Synthesis, Antibacterial Activities, Mode of Action and Acute Toxicity Studies of New Oxazolidinone-Fluoroquinolone Hybrids
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
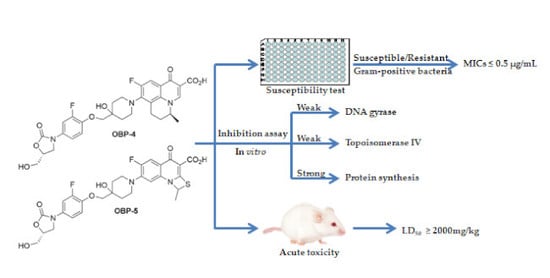
2.2. Antibacterial Activities
2.2.1. In Vitro Activities of Oxazolidinone-Fluoroquinolone Hybrids
2.2.2. In Vitro Activities of Hybrids OBP-4 and OBP-5 against Clinical Isolates
2.3. Mode of Action of Hybrids OBP-4 and OBP-5
2.3.1. In Vitro Enzyme Assays of Hybrids OBP-4 and OBP-5
2.3.2. Molecular Docking of Hybrids OBP-4 and OBP-5
2.4. Acute Toxicity Test
3. Materials and Methods
3.1. General
3.2. Chemistry
3.2.1. Preparation of Hybrids OBP-1, OBP-2 and OBP-3
3.2.2. Preparation of Hybrid OBP-4
3.2.3. Preparation of Hybrid OBP-5
3.2.4. Preparation of Hybrids OBP-6 and OBP-7
3.3. Antibacterial Activities
3.3.1. Bacterial Strains
3.3.2. Susceptibility Testing
3.4. Modes of Action
3.4.1. DNA Gyrase Supercoiling Assay
3.4.2. Topo IV Relaxation Assay
3.4.3. In Vitro Transcription/Translation Assay
3.4.4. Molecular Docking Stusy
3.5. Acute Toxicity Test
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zinner, S.H. Antibiotic use: Present and future. New Microbiol. 2007, 30, 321–325. [Google Scholar] [PubMed]
- Li, B.; Webster, T.J. Bacteria antibiotic resistance: New challenges and opportunities for implant-associated orthopedic infections. J. Orthop. Res. 2018, 36, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Vivas, R.; Barbosa, A.A.T.; Dolabela, S.S.; Jain, S. Multidrug-resistant bacteria and alternative methods to control them: An overview. Microb. Drug Resist. 2019. [Google Scholar] [CrossRef]
- Watkins, R.R.; Bonomo, R.A. Overview: Global and local impact of antibiotic resistance. Infect. Dis. Clin. N. Am. 2016, 30, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Ciorba, V.; Odone, A.; Veronesi, L.; Pasquarella, C.; Signorelli, C. Antibiotic resistance as a major public health concern: Epidemiology and economic impact. Ann. Iq. 2015, 27, 562–579. [Google Scholar]
- Thabit, A.K.; Crandon, J.L.; Nicolau, D.P. Antimicrobial resistance: Impact on clinical and economic outcomes and the need for new antimicrobials. Expert Opin. Pharmacother. 2015, 16, 159–177. [Google Scholar] [CrossRef] [PubMed]
- Tillotson, G.S.; Zinner, S.H. Burden of antimicrobial resistance in an era of decreasing susceptibility. Expert Rev. Anti Infect. Ther. 2017, 15, 663–676. [Google Scholar] [CrossRef]
- Sender, R.; Fuchs, S.; Milo, R. Revised Estimates for the number of human and bacteria cells in the body. PLoS. Biol. 2016, 14, e1002533. [Google Scholar] [CrossRef]
- Parkes, A.L.; Yule, I.A. Hybrid antibiotics—Clinical progress and novel designs. Expert Opin. Drug Discov. 2016, 11, 665–680. [Google Scholar] [CrossRef]
