Effects of Methyl Substitution and Leaving Group on E2/SN2 Competition for Reactions of F− with RY (R = CH3, C2H5, iC3H7, tC4H9; Y = Cl, I)
Abstract
:1. Introduction
2. Results and Discussion
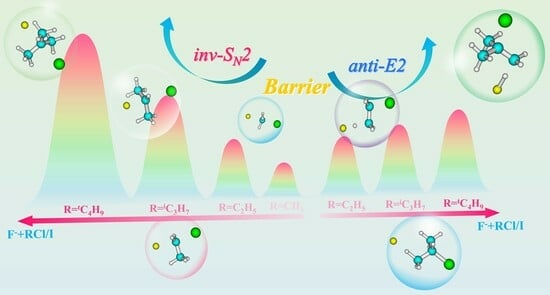
2.1. Potential Energy Surfaces of F− + RY Reactions
2.2. Effects of α-Methyl Substitution
2.3. Effects of Leaving Group
3. Computational Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Villano, S.M.; Eyet, N.; Lineberger, W.C.; Bierbaum, V.M. Reactions of α-Nucleophiles with Alkyl Chlorides: Competition between SN2 and E2 Mechanisms and the Gas-Phase α-Effect. J. Am. Chem. Soc. 2009, 131, 8227–8233. [Google Scholar] [CrossRef] [PubMed]
- Garver, J.M.; Fang, Y.R.; Eyet, N.; Villano, S.M.; Bierbaum, V.M.; Westaway, K.C. A Direct Comparison of Reactivity and Mechanism in the Gas Phase and in Solution. J. Am. Chem. Soc. 2010, 132, 3808–3814. [Google Scholar] [CrossRef] [PubMed]
- Carrascosa, E.; Meyer, J.; Michaelsen, T.; Stei, M.; Wester, R. Conservation of direct dynamics in sterically hindered SN2/E2 reactions. Chem. Sci. 2018, 9, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Gronert, S. Theoretical Studies of Elimination Reactions. 1. Reactions of F− and PH2− with CH3CH2Cl. Competition between SN2 and E2 Mechanisms for First- and Second-Row Nucleophiles. J. Am. Chem. Soc. 1991, 113, 6041–6048. [Google Scholar] [CrossRef]
- Haib, J.; Stahl, D. Competition Between Substitution (SN2), Elimination (E2) and Addition Elimination (AE) Reactions in the Gas Phase. Org. Mass. Spectrom. 1992, 27, 377–382. [Google Scholar] [CrossRef]
- Hu, W.P.; Truhlar, D.G. Factors Affecting Competitive Ion-Molecule Reactions: ClO− + C2H5Cl and C2D5Cl via E2 and SN2 Channels. J. Am. Chem. Soc. 1996, 118, 860–869. [Google Scholar] [CrossRef]
- Glad, S.S.; Jensen, F. Kinetic Isotope Effects and Transition State Geometries. A Theoretical Investigation of E2 Model Systems. J. Org. Chem. 1997, 62, 253–260. [Google Scholar] [CrossRef]
- Chung, D.S.; Kim, C.K.; Lee, I. Theoretical Studies of Competitive Gas-Phase SN2 and E2 Reactions of NCCH2CH2Cl with OH− and SH−. J. Phys. Chem. A 1997, 101, 9097–9104. [Google Scholar] [CrossRef]
- Bickelhaupt, F.M. Understanding Reactivity with Kohn-Sham Molecular Orbital Theory: E2-SN2 Mechanistic Spectrum and Other Concepts. J. Comput. Chem. 1999, 20, 114–128. [Google Scholar] [CrossRef]
- Mugnai, M.; Cardini, G.; Schettino, V. Substitution and Elimination Reaction of F− with C2H5Cl: An ab Initio Molecular Dynamics Study. J. Phys. Chem. A 2003, 107, 2540–2547. [Google Scholar] [CrossRef]
- Vayner, G.; Houk, K.N.; Jorgensen, W.L.; Brauman, J.I. Steric Retardation of SN2 Reactions in the Gas Phase and Solution. J. Am. Chem. Soc. 2004, 126, 9054–9058. [Google Scholar] [CrossRef]
- Ochran, R.A.; Uggerud, E. SN2 reactions with allylic substrates—Trends in reactivity. Int. J. Mass. Spectrom. 2007, 265, 169–175. [Google Scholar] [CrossRef]
- Bento, A.P.; Solà, M.; Bickelhaupt, F.M. E2 and SN2 Reactions of X− + CH3CH2X(X = F, Cl); an ab Initio and DFT Benchmark Study. J. Chem. Theory Comput. 2008, 4, 929–940. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.P.; Sun, X.M.; Wei, X.G.; Ren, Y.; Wong, N.B.; Li, W.K. Exploring the Reactivity Trends in the E2 and SN2 Reactions of X− + CH3CH2Cl (X = F, Cl, Br, HO, HS, HSe, NH2, PH2, AsH2,CH3, SiH3, and GeH3). J. Chem. Theory Comput. 2009, 5, 1597–1606. [Google Scholar] [CrossRef]
- Wolters, L.P.; Ren, Y.; Bickelhaup, F.M. Understanding E2 versus SN2 Competition under Acidic and Basic Conditions. ChemistryOpen 2014, 3, 29–36. [Google Scholar] [CrossRef]
