Direct Base-Assisted C‒H Cyclonickelation of 6-Phenyl-2,2′-bipyridine
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Materials
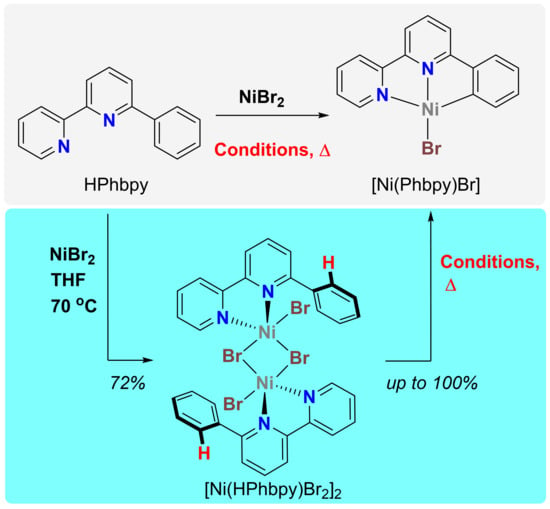
4.2. Cyclonickelation Experiments
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Labinger, J.A. Platinum-Catalyzed C−H Functionalization. Chem. Rev. 2017, 117, 8483–8496. [Google Scholar] [CrossRef] [PubMed]
- Cope, A.C.; Siekman, R.W. Formation of Covalent Bonds from Platinum or Palladium to Carbon by Direct Substitution. J. Am. Chem. Soc. 1965, 87, 3272–3273. [Google Scholar] [CrossRef]
- Cope, A.C.; Friedrich, E.C. Electrophilic Aromatic Substitution Reactions by Platinum(II) and Palladium(II) Chlorides on N,N-Dimethylbenzylamines. J. Am. Chem. Soc. 1968, 90, 909–913. [Google Scholar] [CrossRef]
- Chassot, L.; Mueller, E.; Von Zelewsky, A. cis-Bis(2-phenylpyridine)platinum(II) (CBPPP): A Simple Molecular Platinum Compound. Inorg. Chem. 1984, 23, 4249–4253. [Google Scholar] [CrossRef]
- Constable, E.C.; Henney, R.P.G.; Leese, T.A.; Tocher, D.A. Cyclometallation Reactions of 6-Phenyl-2,2’-bipyridine; a Potential C,N,N-Donor Analogue of 2,2’:6’,2”-Terpyridine. Crystal and Molecular Structure of Dichlorobis(6-phenyl-2,2’-bipyridine)ruthenium(II). J. Chem. Soc., Dalton Trans. 1990, 443–449. [Google Scholar] [CrossRef]
- Constable, E.C.; Henney, R.P.G.; Leese, T.A.; Tocher, D.A. Cyclopalladated and Cycloplatinated Complexes of 6-Phenyl-2,2′-bipyridine: Platinum-Platinum Interactions in the Solid State. J. Chem. Soc. Chem. Commun. 1990, 513–515. [Google Scholar] [CrossRef]
- Berenguer, J.R.; Lalinde, E.; Moreno, M.T. Luminescent cyclometalated-pentafluorophenyl PtII, PtIV and heteropolynuclear complexes. Coord. Chem. Rev. 2018, 366, 69–90. [Google Scholar] [CrossRef]
- Tang, M.-C.; Chan, A.K.-W.; Chan, M.-Y.; Yam, V.W.-W. Platinum and Gold Complexes for OLEDs. Top. Curr. Chem. (Z) 2016, 374, 1–43. [Google Scholar] [CrossRef]
- Kalinowski, J.; Fattori, V.; Cocchi, M.; Williams, J.A.G. Light-emitting devices based on organometallic platinum complexes as emitters. Coord. Chem. Rev. 2011, 255, 2401–2425. [Google Scholar] [CrossRef]
- Williams, J.A.G. Metal complexes of pincer ligands: Excited states, photochemistry, and luminescence. Top. Organomet. Chem. 2011, 40, 89–130. [Google Scholar]
- Yang, S.; Meng, F.; Wu, X.; Yin, Z.; Liu, X.; You, C.; Wang, Y.; Su, S.; Zhu, W. Dinuclear platinum(II) complex dominated by a zig-zag-type cyclometalated ligand: A new approach to realize high-efficiency near infrared emission. J. Mater. Chem. C 2018, 6, 5769–5777. [Google Scholar] [CrossRef]
- Sivchik, V.; Sarker, R.K.; Liu, Z.-Y.; Chung, K.-Y.; Grachova, E.V.; Karttunen, A.J.; Chou, P.-T.; Koshevoy, I.O. Improvement of the Photophysical Performance of Platinum-Cyclometalated Complexes in Halogen-Bonded Adducts. Chem. Eur. J. 2018, 24, 11475–11484. [Google Scholar] [CrossRef]
- Soellner, J.; Strassner, T. The “Enders Triazole” Revisited: Highly Efficient, Blue Platinum(II) Emitters. Organometallics 2018, 37, 1821–1824. [Google Scholar] [CrossRef]
- Schulze, B.; Friebe, C.; Jäger, M.; Görls, H.; Birckner, E.; Winter, A.; Schubert, U.S. PtII Phosphors with Click-Derived 1,2,3-Triazole-Containing Tridentate Chelates. Organometallics 2018, 37, 145–155. [Google Scholar] [CrossRef]