- Barbachyn, M.R. Recent advances in the discovery of hybrid antibacterial agents. Annu. Rep. Med. Chem. 2008, 43, 281–290. [Google Scholar]
- Robertson, G.T.; Bonventre, E.J.; Doyle, T.B.; Du, Q.; Duncan, L.; Morris, T.W.; Roche, E.D.; Yan, D.; Lynch, A.S. In vitro evaluation of CBR-2092, a novel rifamycin-quinolone hybrid antibiotic: Studies of the mode of action in Staphylococcus aureus. Antimicrob. Agents. Chemother. 2008, 52, 2313–2323. [Google Scholar] [CrossRef]
- Pokrovskaya, V.; Belakhov, V.; Hainrichson, M.; Yaron, S.; Baasov, T. Design, synthesis, and evaluation of novel fluoroquinolone-aminoglycoside hybrid antibiotics. J. Med. Chem. 2009, 52, 2243–2254. [Google Scholar] [CrossRef]
- Blais, J.; Lewis, S.R.; Krause, K.M.; Benton, B.M. Antistaphylococcal activity of TD-1792, a multivalent glycopeptide-cephalosporin antibiotic. Antimicrob. Agents. Chemother. 2012, 56, 1584–1587. [Google Scholar] [CrossRef]
- Louie, T.; Nord, C.E.; Talbot, G.H.; Wilcox, M.; Gerding, D.N.; Buitrago, M.; Kracker, H.; Charef, P.; Cornely, O.A. Multicenter, double-blind, randomized, phase 2 study evaluating the novel antibiotic cadazolid in patients with Clostridium difficile infection. Antimicrob. Agents. Chemother. 2015, 59, 6266–6273. [Google Scholar] [CrossRef] [PubMed]
- Pokrovskaya, V.; Baasov, T. Dual-acting hybrid antibiotics: A promising strategy to combat bacterial resistance. Expert Opin. Drug Discov. 2010, 5, 883–902. [Google Scholar] [CrossRef]
- Shaw, K.J.; Barbachyn, M.R. The oxazolidinones: Past, present, and future. Ann. N. Y. Acad. Sci. 2011, 1241, 48–70. [Google Scholar] [CrossRef]
- Bender, J.K.; Fleige, C.; Lange, D.; Klare, I.; Werner, G. Rapid emergence of highly variable and transferable oxazolidinone and phenicol resistance gene optrA in German Enterococcus spp. clinical isolates. Int. J. Antimicrob. Agents 2018, 52, 819–827. [Google Scholar] [CrossRef] [PubMed]
- De Lastours, V.; Fantin, B. Resistance to fluoroquinolones in 2013: What are the consequences in internal medicine? Rev. Med. Interne 2014, 35, 601–608. [Google Scholar] [CrossRef]
- Hubschwerlen, C.; Specklin, J.L.; Sigwalt, C.; Schroeder, S.; Locher, H.H. Design, synthesis and biological evaluation of oxazolidinone-quinolone hybrids. Bioorg. Med. Chem. 2003, 11, 2313–2319. [Google Scholar] [CrossRef]
- Hubschwerlen, C.; Specklin, J.L.; Baeschlin, D.K.; Borer, Y.; Haefeli, S.; Sigwalt, C.; Schroeder, S.; Locher, H.H. Structure-activity relationship in the oxazolidinone-quinolone hybrid series: Influence of the central spacer on the antibacterial activity and the mode of action. Bioorg. Med. Chem. Lett. 2003, 13, 4229–4233. [Google Scholar] [CrossRef] [PubMed]
- Sabine Schubert, A.D. Low propensity for development of resistance to MCB3681, the active moiety of oxaquin (MCB3837), in Gram-positive bacteria with vancomycin-, linezolid-, methicillin- and/or ciprofloxacin resistances. In Proceedings of the 46th ICAAC, San Francisco, CA, USA, 27–30 September 2006. [Google Scholar]
