1. Introduction
Widely applied in human disease treatment, animal husbandry, poultry farming, and aquaculture, antibiotics have been one of the most important drugs in the world [
1,
2,
3,
4]. However, their overuse and misuse have led to the emergence of antibiotic-resistant bacteria, which is a major global health threat [
5,
6,
7]. Antibiotics have also been found in the environment, particularly in water bodies, which can have harmful effects on the ecological balance and potentially pose a risk to human health [
8,
9,
10]. Furthermore, antibiotics are difficult to self-degrade in natural ecosystems, making it essential to study methods for breaking them down into small molecules [
11].
Over the past decades, various methods have been developed to address this issue, including biodegradation [
12,
13,
14,
15], ozonation [
16,
17], and photocatalysis [
18,
19,
20,
21], etc. Among the diverse methods, photocatalysis has received significant attention as a highly promising approach for treating antibiotic contaminants. This is primarily due to its cost-effectiveness and environmentally friendly nature, as it utilizes ambient conditions and sunlight to degrade antibiotics [
22]. Semiconductor materials have demonstrated exceptional performances as photocatalytic materials. As early as 1972, the Japanese scholars Fujishima et al. first reported TiO
2 as a photocatalytic electrode for splitting water [
23]. Since then, TiO
2 has been widely used in photocatalysis due to its unique properties (e.g., chemical stability, non-toxic, strong tenability, environmentally friendly, and economically viable) [
24,
25]. However, there are still challenges in optimizing the photocatalytic efficiency of TiO
2, such as improving its visible light response and reducing the recombination of electron–hole pairs [
26].
The inverse opal structure is a well-defined three-dimensional periodic arrangement of pores that are interconnected by thin walls, with the size of the pores and thickness of the walls being adjustable through the fabrication process. This unique structure offers several advantages for photocatalysis [
27,
28]. Firstly, the high surface area of the inverse opal provides numerous active sites for photocatalytic reactions, allowing for an enhanced catalytic efficiency [
29]. Secondly, the periodic arrangement of the pores and walls creates a photonic bandgap, leading to improved light absorption and scattering within the structure [
30,
31,
32]. This enhanced light–matter interaction can further boost the photocatalytic performance of the material [
33]. Overall, preparing TiO
2 in an inverse opal structure (IO-TiO
2) could effectively improve its photocatalytic performance. While IO-TiO
2 has indeed shown an improved photocatalytic performance compared to bulk TiO
2, further enhancements in its catalytic performance are still necessary. The key to addressing this issue is promoting the charge flow in the photocatalyst [
34]. A promising approach is to construct a heterojunction by combining IO-TiO
2 with another semiconductor. An artificial heterojunction can generate a built-in electric field (BIEF) due to the mismatch in Fermi levels between the two materials. The presence of a BIEF can significantly facilitate the separation and collection of photo-generated carriers, ultimately improving the photocatalytic performance [
35].
ZnIn
2S
4, a ternary semiconductor, is known for its narrow bandgap (~2.02–2.59 eV), suitable redox potentials, and good chemical stability, and it can absorb light with wavelengths up to 614 nm [
36]. As a heavy-metal-free semiconductor, it is a promising material for constructing a TiO
2-based heterojunction, extending the light absorption range of TiO
2 and enhancing its photocatalytic efficiency by creating a BIEF. Various structural types of TiO
2-based ZnIn
2S
4 photocatalysts have been developed, including nanofibers [
37], nanosheets, nanoflowers [
38], and hollow nanospheres [
39]. However, the chemical synthesis processes used to create TiO
2-based ZnIn
2S
4 photocatalysts often struggle to achieve the perfect contact between different materials (1D/2D, 2D/2D, and 2D/3D) at the nano-level in practical situations [
40]. This imperfect contact can limit the efficiency and effectiveness of the heterojunction. Therefore, it is crucial to develop a method for constructing a heterojunction with perfect contact, ensuring the optimal interaction between the materials and maximizing the photocatalytic performance. Quantum dots (QDs) possess distinct properties that deviate significantly from their bulk materials [
41]. QDs are confined in all three spatial dimensions, which leads to a quantum confinement effect and enables the tunability of band gaps [
42]. This effect is most pronounced when the size is smaller than the excitonic Bohr radius of the particular QDs, and becomes less pronounced beyond this length scale. Different from 1D, 2D, or 3D materials, 0D QDs would be uniform and tightly attached to the inverse opal skeleton [
43].