- Sesolis, H.; Chan, C.K.-M.; Gontard, G.; Fu, H.L.-K.; Yam, V.W.-W.; Amouri, H. Dinuclear (N∧C∧N) Pincer Pt(II) Complexes with Bridged Organometallic Linkers: Synthesis, Structures, Self-Aggregation, and Photophysical Properties. Organometallics 2017, 36, 4794–4801. [Google Scholar] [CrossRef]
- Schneider, L.; Sivchik, V.; Chung, K.-Y.; Chen, Y.-T.; Karttunen, A.J.; Chou, P.-T.; Koshevoy, I.O. Cyclometalated Platinum(II) Cyanometallates: Luminescent Blocks for Coordination Self-Assembly. Inorg. Chem. 2017, 56, 4459–4467. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Tong, G.S.M.; Wan, Q.; Cheng, G.; Tong, W.-Y.; Ang, W.-H.; Kwong, W.-L.; Che, C.-M. Highly phosphorescent platinum(II) emitters: Photophysics, materials and biological applications. Chem. Sci. 2016, 7, 1653–1673. [Google Scholar] [CrossRef] [Green Version]
- Solomatina, A.I.; Chelushkin, P.S.; Abakumova, T.O.; Zhemkov, V.A.; Kim, M.; Bezprozvanny, I.; Gurzhiy, V.V.; Melnikov, A.S.; Anufrikov, Y.A.; Koshevoy, I.O.; et al. Reactions of Cyclometalated Platinum(II) [Pt(N∧C)(PR3)Cl] Complexes with Imidazole and Imidazole-Containing Biomolecules: Fine-Tuning of Reactivity and Photophysical Properties via Ligand Design. Inorg. Chem. 2019, 58, 204–217. [Google Scholar] [CrossRef]
- Zhang, Y.; Luo, Q.; Zheng, W.; Wang, Z.; Lin, Y.; Zhang, E.; Lü, S.; **a, L.; Rodimaa, T.; et al. Acidities of strong neutral Brønsted acids in different media. J. Phys. Org. Chem. 2013, 26, 162–170. [Google Scholar] [CrossRef]
- Yang, P.; Yang, Y.; Zhang, C.; Yang, X.J.; Hu, H.M.; Gao, Y.; Wu, B. Synthesis, structure, and catalytic ethylene oligomerization of nickel(II) and cobalt(II) complexes with symmetrical and unsymmetrical 2,9-diaryl-1,10-phenanthroline ligands. Inorg. Chim. Acta 2009, 362, 89–96. [Google Scholar] [CrossRef]
- Roy, P.; Bour, J.R.; Kampf, J.W.; Sanford, M.S. Catalytically Relevant Intermediates in the Ni-Catalyzed C(sp2)–H and C(sp3)–H Functionalization of Aminoquinoline Substrates. J. Am. Chem. Soc. 2019, 141, 17382–17387. [Google Scholar] [CrossRef]
- Reichardt, C.; Welton, T. Solvents and Solvent Effects in Organic Chemistry, 4th ed.; WILEY-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2011; p. 455. [Google Scholar]
- Hartwig, J.F. Organotransition Metal Chemistry: From Bonding to Catalysis; University Science Books: Millvalley, CA, USA, 2010; p. 9. [Google Scholar]
- Pattanayak, P.; Pratihar, J.L.; Patra, D.; Burrows, A.; Mohan, M.; Chattopadhyay, S.; Lal Pratihar, J.; Patra, D.; Burrows, A.; Mohan, M.; et al. Regiospecific ortho-aromatic hydroxylation via cyclonickelation using hydrogen peroxide and other oxygen donors: Synthesis of metalloazosalophens. Eur. J. Inorg. Chem. 2007, 2007, 4263–4271. [Google Scholar] [CrossRef]
- Sripothongnak, S.; Barone, N.; Ziegler, C.J. C–H bond activation and ring oxidation in nickel carbahemiporphyrazines. Chem. Commun. 2009, 30, 4584–4586. [Google Scholar] [CrossRef] [PubMed]
- Burla, M.C.; Caliandro, R.; Carrozzini, B.; Cascarano, G.L.; Cuocci, C.; Giacovazzo, C.; Mallamo, M.; Mazzone, A.; Polidori, G. Crystal structure determination and refinement via SIR2014. J. Appl. Crystallogr. 2015, 48, 306–309. [Google Scholar] [CrossRef]
- Sheldrick. G.M. SHELXL 2016. Acta Crystallogr., Sect. A: Found. Crystallogr. 2008, 64, 112–122.