- Locher, H.H.; Caspers, P.; Bruyere, T.; Schroeder, S.; Pfaff, P.; Knezevic, A.; Keck, W.; Ritz, D. Investigations of the mode of action and resistance development of cadazolid, a new antibiotic for treatment of Clostridium difficile infections. Antimicrob. Agents. Chemother. 2014, 58, 901–908. [Google Scholar] [CrossRef] [PubMed]
- Gerding, D.N.; Hecht, D.W.; Louie, T.; Nord, C.E.; Talbot, G.H.; Cornely, O.A.; Buitrago, M.; Best, E.; Sambol, S.; Osmolski, J.R.; et al. Susceptibility of Clostridium difficile isolates from a Phase 2 clinical trial of cadazolid and vancomycin in C. difficile infection. J. Antimicrob. Chemother. 2016, 31, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Correia, S.; Poeta, P.; Hebraud, M.; Capelo, J.L.; Igrejas, G. Mechanisms of quinolone action and resistance: Where do we stand? J. Med. Microbiol. 2017, 66, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Hiasa, H.; DiGate, R.J.; Marians, K.J. Decatenating activity of Escherichia coli DNA gyrase and topoisomerases I and III during oriC and pBR322 DNA replication in vitro. J. Biol. Chem. 1994, 269, 2093–2099. [Google Scholar] [PubMed]
- Baldoni, D.; Gutierrez, M.; Timmer, W.; Dingemanse, J. Cadazolid, a novel antibiotic with potent activity against Clostridium difficile: Safety, tolerability and pharmacokinetics in healthy subjects following single and multiple oral doses. J. Antimicrob. Chemother. 2014, 69, 706–714. [Google Scholar] [CrossRef] [PubMed]
- Clewley, R.G.; Cross, G.G.; Fischer, A.; Henderson, G.N. Formation of 4-halo-4- nitrocyclohexa-2,5-dienones on nitration of p-halophenols and p-halophenyl acetates. Tetrahedron 1989, 45, 1299–1310. [Google Scholar] [CrossRef]
- Wessig, P.; Möllnitz, K.; Eiserbeck, C. Oligospiroketals as novel molecular rods. Chemistry 2007, 13, 4859–4872. [Google Scholar] [CrossRef]
- Yang, Y.; Ji, R.; Hu, Z.; Chen, K. A new practical route to stereospecific synthesis of (S)-(-)-ofloxacin. Acta Pharm. Sin. 1998, 33, 828–831. [Google Scholar]
- Kose, E.; Karabacak, M.; Atac, A. The spectroscopic and quantum chemical studies of 3,4-difluoroaniline. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2015, 143, 265–280. [Google Scholar] [CrossRef] [PubMed]
- Segawa, J.; Kitano, M.; Kazuno, K.; Matsuoka, M.; Shirahase, I.; Ozaki, M.; Matsuda, M.; Tomii, Y.; Kise, M. Studies on pyridonecarboxylic acids. 1. Synthesis and antibacterial evaluation of 7-substituted-6-halo-4-oxo-4H-[1,3]thiazeto[3,2-a]quinoline-3-carboxylic acids. J. Med. Chem. 1992, 35, 4727–4738. [Google Scholar] [CrossRef]
