Extracellular Vesicles: New Players in Lymphomas
Abstract
:1. Introduction
2. EV Biogenesis
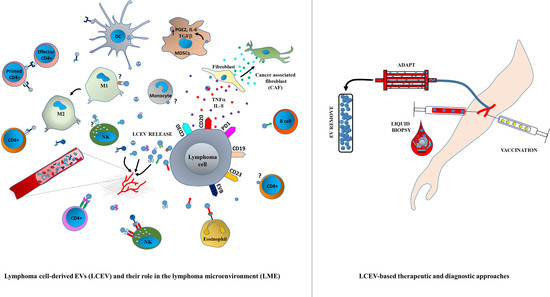
3. LCEVs and LME
4. Immune Regulation by Tumor-Derived EVs
5. EV-Mediated Lymphoma Immune Escape
6. LCEVs and Angiogenesis
7. Liquid Biopsy as a New EV-Based Screening Method
8. LCEVs as Markers of Disease and Progression
9. LCEVs as Mediators of Drug Resistance
10. EV-Based Lymphoma Therapies
10.1. EV-Based Vaccines
10.2. Neutralizing EV-Based Approaches
10.3. Targeting EV Biogenesis
10.4. Extracorporeal Removal of LCEVs
11. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lynch, R.C.; Gratzinger, D.; Advani, R.H. Clinical Impact of the 2016 Update to the WHO Lymphoma Classification. Curr. Treat. Options Oncol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Swerdlow, S.H.; Campo, E.; Pileri, S.A.; Harris, N.L.; Stein, H.; Siebert, R.; Advani, R.; Ghielmini, M.; Salles, G.A.; Zelenetz, A.D.; et al. The 2016 revision to the World Health Organization (WHO) classification of lymphoid neoplasms. Blood 2016. [Google Scholar] [CrossRef] [PubMed]
- Galardy, P.J.; Bedekovics, T.; Hermiston, M.L. Targeting childhood, adolescent and young adult non-Hodgkin lymphoma: Therapeutic horizons. Br. J. Haematol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Tarte, K. Role of the microenvironment across histological subtypes of NHL. Hematology 2017. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011. [Google Scholar] [CrossRef] [PubMed]
- Boyiadzis, M.; Whiteside, T.L. The emerging roles of tumor-derived exosomes in hematological malignancies. Leukemia 2017. [Google Scholar] [CrossRef] [PubMed]
- Tetta, C.; Ghigo, E.; Silengo, L.; Deregibus, M.C.; Camussi, G. Extracellular vesicles as an emerging mechanism of cell-to-cell communication. Endocrine 2013. [Google Scholar] [CrossRef] [PubMed]
- Bebelman, M.P.; Smit, M.J.; Pegtel, D.M.; Baglio, S.R. Biogenesis and function of extracellular vesicles in cancer. Pharmacol. Ther. 2018. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Hochberg, F.H.; Jones, P.S. Extracellular vesicles: The growth as diagnostics and therapeutics; a survey. J. Extracell. Vesicles 2018. [Google Scholar] [CrossRef]
- Rackov, G.; Garcia-Romero, N.; Esteban-Rubio, S.; Carrión-Navarro, J.; Belda-Iniesta, C.; Ayuso-Sacido, A. Vesicle-mediated control of cell function: The role of extracellular matrix and microenvironment. Front. Physiol. 2018. [Google Scholar] [CrossRef]
- Ko, J.; Carpenter, E.; Issadore, D. Detection and isolation of circulating exosomes and microvesicles for cancer monitoring and diagnostics using micro-/nano-based devices. Analyst 2016. [Google Scholar] [CrossRef] [PubMed]
- Ohno, S.I.; Drummen, G.P.C.; Kuroda, M. Focus on extracellular vesicles: Development of extracellular vesicle-based therapeutic systems. Int. J. Mol. Sci. 2016, 17, 172. [Google Scholar] [CrossRef] [PubMed]