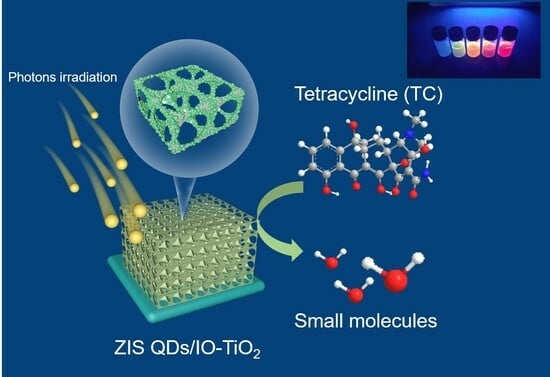
Herein, building upon our previous work about CdS quantum dots incorporated into hierarchical IO-TiO
2 [
44], we further investigated a series of heavy-metal-free 0D group II–III–VI (II–Zn/III–In/VI–S) ZnIn
2S
4 quantum dots (QDs). These ZnIn
2S
4 QDs were introduced to construct perfect contact 0D/3D ZnIn
2S
4 QDs/IO-TiO
2 (denoted as ZIS QDs/IO-TiO
2) based on a Z-scheme mechanism. Guided by the reported bandgap-tuning works, group I element Ag was doped into ZIS QDs to tune up the QDs’ band gap. Inspired by previous work, Ag-ZIS QDs/IO-TiO
2 was synthesized via a simple one-step process and demonstrated an excellent photocatalytic degradation of antibiotics under visible light illumination [
45]. Various physicochemical characterizations were conducted to investigate the advantages of coupling the slow photon effect and Z-scheme charge transfer. The superior catalytic activity observed in the Ag-ZIS QDs/IO-TiO
2 heterojunction can be attributed to the efficient separation of the photo carriers, as supported by the corresponding characterization results. This coupling strategy opens up avenues for the development of efficient solar energy conversion devices, environmental remediation technologies, and other advanced applications that require improved light harvesting and charge transfer capabilities, making it a promising candidate for photocatalytic degradation applications.
3. Materials and Methods
3.1. Reagents and Apparatus
Silver nitrate (AgNO3), zinc acetate (Zn(OAc)2·2H2O), thioacetamide (TAA), indium nitrate (In(NO3)3·4.5H2O), and L-cysteine (HSCH2CH(NH2)CO2H) were purchased from Sigma-Aldrich. Sulfur powder, sodium hydroxide (NaOH), and ethanol were obtained from Sinopharm Chemical Reagent Co., Ltd. All the reagents were of analytical grade and were used as received, without any additional purification.
3.2. Synthesis of Ag: ZnIn2S4 Quantum Dots (Ag: ZIS QDs)
Ag-doped ZnIn
2S
4 quantum dots (Ag: ZIS QDs) were synthesized using a hydrothermal method [
49]. In a typical synthesis, zinc acetate dihydrate (1.7 mmol) and indium(III) acetate (3.4 mmol) were dissolved in 70 mL of distilled water to form the precursor solution. Various amounts of silver nitrate were introduced to the precursor solution to prepare the Ag (X): ZIS QDs, where X represents the molar ratios of the doped silver. Subsequently, an aqueous solution of L-cysteine (0.17 mmol) was added under vigorous stirring, and the pH of the mixture was adjusted to 8.5 using a 1.0 M NaOH solution. Excess thioacetamide (TAA) (6.5 mmol) was then rapidly added to the mixture. The resulting solution was sealed in a Teflon-lined stainless steel autoclave and heated at 110 °C for 4 h. After cooling to room temperature, the various Ag: ZIS QDs were collected through centrifugation and subsequently washed three times using a water and ethanol mixture (with a volume ratio of 30:70).