- Matuszak, N.; Muccioli, G.G.; Labar, G.; Lambert, D.M. Synthesis and in vitro evaluation of N-substituted maleimide derivatives as selective monoglyceride lipase inhibitors. J. Med. Chem. 2009, 52, 7410–7420. [Google Scholar] [CrossRef] [PubMed]
- Wan, P.H.; Kee, J.L. Baylis-Hillman route to several quinolone antibiotic intermediates. Chem. Inform. 2006, 37, 963–968. [Google Scholar]
- Egawa, H.; Kataoka, M.; Shibamori, K.I.; Miyamato, T.; Nakano, J.; Matsumoto, J.I. A new synthetic route to 7-halo-1-cyclopropyl-6-fluoro-1,4-dihydro-4oxoquinoline-3- carboxylic acid, an intermediate for the synthesis of quinolone antibacterial agents. J. Heterocyclic. Chem. 1987, 18, 181–185. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; 26th Informational Supplement; CLSI document M100-S26; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2016. [Google Scholar]
- Eyal, Z.; Matzov, D.; Krupkin, M.; Wekselman, I.; Paukner, S.; Zimmerman, E.; Rozenberg, H.; Bashan, A.; Yonath, A. Structural insights into species-specific features of the ribosome from the pathogen Staphylococcus aureus. Proc. Natl. Acad. Sci. USA 2015, 112, E5805–E5814. [Google Scholar] [CrossRef]
- Taher, E.S.; Ibrahim, T.S.; Fares, M.; Al-Mahmoudy, A.M.M.; Radwan, A.F.; Orabi, K.Y.; El-Sabbagh, O.I. Novel benzenesulfonamide and 1,2-benzisothiazol-3(2H)-one-1,1- dioxide derivatives as potential selective COX-2 inhibitors. Eur. J. Med. Chem. 2019, 171, 372–382. [Google Scholar] [CrossRef]
- Friesner, R.A.; Murphy, R.B.; Repasky, M.P.; Frye, L.L.; Greenwood, J.R.; Halgren, T.A.; Sanschagrin, P.C.; Mainz, D.T. Extra precision glide: Docking and scoring incorporating a model of hydrophobic enclosure for protein-ligand complexes. J. Med. Chem. 2006, 49, 6177–6196. [Google Scholar] [CrossRef]
- OECD 425 Guideline for Testing of Chemical. Acute Oral Toxicity-Up-and-Down Procedure; OECD: Paris, France, 2001. [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |










| Organism (No.) | MIC (µg/mL) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| OBP-1 | OBP-2 | OBP-3 | OBP-4 | OBP-5 | OBP-6 | OBP-7 | CZD | LZD | MXF | |
| S. pneumoniae (ATCC49619) | 0.25 | 0.25 | 0.5 | 0.031 | 0.062 | 0.5 | 2 | 0.062 | 1 | 0.062 |
| S. agalactiaeR (13B317) | 0.5 | 0.25 | 0.5 | 0.031 | 0.031 | 2 | 4 | 0.125 | 1 | 4 |
| E. faecalis (ATCC29212) | 1 | 1 | 1 | 0.25 | 1 | 4 | 1 | 0.5 | 4 | 0.125 |
| VRE (13B330) | 1 | 1 | 2 | 0.25 | 0.5 | 64 | 128 | 0.25 | 2 | 64 |
| MRSA (13B262) | 2 | 1 | 2 | 0.5 | 0.5 | 64 | 128 | 0.5 | 4 | 8 |
| S. aureus (ATCC29213) | 1 | 0.25 | 2 | 0.016 | 0.25 | 0.062 | 0.25 | 0.5 | 4 | 0.031 |
| H. influenzae (ATCC49247) | 1 | 0.5 | 2 | 0.25 | 0.25 | 0.016 | 0.031 | 1 | 8 | 0.008 |
| P. Aeruginosa (ATCC27853) | 64 | 64 | 128 | 64 | 64 | 16 | 64 | 64 | 128 | 4 |
| E. coli (ATCC25922) | 4 | 4 | 64 | 4 | 4 | 1 | 0.5 | 8 | 128 | 0.016 |
| Organism (No. Tested) | Agents | MIC (µg/mL) | CLSI a %S/%R | ||