- Abels, E.R.; Breakefield, X.O. Introduction to Extracellular Vesicles: Biogenesis, RNA Cargo Selection, Content, Release, and Uptake. Cell Mol. Neurobiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Lötvall, J.; Hill, A.F.; Hochberg, F.; Buzás, E.I.; Vizio, D.D.; Gardiner, C.; Gho, Y.S.; Kurochkin, I.V.; Mathivanan, S.; Quesenberry, P.; et al. Minimal experimental requirements for definition of extracellular vesicles and their functions: A position statement from the International Society for Extracellular Vesicles. J. Extracell. Vesicles 2014. [Google Scholar] [CrossRef] [PubMed]
- Hurwitz, S.N.; Conlon, M.M.; Rider, M.A.; Brownstein, N.C.; Meckes, D.G. Nanoparticle analysis sheds budding insights into genetic drivers of extracellular vesicle biogenesis. J. Extracell. Vesicles 2016. [Google Scholar] [CrossRef] [PubMed]
- Malloci, M.; Perdomo, L.; Veerasamy, M.; Andriantsitohai, R.; Simard, G.; Martınez, M.C. Extracellular Vesicles: Mechanisms in Human Health and Disease. Antioxid. Redox Signal. 2018. [Google Scholar] [CrossRef] [PubMed]
- Trajkovic, K.; Hsu, C.; Chiantia, S.; Rajendran, L.; Wenzel, D.; Wieland, F.; Schwille, P.; Brügger, B.; Simons, M. Ceramide Triggers Budding of Exosome Vesicles into Multivesicular Endosomes. Science 2008. [Google Scholar] [CrossRef]
- Ratajczak, M.Z.; Ratajczak, D.; Pedziwiatr, D. Extracellular microvesicles (ExMVs) in cell to cell communication: A role of telocytes. Adv. Exp. Med. Biol. 2016. [Google Scholar] [CrossRef]
- Tian, T.; Wang, Y.; Wang, H.; Zhu, Z.; ** through inhibition of exosomal export by indomethacin increases cytostatic efficacy of doxorubicin and pixantrone. Clin. Cancer Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.C.; Ribeiro, M.L.; Sarian, L.O.; Ortega, M.M.; Derchain, S.F. Exosomes-mediate microRNAs transfer in breast cancer chemoresistance regulation. Am. J. Cancer Res. 2016. [Google Scholar] [CrossRef]
- Ruella, M.; Klichinsky, M.; Kenderian, S.S.; Shestova, O.; Ziober, A.; Kraft, D.O.; Gill, S. Overcoming the Immunosuppressive Tumor Microenvironment of Hodgkin Lymphoma Using Chimeric Antigen Receptor T Cells. Cancer Discov. 2017. [CrossRef] [PubMed]
- Filley, A.C.; Henriquez, M.; Dey, M. CART Immunotherapy: Development, Success, and Translation to Malignant Gliomas and Other Solid Tumors. Front. Oncol. 2018. [Google Scholar] [CrossRef]
- Aoki, T.; Steidl, C. Novel Biomarker Approaches in Classic Hodgkin Lymphoma. Cancer J. 2018. [Google Scholar] [CrossRef]
- Bröckelmann, P.J.; Borchmann, P.; Engert, A. Current and future immunotherapeutic approaches in Hodgkin lymphoma. Leuk. Lymphoma 2016. [Google Scholar] [CrossRef]
- Stickney, D.R.; Foon, K.A. Biologic response modifiers: Therapeutic approaches to lymphoproliferative diseases. Curr. Opin. Oncol. 1992, 4, 847–855. [Google Scholar] [CrossRef] [PubMed]
- Sheng, J.; Qin, H.; Zhang, D.; Zhang, X.; Liu, L.; Li, B. New Strategies for Therapeutic Cancer Vaccines. Anticancer Agents Med. Chem. 2018. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.; Liu, Q.; Wei, J.; Liu, B. Personalized cancer neoantigen vaccines come of age. Theranostics 2018. [Google Scholar] [CrossRef] [PubMed]