3.3. Synthesis of PS Microspheres
The synthesis of the PS spheres followed this procedure [
50]. Initially, potassium persulfate (4 mmol) and sodium dodecyl sulfate (1.5 mmol) were dissolved in a 40 mL aqueous ethanol solution (50%
v/
v). The mixture was then heated to 75 °C under a nitrogen atmosphere, followed by the addition of 10 mL of styrene. Continuous stirring was maintained for 19 h, resulting in the formation of a white emulsion, creating a suspension of PS microspheres. For the fabrication of ordered PS array film templates, 500 μL of the prepared PS spheres suspension was dispersed into 25 mL of deionized water and subsequently sonicated for 30 min to ensure a uniform dispersion. Cleaned ITO-coated glass slides were then subjected to alternating ultrasonic cleaning with ethanol, acetone, and deionized water for 5 min. Following this, the slides were vertically immersed in a beaker and placed in a 75 °C oven. As the liquid evaporated, a hexagonal close-packed PS array film formed on the slides.
3.4. Fabrication of Ordered PS Array Film Templates
To achieve a consistent dispersion, we introduced the prepared PS spheres suspension (500 μL) into 25 mL of deionized water and subjected it to 30 min of sonication. The ITO-coated glass slides underwent a thorough cleaning process, including sequential ultrasonic treatments with ethanol, acetone, and deionized water, each lasting for 5 min.
3.5. Synthesis of IO-TiO2 Structure
The IO-TiO2 structure was synthesized using an infiltration method. The sacrificial templates were fabricated by filling the interstitial spaces between the PS spheres with the TiO2 precursor. The procedure was as follows: the TiO2 precursor, comprising TiBALDH, 0.1 M HCl, and ethanol (1:1:1.5 volume ratio), was stirred for 1 h at room temperature. Subsequently, 10 μL of the precursor was deposited onto the pre-treated PS template and incubated at 35 °C for 4 h. The temperature of the electrodes was then raised from room temperature to 450 °C at a ramp rate of 1.0 °C·min−1 and held at this temperature for 2 h, resulting in the formation of the IO-TiO2 structure.
3.6. Photocatalytic Activity Test
The photocatalytic performance of the photocatalysts was assessed by degrading TC under visible light. In each experiment, 50 mg of the Ag-ZIS QDs/IO-TiO2 sample was dispersed in an aqueous solution containing TC (10 mg/L). Before exposing it to light, the suspension was stirred in the dark for 1 h to establish an adsorption–desorption equilibrium between the photocatalyst and the TC. During the irradiation process, visible light was generated using a 250 W xenon lamp filtered through a UV cutoff filter (λ > 420 nm). Over the course of the photocatalytic process, 5 mL of the suspension was extracted at 30 min intervals. The concentration of TC in the solution was monitored via UV-vis spectroscopy, measuring the absorbance at a characteristic wavelength of 357 nm. The degradation efficiency was calculated using the formula −ln(C/C0), where C represents the TC concentration at each irradiation time and C0 is the initial concentration.
3.7. Characterization
The structural analysis of the samples was conducted using X-ray diffraction (XRD) with X’TRA and Cu Kα (ARL Co.) (D8ADVANCE, Bruker, Salbuluken, Germany) equipment. X-ray photoelectron spectroscopy (XPS) measurements were carried out utilizing a PHI 5000 Versa Probe (UlVAC-PHI Co., Chigasaki, Japan). For obtaining high-resolution transmission electron microscopy (HRTEM) images of the Ag (X): ZIS QDs, a JEOL JEM-2100 transmission electron microscope (Hitachi, Tokyo, Japan) was employed.