|---|---|---|---|---|---|
| MIC50 | MIC90 | Range | |||
| S. aureus (46) | OBP-4 | ≤0.0625 | 0.125 | ≤0.0625–0.25 | -/- |
| OBP-5 | ≤0.0625 | ≤0.0625 | ≤0.0625–0.25 | -/- | |
| ENR | 2 | 4 | ≤0.0625–8 | ||
| CIP | 16 | 32 | ≤0.0625–32 | 37.9/62.1 | |
| GAFX | 1 | 2 | ≤0.0625–2 | 45.5/42.4 | |
| NDFX | 0.5 | 0.5 | ≤0.0625–2 | ||
| LZD | 2 | 2 | 0.5–2 | 100.0/- | |
| VAN | 0.5 | 0.5 | 0.25–0.5 | 100.0/- | |
| S. haemolyticus (32) | OBP-4 | 0.125 | 0.125 | ≤0.0625–0.5 | -/- |
| OBP-5 | ≤0.0625 | ≤0.0625 | ≤0.0625–0.125 | -/- | |
| ENR | ≤0.0625 | 0.5 | ≤0.0625–8 | ||
| CIP | 0.25 | 1 | 0.125–16 | 96.2/3.8 | |
| GAFX | 0.125 | 0.25 | ≤0.0625–2 | 96.2/3.8 | |
| NDFX | ≤0.0625 | 0.5 | ≤0.0625–4 | ||
| LZD | 0.5 | 2 | 0.125–8 | 96.2/3.8 | |
| VAN | 0.5 | 2 | ≤0.0625–4 | 96.2/- | |
| S. epidermidis (10) | OBP-4 | 0.125 | 0.25 | ≤0.0625–0.25 | -/- |
| OBP-5 | ≤0.0625 | 0.125 | ≤0.0625–0.125 | -/- | |
| ENR | 2 | 4 | 0.125–4 | ||
| CIP | 8 | 16 | 0.25–16 | 20.0/60.0 | |
| GAFX | 1 | 2 | 0.125–2 | 30.0/50.0 | |
| NDFX | 1 | 2 | ≤0.0625–2 | ||
| LZD | 1 | 1 | 0.5–1 | 100.0/- | |
| VAN | 1 | 2 | 1–2 | 100.0/- | |
| S. suis (9) | OBP-4 | ≤0.0625 | 0.25 | ≤0.0625–2 | -/- |
| OBP-5 | ≤0.0625 | 0.125 | ≤0.0625–2 | -/- | |
| ENR | 0.25 | 4 | ≤0.0625–8 | ||
| CIP | 0.25 | 8 | 0.125–16 | ||
| GAFX | ≤0.0625 | 4 | ≤0.0625–4 | ||
| NDFX | ≤0.0625 | 2 | ≤0.0625–4 | ||
| LZD | 0.5 | 4 | 0.5–4 | ||
| VAN | 0.25 | 2 | 0.125–2 | ||
| S. pneumoniae (13) | OBP-4 | 0.25 | 0.25 | ≤0.0625–0.5 | -/- |
| OBP-5 | ≤0.0625 | 0.25 | ≤0.0625–0.25 | -/- | |
| ENR | 32 | >32 | 2–>32 | ||
| CIP | 16 | >32 | 4–>32 | -/100.0 | |
| GAFX | 4 | 32 | 4–32 | 7.7/92.3 | |
| NDFX | 16 | >32 | 8–>32 | ||
| LZD | 0.5 | 0.5 | 0.5–4 | 76.9/- | |
| VAN | 1 | 32 | 0.5–>32 | 53.8/- | |
| E. faecalis (72) | OBP-4 | 0.25 | 0.25 | ≤0.0625–1 | -/- |
| OBP-5 | 0.125 | 0.125 | ≤0.0625–0.5 | -/- | |
| ENR | 0.5 | 0.5 | ≤0.0625–2 | ||
| CIP | 0.5 | 1 | 0.125–2 | 97.3/- | |
| GAFX | 0.25 | 0.5 | 0.125–2 | 94.6/- | |
| NDFX | 0.25 | 1 | 0.125–2 | ||
| LZD | 2 | 2 | 0.5–8 | 97.3/2.7 | |
| E. faecium (47) | OBP-4 | 0.5 | 0.25 | 0.25–2 | -/- |
| OBP-5 | 0.25 | 0.25 | ≤0.0625–1 | -/- | |
| ENR | 2 | 32 | 0.5–>32 | ||
| CIP | 2 | 32 | 0.25–>32 | 51.7/37.9 | |
| GAFX | 1 | 8 | 0.25–8 | 86.2/13.8 | |
| NDFX | 2 | 8 | 0.5–16 | ||
| LZD | 2 | 4 | 0.5–16 | 82.8/13.8 | |
| VAN | 2 | 4 | 0.5–4 | 100.0/- | |
| H. parasuis (34) | OBP-4 | 1 | 4 | ≤0.0625–4 | -/- |
| OBP-5 | 0.5 | 2 | ≤0.0625–4 | -/- | |
| ENR | ≤0.0625 | 1 | ≤0.0625–1 | ||
| CIP | ≤0.0625 | 1 | ≤0.0625–1 | ||
| GAFX | ≤0.0625 | 1 | ≤0.0625–1 | ||
| NDFX | ≤0.0625 | 1 | ≤0.0625–1 | ||
| LZD | 16 | >32 | 8–>32 | ||
| VAN | 8 | >32 | 2–>32 | ||
| Organism (No. Tested) | Agents | No. of Strains (Cumulative %) Inhibited at Each MIC (µg/mL) | ||||||
|---|---|---|---|---|---|---|---|---|
