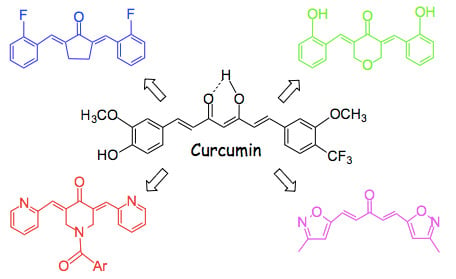
Eliminating the Heart from the Curcumin Molecule: Monocarbonyl Curcumin Mimics (MACs)
Abstract
:1. Introduction


2. Structural Diversity
2.1. 2D Diversity

2.2. 3D Diversity

3. Inflammation Control in Vitro and in Vivo by MACs

3.1. NF-κB/TNF-α




3.2. 5-LOX/COX-2

4. Cancer Mediation in Vitro and in Vivo by MACs
4.1. In Vitro Probes of Cancer Cell Lines and Signaling Factors








4.2. In Vivo Cancer Models, Tumor Growth and Regression: MACs

5. Bacterial Growth

6. Tuberculosis


7. Alzheimer’s Disease


8. Malaria

9. Summary
Acknowledgments
Abbreviation
| MACs | monocarbonyl analogs of curcumin |
| Aβ | amyloid β |
| AD | Alzheimer’s Disease |
| AKT | protein kinase B |
| ALR | aldose reductase |
| AP-1 | the activator protein 1 |
| ATP | adenosine triphosphate |
| COX-2 | cyclooxygenase |
| DNMT | DNA methyltransferase |
| ERK | extracellular-signal regulated kinase |
| HIF | hypoxia inducible factor |
| HSP | heat shock protein |
| HUVEC | human umbilical vein endothelial cells |
| IL | interleukin |
| i.p. | intraperitoneal |
| i.v. | intravenous |
| JNK | c-Jun N-terminal kinase |
| MAPK | mitogen-activated protein kinase |
| MID | minimum inhibitory concentration |
| MTD | maximum tolerance dose |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| LOX | lipoxygenase |
| LPS | lipopolysaccharide |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| NCI | National Cancer Institute |
| NF-κB | nuclear factor κB |
| NO | nitric oxide |
| NSAID | nonsteroidal anti-inflammatory drug |
| ODC | ornithine decarboxylase |
| p38 | stress-activated protein kinase |
| PGE | prostaglandine E |
| p.o. | per os (by mouth) |
| RAW264.7 | mouse macrophage-like cell line |
| ROS | reactive oxygen species |
| SAR | structure-activity relationship |
| STAT | signal transducer and activator of transcription |
| TB | tuberculosis |
| TF | tissue factor |
| TNF | tumor necrosis factor |
| TPA | 12-O-tetradecanoyl-13-acetate |
| VEGF | vascular endothelial growth factor |
| VHL | von Hippel-Lindau |
Conflicts of Interest
References and Notes
- Li, S.; Yuan, W.; Deng, G.; Wang, P.; Yang, P.; Aggarwal, B.B. Chemical Composition and Product Quality Control of Turmeric (Curcuma longa L.). Pharm. Crops 2011, 2, 28–54. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ (accessed on 27 October 2014).
- Mishra, S.B.; Mukerjee, A.; Singh, S. Turmeric: A time tested folk medicine with Ayurvedic perspective. J. Pharm. Biomed. Res. 2011, 1, 67–70. [Google Scholar]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Ji, H.-F. The pharmacology of curcumin: Is it the degradation products? Trends Mol. Med. 2012, 18, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Gordon, O.N.; Schneider, C. Vanillin and ferulic acid: not the major degradation products of curcumin. Trends Mol. Med. 2012, 18, 361–363. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Ji, H.-F. Low stability remedies the low bioavailability of curcumin. Trends Mol. Med. 2012, 18, 363–364. [Google Scholar] [CrossRef]