- X-RED32 1.31 & X-SHAPE; Version 1.06; STOE & Cie GmbH: Darmstadt, Germany, 2006.
- APEX2—Software Suite for Crystallographic Programs; Bruker AXS, Inc.: Madison, WI, USA, 2010.
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A Complete Structure Solution, Refinement and Analysis Program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. Sect. C: Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SADABS-2008/1—Bruker AXS Area Detector Scaling and Absorption Correction; Bruker AXS, Inc.: Madison, WI, USA, 2008. [Google Scholar]
Sample Availability: Samples of the compounds [Ni(Phbpy)Br], [Ni(HPhbpy)Br2]2, and [Ni(Phbpy)(OAc)] are available from the authors. |






| a | NiX2 | Base (eq.) | Solvent | Time (h) | T (°C) | Yield (%) | Analysis | |
|---|---|---|---|---|---|---|---|---|
| 1 | A | NiBr2 | no base | toluene | 60 | 111 g | 0 | visual b |
| 2 | A | NiBr2 | NEt3 (11.5) | toluene | 48 | 111 g | 1.5 | 1H NMR c |
| 3 | A | NiBr2 | NaHCO3 (2) | toluene/MeOH | 3 | 111 g | 2.5 | 1H NMR |
| 4 | A | NiBr2 | Na2CO3 (4) | solid | 1 | 170 | 0 | visual b |
| 5 | B | NiBr2 | Cs2CO3 (2) | toluene | 66 | 111 g | trace | 1H NMR |
| 6 | B | NiBr2 | Na2CO3 (3) | toluene | 90 | 111 g | 19 | 1H NMR |
| 7 | C | [Ni(HPhbpy)Br2] | NaOtBu (2) | diethyl ether | 16 | 23 | 0 | 1H NMR |
| 8 | C | [Ni(HPhbpy)Br2] | KOAc/K2CO3 (2/4) | toluene | 16 | 111 g | 6 | 1H NMR |
| 9 | C | [Ni(HPhbpy)Br2] | KOAc/K2CO3 (2/4) | toluene | 62 | 111 g | 28 | 1H NMR |
| 10 | A | Ni(OAc)2.4H2O | - | toluene/EtOH//DME | 15 | 111 | 5 d | XRD |
| 11 | A | Ni(OAc)2 | - | solid | 1 | 250 | 0 e | visual b |
| 12 | A | Ni(OAc)2 | - | solid | 1 | 180 | 4 f | XRD |
| Entry | Solvent | Time (h) | Tbat (°C) | Tb.p. (°C) | Yield (%) | |
|---|---|---|---|---|---|---|
| 13 | toluene | 72 | 120 | 111 | 0.099 | 43 |
| 14 | chlorobenzene | 64 | 160 | 131 | 0.188 | 91 |
| 15 | 1,2-dichlorobenzene | 25 | 190 | 180 | 0.225 | 98 |
| 16 | p-xylene | 25 | 160 | 138 | 0.074 | 100 |
| 17 | benzonitrile | 20 | 200 | 190 | 0.333 | 0 |
| 18 | p-xylene c (from NiBr2) | 64 | 160 | 138 | 0.074 | 93 |
| 19 | 1,2-dichlorobenzene d (no base) | 64 | 190 | 180 | 0.225 | 15 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vogt, N.; Sivchik, V.; Sandleben, A.; Hörner, G.; Klein, A. Direct Base-Assisted C‒H Cyclonickelation of 6-Phenyl-2,2′-bipyridine. Molecules 2020, 25, 997. https://doi.org/10.3390/molecules25040997
Vogt N, Sivchik V, Sandleben A, Hörner G, Klein A. Direct Base-Assisted C‒H Cyclonickelation of 6-Phenyl-2,2′-bipyridine. Molecules. 2020; 25(4):997. https://doi.org/10.3390/molecules25040997
Chicago/Turabian StyleVogt, Nicolas, Vasily Sivchik, Aaron Sandleben, Gerald Hörner, and Axel Klein. 2020. "Direct Base-Assisted C‒H Cyclonickelation of 6-Phenyl-2,2′-bipyridine" Molecules 25, no. 4: 997. https://doi.org/10.3390/molecules25040997