- Tajti, V.; Czakó, G. Benchmark ab Initio Characterization of the Complex Potential Energy Surface of the F− + CH3CH2Cl Reaction. J. Phys. Chem. A 2017, 121, 2847–2854. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhang, J.X.; ** of the F− + CH3NH2 reaction. Phys. Chem. Chem. Phys. 2022, 24, 20249. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Montgomery, J.A.; Scalmani, G.; Barone, V.; Mennucci, B.; et al. Gaussian 09; Revision A.01; Gaussian Inc.: Wallingford, CT, USA, 2009. [Google Scholar]






| Cl | I | ||||||
|---|---|---|---|---|---|---|---|
| R | Species | MP2 | CCSD(T) a | Exptl c | MP2 | CCSD(T) b | Exptl c |
| CH3 | 1dRCH | 16.9 | 17.6 | −18.9 | −20.3 | ||
| 1TSRC | −15.1 | −15.8 | −15.8 | −17.6 | |||
| 1bRC | −15.6 | −16.1 | −16.2 | −18.3 | |||
| 1bTS | −12.3 | −12.8 | −15.6 | −18.1 | |||
| 1dTS | 32.1 | 30.7 | 22.8 | 18.3 | |||
| 1dPC | −40.7 | −41.6 | −49.6 | −55.3 | |||
| 1bPC | −40.7 | −41.6 | −49.6 | −55.3 | |||
| 1P2 | −30.6 | −31.8 | −31.1 | −40.9 | −48.0 | −42.3 | |
| C2H5 | 2c/2dRCH | −17.8 | −18.0 | −18.7 | −19.6 | ||
| 2a/2bRC | −17.5 | −17.7 | −19.4 | −20.1 | |||
| 2aTS | −11.2 | −11.1 | −15.8 | −16.0 | |||
| 2bTS | −11 | −11.3 | −15.4 | −16.9 | |||
| 2cTS | −0.1 | −6.8 | −4.2 | −4.9 | |||
| 2dTS | 31.1 | 30.0 | 21.5 | 19.2 | |||
| 2aPC | −31.9 | −33.6 | −41.5 | −42.7 | |||
| 2bPC | −42.4 | −44.6 | −51.8 | −50.9 | |||
| 2cPC | −44.2 | −41.6 | −49.0 | −54.0 | |||
| 2dPC | −42.4 | −44.6 | −51.8 | −54.0 | |||
| 2P1 | −15.5 | −18.1 | −22.5 | −26.3 | −26.2 | −37.8 | |
| 2P2 | −31.3 | −34.0 | −32.0 | −42.1 | −44.0 | −48.7 | |
| iC3H7 | 3a/3bRC | −20.7 | −21.4 | −21.9 | −22.8 | ||
| 3c/3dRC | −15.5 | −16.3 | −16.9 | −17.9 | |||
| 3aTS | −11.6 | −12.4 | −16.9 | −17.5 | |||
| 3bTS | −9.1 | −11.0 | −14.1 | −16.3 | |||
| 3cTS | 0.3 | −1.0 | −4.3 | −5.4 | |||
| 3dTS | 31.5 | 28.8 | 21.4 | - | |||
| 3aPC | −31.8 | −33.8 | −41.8 | −44.4 | |||
| 3bPC | −43.9 | −46.9 | −54.0 | −57.9 | |||
| 3cPC | −44.4 | −47.3 | −48.8 | −52.8 | |||
| 3dPC | −44.4 | −47.3 | −54.0 | - | |||
| 3P1 | −13.9 | −16.5 | −22.1 | −24.8 | −28.4 | −37.7 | |
| 3P2 | −31.8 | −34.9 | −30.3 | −42.8 | −46.8 | −45.9 | |
| tC4H9 | 4a/4bRC | −23.1 | −23.3 | −24.4 | −25.6 | ||
| 4c/4dRC | −15.6 | −16.1 | −17.2 | −17.8 | |||
| 4aTS | −11.9 | −17.6 | −17.7 | −24.4 | |||
| 4bTS | −2.3 | - | −7.5 | - | |||
| 4cTS | 0.8 | −4.6 | −3.9 | −10.8 | |||
| 4dTS | 39.9 | - | 28.8 | - | |||
| 4aPC | −33.4 | −37.8 | −43.4 | −51.2 | |||
| 4bPC | −46.2 | - | −56.1 | - | |||
| 4cPC | −44.0 | −49.1 | −49.1 | −57.4 | |||
| 4dPC | −46.2 | - | −56.1 | - | |||
| 4P1 | −12.6 | −18.2 | −22.1 | −23.9 | −37.6 | −37.6 | |
| 4P2 | −31.9 | - | −29.5 | −43.3 | - | −37.8 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhen, W.; Zhao, S.; Fu, G.; Wang, H.; Sun, J.; Yang, L.; Zhang, J. Effects of Methyl Substitution and Leaving Group on E2/SN2 Competition for Reactions of F− with RY (R = CH3, C2H5, iC3H7, tC4H9; Y = Cl, I). Molecules 2023, 28, 6269. https://doi.org/10.3390/molecules28176269
Zhen W, Zhao S, Fu G, Wang H, Sun J, Yang L, Zhang J. Effects of Methyl Substitution and Leaving Group on E2/SN2 Competition for Reactions of F− with RY (R = CH3, C2H5, iC3H7, tC4H9; Y = Cl, I). Molecules. 2023; 28(17):6269. https://doi.org/10.3390/molecules28176269
Chicago/Turabian StyleZhen, Wenqing, Siwei Zhao, Gang Fu, Hongyi Wang, Jianmin Sun, Li Yang, and Jiaxu Zhang. 2023. "Effects of Methyl Substitution and Leaving Group on E2/SN2 Competition for Reactions of F− with RY (R = CH3, C2H5, iC3H7, tC4H9; Y = Cl, I)" Molecules 28, no. 17: 6269. https://doi.org/10.3390/molecules28176269