- Marron, T.U.; Ronner, L.; Martin, P.E.; Flowers, C.R.; Brody, J.D. Vaccine strategies for the treatment of lymphoma: Preclinical progress and clinical trial update. Immunotherapy 2016. [Google Scholar] [CrossRef] [PubMed]
- Gilboa, E. The makings of a tumor rejection antigen. Immunity 1999. [Google Scholar] [CrossRef]
- Viaud, S.; Théry, C.; Ploix, S.; Tursz, T.; Lapierre, V.; Lantz, O.; Zitvogel, L.; Chaput, N. Dendritic cell-derived exosomes for cancer immunotherapy: What’s next? Cancer Res. 2010. [Google Scholar] [CrossRef]
- Menay, F.; Herschlik, L.; De Toro, J.; Cocozza, F.; Tsacalian, R.; Gravisaco, M.J.; Di Sciullo, M.P.; Vendrell, A.; Waldner, C.I.; Mongini, C. Exosomes isolated from ascites of T-cell lymphoma-bearing mice expressing surface CD24 and HSP-90 induce a tumor-specific immune response. Front. Immunol. 2017. [Google Scholar] [CrossRef]
- Chaput, N.; Taïeb, J.; Schartz, N.E.C.; André, F.; Angevin, E.; Zitvogel, L. Exosome-based immunotherapy. Cancer Immunol. Immunother. 2004. [Google Scholar] [CrossRef]
- Taieb, J.; Chaput, N.; Schartz, N.; Roux, S.; Novault, S.; Menard, C.; Ghiringhelli, F.; Terme, M.; Carpentier, A.F.; Darrasse-Jese, G.; et al. Chemoimmunotherapy of Tumors: Cyclophosphamide Synergizes with Exosome Based Vaccines. J. Immunol. 2006. [Google Scholar] [CrossRef]
- Salles, G.; Barrett, M.; Foà, R.; Maurer, J.; O’Brien, S.; Valente, N.; Wenger, M.; Maloney, D.G. Rituximab in B-Cell Hematologic Malignancies: A Review of 20 Years of Clinical Experience. Adv. Ther. 2017. [Google Scholar] [CrossRef]
- Leonard, J.P.; Goldenberg, D.M. Preclinical and clinical evaluation of epratuzumab (anti-CD22 IgG) in B-cell malignancies. Oncogene 2007. [Google Scholar] [CrossRef] [PubMed]
- Furman, R.; Forero-Torres, A.; Shustov, A.; Drachman, J. A phase I study of dacetuzumab (SGN-40, a humanized anti-CD40 monoclonal antibody) in patients with chronic lymphocytic leukemia. Leuk Lymphoma. 2010. [Google Scholar] [CrossRef] [PubMed]
- Blanc, V.; Bousseau, A.; Caron, A.; Carrez, C.; Lutz, R.J.; Lambert, J.M. SAR3419: An anti-CD19-maytansinoid immunoconjugate for the treatment of B-cell malignancies. Clin. Cancer Res. 2011. [Google Scholar] [CrossRef] [PubMed]
- Nagorsen, D.; Baeuerle, P.A. Immunomodulatory therapy of cancer with T cell-engaging BiTE antibody blinatumomab. Exp. Cell Res. 2011. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Lapalombella, R.; Joshi, T.; Cheney, C.; Gowda, A.; Hayden-Ledbetter, M.S.; Baum, P.R.; Lin, T.S.; Jarjoura, D.; Lehman, A.; et al. Targeting CD37-positive lymphoid malignancies with a novel engineered small modular immunopharmaceutical. Blood 2007. [Google Scholar] [CrossRef]
- Huang, M.-B.; Gonzalez, R.R.; Lillard, J.; Bond, V.C. Secretion modification region-derived peptide blocks exosome release and mediates cell cycle arrest in breast cancer cells. Oncotarget 2017. [Google Scholar] [CrossRef] [PubMed]