- Liang, G.; Shao, L.; Wang, Y.; Zhao, C.; Chu, Y.; ** novel antibacterial drugs. Biochem. J. 2009, 423, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Plaza, A.; Keffer, J.L.; Bifulco, G.; Lloyd, J.R.; Bewley, C.A. Chrysophaentins A–H, antibacterial bisdiarylbutene macrocycles that inhibit the bacterial cell division protein FtsZ. J. Am. Chem. Soc. 2010, 132, 9069–9077. [Google Scholar] [CrossRef] [PubMed]
- Chatterji, B.P.; **dal, B.; Srivastava, S.; Panda, D. Microtubules as antifungal and antiparasitic drug targets. Expert Opin. Ther. Pat. 2011, 21, 167–186. [Google Scholar] [CrossRef] [PubMed]
- Liang, G.; Yang, S.; Jiang, L.; Zhao, Y.; Shao, L.; **ao, J.; Ye, F.; Li, Y.; Li, X. Synthesis and anti-bacterial properties of mono-carbonyl analogues of curcumin. Chem. Pharm. Bull. 2008, 56, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Selvakumar, B.; Venkataraman, R. Synthesis and Biological Evaluation of Some Curcumin Analogs and Their Derivatives. Rasāyan J. Chem. 2010, 3, 260–265. [Google Scholar]
- Vilekar, P.; Ding, C.; Lagisetty, P.; Awasthi, V.; Awasthi, S. Antibacterial Activity of Synthetic Curcumin Derivatives: 3,5-Bis(benzylidene)-4-Piperidone (EF24) and EF24-Dimer Linked via Diethylenetriaminepentacetic Acid (EF2DTPA). Appl. Biochem. Biotechnol. 2014, 172, 3363–3373. [Google Scholar] [CrossRef] [PubMed]
- Tuberculosis. WHO. Available online: http://www.who.int/mediacentre/factsheets/fs104/en/ (accessed on 26 September 2014).
- Treatment. Centers for Disease Control and Prevention. Available online: http://www.cdc.gov/tb/topic/treatment/default.htm (accessed on 25 September 2014).
- How many TB Cases have been Successfully Treated? WHO. Available online: http://www.who.int/gho/tb/epidemic/treatment/en/ (accessed on 27 September 2014).
- Drug-resistant Tuberculosis Now at Record Levels. WHO. Available online: http://www.who.int/mediacentre/news/releases/2010/drug_resistant_tb_20100318/en/ (accessed on 27 September 2014).
- Schraufstätter, E.; Bernt, H. Antibacterial Action of Curcumin and Related Compounds. Nature 1949, 164, 456–457. [Google Scholar] [CrossRef] [PubMed]
- Pasca, M.R.; Guglierame, P.; Arcesi, F.; Bellinzoni, M.; Rossi, E.D.; Riccardi, G. Rv2686c-Rv2687c-Rv2688c, an ABC Fluoroquinolone Efflux Pump in Mycobacterium Tuberculosis. Antimicrob. Agents Chemother. 2004, 48, 3175–3178. [Google Scholar] [CrossRef] [PubMed]
- Silva, P.E.A.; Bigi, F.; Santangelo, M.D.L.P.; Romano, M.I.; Martin, C.; Cataldi, A.; Ainsa, J.A. Characterization of P55, a Multidrug Efflux Pump in Mycobacterium Bovis and Mycobacterium Tuberculosis. Antimicrob. Agents Chemother. 2001, 45, 800–804. [Google Scholar] [CrossRef] [PubMed]
- Balganesh, M.; Dinesh, N.; Sharma, S.; Kuruppath, S.; Nair, A.V.; Sharma, U. Efflux Pumps of Mycobacterium Tuberculosis Play a Significant Role in Antituberculosis Activity of Potential Drug Candidates. Antimicrob. Agents Chemother. 2012, 56, 2643–2651. [Google Scholar] [CrossRef] [PubMed]
- Dube, D.; Kukshal, V.; Srivastava, S.K.; Tripathi, R.P.; Ramachandran, R. NAD+-dependent DNA Ligase (Rv3014c) From M. tuberculosis: Strategies for Inhibitor Design. Med. Chem. Res. 2008, 17, 189–198. [Google Scholar] [CrossRef]