| ≤0.0625 | 0.125 | 0.25 | 0.5 | 1 | 2 | 4 | ||
| S. aureus (46) | OBP-4 | 26(56.5) | 17(93.5) | 3(100.0) | ||||
| OBP-5 | 42(91.3) | 3(97.8) | 1(100.0) | |||||
| S. haemolyticus (32) | OBP-4 | 14(43.8) | 16(93.8) | 2(100.0) | ||||
| OBP-5 | 30(93.4) | 2(100.0) | ||||||
| S. epidermidis (10) | OBP-4 | 2(20.0) | 4(60.0) | 4(100.0) | ||||
| OBP-5 | 8(80.0) | 2(20.0) | ||||||
| S. suis (9) | OBP-4 | 5(55.6) | 2(77.8) | 1(88.9) | 1(100) | |||
| OBP-5 | 5(55.6) | 3(88.9) | 1(100) | |||||
| S. pneumoniae (13) | OBP-4 | 2(15.4) | 3(38.5) | 7(92.3) | 1(100.0) | |||
| OBP-5 | 7(53.8) | 4(84.6) | 2(100.0) | |||||
| E. faecalis (72) | OBP-4 | 9(12.5) | 17(36.1) | 43(95.8) | 2(98.6) | 1(100.0) | ||
| OBP-5 | 17(23.6) | 49(91.7) | 5(98.6) | 1(100.0) | ||||
| E. faecium (47) | OBP-4 | 8(17.2) | 37(95.7) | 1(97.9) | 1(100.0) | |||
| OBP-5 | 3(6.4) | 7(21.3) | 33(91.5) | 3(97.9) | 1(100.0) | |||
| H. parasuis (34) | OBP-4 | 4(11.8) | 3(20.6) | 5(35.3) | 9(61.8) | 6(79.4) | 7(100.0) | |
| OBP-5 | 5(14.7) | 1(17.6) | 4(29.4) | 6(47.1) | 7(67.6) | 10(97.1) | 1(100.0) | |
| Organism | MIC (µg/mL) | |||||||
|---|---|---|---|---|---|---|---|---|
| OBP-4 | OBP-5 | ENR | CIP | GAFX | NDFX | LZD | VAN | |
| MRSA | 0.25 | 0.125 | 0.25 | 0.125 | 0.125 | ≤0.0625 | 4 | 1 |
| MRSE | ≤0.0625 | ≤0.0625 | ≤0.0625 | 0.125 | ≤0.0625 | 0.125 | 0.25 | 1 |
| S. aureusa | 0.125 | ≤0.0625 | 2 | 32 | 2 | 0.5 | 2 | 0.25 |
| S. haemolyticusa | 0.5 | ≤0.0625 | 8 | 16 | 2 | 4 | 8 | 2 |
| S. pneumoniaeb | 0.25 | ≤0.0625 | >32 | >32 | >32 | >32 | 0.5 | >32 |
| E. faecalissc | 1 | 0.25 | 1 | 1 | 0.5 | 1 | 8 | 2 |
| E. faeciumd | 0.5 | 0.125 | >32 | >32 | 8 | 16 | 16 | 4 |
| E. faeciumb | 0.5 | 0.25 | >32 | >32 | 16 | 32 | 2 | 16 |
| Compd. | IC50 (μM) | ||
|---|---|---|---|
| DNA Gyrase | Topo IV | Inhib. of Protein Synthesis | |
| OBP-4 | 1–5 | 10–15 | 5 |
| OBP-5 | 20 | >40 | 2 |
| CIP | ˂0.75 | 3–6 | NT |
| LZD | NT | NT | 5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, L.; Shao, L.; Li, J.; Cui, H.; Li, B.; Zhou, X.; Lv, P.; Zhang, J. Synthesis, Antibacterial Activities, Mode of Action and Acute Toxicity Studies of New Oxazolidinone-Fluoroquinolone Hybrids. Molecules 2019, 24, 1641. https://doi.org/10.3390/molecules24081641
Liu L, Shao L, Li J, Cui H, Li B, Zhou X, Lv P, Zhang J. Synthesis, Antibacterial Activities, Mode of Action and Acute Toxicity Studies of New Oxazolidinone-Fluoroquinolone Hybrids. Molecules. 2019; 24(8):1641. https://doi.org/10.3390/molecules24081641
Chicago/Turabian StyleLiu, Lili, Li** Shao, **g Li, Haifeng Cui, Bing Li, Xuzheng Zhou, Pengyue Lv, and Jiyu Zhang. 2019. "Synthesis, Antibacterial Activities, Mode of Action and Acute Toxicity Studies of New Oxazolidinone-Fluoroquinolone Hybrids" Molecules 24, no. 8: 1641. https://doi.org/10.3390/molecules24081641