- Takeuchi, T.; Ishidoh, T.; Iijima, H.; Kuriyama, I.; Shimazaki, N.; Koiwai, O.; Kuramochi, K.; Kobayashi, S.; Sugawara, F.; Sakaguchi, K.; et al. Structural Relationship of Curcumin Derivatives Binding to the BRCT Domain of Human DNA Polymerase Lambda. Genes Cells 2006, 11, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, D.K.; Saikia, D.; Tiwari, R.; Ojha, S.; Shanker, K.; Kumar, J.K.; Gupta, A.K.; Tandon, S.; Negi, A.S.; Khanuja, S.P.S. Demethoxycurcumin and its semisynthetic analogues as antitubercular Agents. Planta Med. 2008, 74, 1828–1831. [Google Scholar] [CrossRef] [PubMed]
- Marathe, S.A.; Dasgupta, I.; Gnanadhas, D.P.; Chakravortty, D. Multifaceted Roles of Curcumin: Two Sides of a Coin! Expert Opin. Biol. Ther. 2011, 11, 1485–1499. [Google Scholar] [CrossRef] [PubMed]
- Dimmock, J.R.; Kandepu, N.M.; Das, U.; Zello, G.A.; Nienaber, K.H. Antimycobacterial arylidenecyclohexanones and related Mannich bases. Pharmazie 2004, 59, 502–505. [Google Scholar] [PubMed]
- Das, S.; Das, U.; Bandy, B.; Gorecki, D.K.J.; Dimmock, J.R. 2-[4-(4-Methoxyphenylcarbonyloxy)benzylidene]-6-dimethylaminomethyl cyclohexanone hydrochloride: A Mannich base which inhibits the growth of some drug-resistant strains of Mycobacterium tuberculosis. Pharmazie 2010, 65, 849–850. [Google Scholar]
- Das, U.; Das, S.; Bandy, B.; Sables, J.P.; Dimmock, J.R. N-Aroyl-3,5-bis(benzylidene)-4-piperidones: A novel class of antimycobacterial agents. Bioorg. Med. Chem. 2008, 16, 3602–3607. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, P.; Reeves, A.Z.; Powell, K.R.; Napier, R.J.; Swimm, A.I.; Sun, A.; Giesler, K.; Bommarius, B.; Shinnick, T.M.; Snyder, J.P.; et al. Monocarbonyl analogs of curcumin inhibit growth of antibiotic sensitive and resistant strains of Mycobacterium tuberculosis. Eur. J. Med. Chem. 2015, in press. [Google Scholar]
- Kim, J.-J.; Lee, H.-M.; Shin, D.-M.; Kim, W.; Yuk, J.-M.; **, H.S.; Lee, S.-H.; Cha, G.-H.; Kim, J.-M.; Lee, Z.-W.; et al. Host Cell Autophagy Activated by Antibiotics Is Required for Their Effective Antimycobacterial Drug Action. Cell Host Microbe 2012, 11, 457–468. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Bai, Y.; Sun, A.; Liotta, D.; Snyder, J.P.; Fu, H.; Huang, B. Autophagy and apoptosis in hepatocellular carcinoma induced by EF25-(GSH)2: A novel curcumin analog. PLoS One 2014, 9, e107876. [Google Scholar] [CrossRef] [PubMed]
- Dementia. WHO. Available online: http://www.who.int/mediacentre/factsheets/fs362/en/ (accessed on 27 September 2014).
- Anand, P.; Thomas, S.G.; Kunnumakkara, A.B.; Sundaram, C.; Harikumar, K.B.; Sung, B.; Tharakan, S.T.; Misra, K.; Priyadarsini, I.K.; Rajasekharan, K.N.; et al. Biological Activities of Curcumin and Its Analogues (Congeners) Made by Man and Mother Nature. Biochem. Pharmacol. 2008, 76, 1590–1611. [Google Scholar] [CrossRef] [PubMed]
- Yang, F. Curcumin Inhibits Formation of Amyloid Oligomers and Fibrils, Binds Plaques, and Reduces Amyloid in Vivo. J. Biol. Chem. 2004, 280, 5892–5901. [Google Scholar] [CrossRef] [PubMed]
- A Pilot Study of Curcumin and Ginkgo for Treating Alzheimer’s Disease. Available online: http://clinicaltrials.gov/ct2/show/NCT00164749?term=curcumin+and+alzheimers+disease&rank=1 (accessed on 26 September 2012).
- Ringman, J.M.; Frautschy, S.A.; Cole, G.M.; Masterman, D.L.; Cummings, J.L. A potential role of the curry spice curcumin in Alzheimer’s disease. Curr. Alzheimer Res. 2005, 2, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Alloza, M.; Borrelli, L.A.; Rozkalne, A.; Hyman, B.T.; Bacskai, B.J. Curcumin labels amyloid pathology in vivo, disrupts existing plaques, and partially restores distorted neurites in an Alzheimer mouse model. J. Neurochem. 2007, 102, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Landau, M.; Sawaya, M.R.; Faull, K.F.; Laganowsky, A.; Jiang, L.; Sievers, S.A.; Liu, J.; Barrio, J.R.; Eisenberg, D. Towards a pharmacophore for Amyloid. PLoS Biol. 2011, 9, e1001080. [Google Scholar] [CrossRef] [PubMed]
- Yanagisawa, D.; Shirai, N.; Amatsubo, T.; Taguchi, H.; Hirao, K.; Urushitani, M.; Morikawa, S.; Inubushi, T.; Kato, M.; Kato, F.; et al. Relationship between the tautomeric structures of curcumin derivatives and their Aβ-binding activities in the context of therapies for Alzheimer’s disease. Biomaterials 2010, 32, 4179–4185. [Google Scholar] [CrossRef]
- Payton, F.; Peter Sandusky, P.; Alworth, W.L. NMR Study of the solution structure of curcumin. J. Nat. Prod. 2007, 70, 143–146. [Google Scholar] [CrossRef] [PubMed]
- Orlando, R.A.; Gonzales, A.M.; Royer, R.E.; Deck, L.M.; Vander Jagt, D.L. A chemical analog of curcumin as an improved inhibitor of amyloid Abeta oligomerization. PLoS One 2012, 7, e31869. [Google Scholar] [CrossRef] [PubMed]
- Markesbery, W.R. Oxidative stress hypothesis in Alzheimer’s disease. Free Radic. Biol. Med. 1997, 23, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Ao, G.; Chu, X.; Ji, Y.; Wang, J. Antioxidant properties and PC12 cell protective effects of a novel curcumin analogue (2E,6E)-2,6-bis(3,5-Dimethoxybenzylidene)cyclohexanone (MCH). Int. J. Mol. Sci. 2014, 15, 3970–3988. [Google Scholar] [CrossRef] [PubMed]
- Reddy, R.C.; Vatsala, P.G.; Keshamouni, V.G.; Padmanaban, G.; Rangarajan, P.N. Curcumin for malaria therapy. Biochem. Biophys. Res. Commun. 2005, 326, 472–474. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Karmodiya, K.; Surolia, N.; Surolia, A. Synthesis and exploration of novel curcumin analogues as anti-malarial agents. Bioorg. Med. Chem. 2008, 16, 2894–2902. [Google Scholar] [CrossRef] [PubMed]
- Manohar, S.; Khan, S.I.; Kandi, S.K.; Raj, K.; Sun, G.; Yang, X.; Molina, A.D.C.; Ni, N.; Wang, B.; Rawat, D.S. Synthesis, antimalarial activity and cytotoxic potential of new monocarbonyl analogues of curcumin. Bioorg. Med. Chem. Lett. 2013, 23, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Reference 27 (Bairwa et al. 2014) overviews a sampling of recent SAR studies.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shetty, D.; Kim, Y.J.; Shim, H.; Snyder, J.P. Eliminating the Heart from the Curcumin Molecule: Monocarbonyl Curcumin Mimics (MACs). Molecules 2015, 20, 249-292. https://doi.org/10.3390/molecules20010249
Shetty D, Kim YJ, Shim H, Snyder JP. Eliminating the Heart from the Curcumin Molecule: Monocarbonyl Curcumin Mimics (MACs). Molecules. 2015; 20(1):249-292. https://doi.org/10.3390/molecules20010249
Chicago/Turabian StyleShetty, Dinesh, Yong Joon Kim, Hyunsuk Shim, and James P. Snyder. 2015. "Eliminating the Heart from the Curcumin Molecule: Monocarbonyl Curcumin Mimics (MACs)" Molecules 20, no. 1: 249-292. https://doi.org/10.3390/molecules20010249