- Nanbo, A.; Katano, H.; Kataoka, M.; Hoshina, S.; Sekizuka, T.; Kuroda, M.; Ohba, Y. Infection of Epstein–Barr virus in type III latency modulates biogenesis of exosomes and the expression profile of exosomal miRNAs in the Burkitt lymphoma Mutu cell lines. Cancers 2018, 10, 237. [Google Scholar] [CrossRef]
- Menck, K.; Sönmezer, C.; Worst, T.S.; Schulz, M.; Dihazi, G.H.; Streit, F.; Erdmann, G.; Kling, S.; Boutros, M.; Binder, C.; et al. Neutral sphingomyelinases control extracellular vesicles budding from the plasma membrane. J. Extracell. Vesicles 2017. [Google Scholar] [CrossRef]
- Datta, A.; Kim, H.; McGee, L.; Johnson, A.E.; Talwar, S.; Marugan, J.; Southall, N.; Hu, X.; Lal, M.; Mondal, D.; et al. High-Throughput screening identified selective inhibitors of exosome biogenesis and secretion: A drug repurposing strategy for advanced cancer. Sci. Rep. 2018. [Google Scholar] [CrossRef]
- Zerial, M.; McBride, H. Heterogeneity of Rab effectors. Nat. Rev. Mol. Cell Biol. 2001. [Google Scholar] [CrossRef]
- Hsu, C.; Morohashi, Y.; Yoshimura, S.I.; Manrique-Hoyos, N.; Jung, S.Y.; Lauterbach, M.A.; Bakhti, M.; Grønborg, M.; Möbius, W.; Rhee, J.S.; et al. Regulation of exosome secretion by Rab35 and its GTPase-activating proteins TBC1D10A-C. J. Cell Biol. 2010. [Google Scholar] [CrossRef] [PubMed]
- Ostrowski, M.; Carmo, N.B.; Krumeich, S.; Fanget, I.; Raposo, G.; Savina, A.; Moita, C.F.; Schauer, K.; Hume, A.N.; Freitas, R.P.; et al. Rab27a and Rab27b control different steps of the exosome secretion pathway. Nat. Cell Biol. 2010. [Google Scholar] [CrossRef] [PubMed]
- Savina, A.; Furlán, M.; Vidal, M.; Colombo, M.I. Exosome release is regulated by a calcium-dependent mechanism in K562 cells. J. Biol. Chem. 2003. [Google Scholar] [CrossRef] [PubMed]
- Chalmin, F.; Ladoire, S.; Mignot, G.; Vincent, J.; Bruchard, M.; Remy-Martin, J.P.; Boireau, W.; Rouleau, A.; Simon, B.; Lanneau, D.; et al. Membrane-associated Hsp72 from tumor-derived exosomes mediates STAT3-dependent immunosuppressive function of mouse and human myeloid-derived suppressor cells. J. Clin. Investig. 2010. [Google Scholar] [CrossRef] [PubMed]
- Marleau, A.M.; Chen, C.S.; Joyce, J.A.; Tullis, R.H. Exosome removal as a therapeutic adjuvant in cancer. J. Transl. Med. 2012. [Google Scholar] [CrossRef] [PubMed]
- Theodoraki, M.N.; Yerneni, S.S.; Hoffmann, T.K.; Gooding, W.E.; Whiteside, T.L. Clinical significance of PD-L1 exosomes in plasma of head and neck cancer patients. Clin. Cancer Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Huang, A.C.; Zhang, W.; Zhang, G.; Wu, M.; Xu, W.; Yu, Z.; Yang, J.; Wang, B.; Sun, H.; et al. Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature 2018. [Google Scholar] [CrossRef]
- Jabbour, E.; Peslin, N.; Arnaud, P.; Ferme, C.; Carde, P.; Vantelon, J.M.; Bocaccio, C.; Bourhis, J.H.; Koscielny, S.; Ribrag, V. Prognostic value of the age-adjusted International Prognostic Index in chemosensitive recurrent or refractory non-Hodgkin’s lymphomas treated with high-dose BEAM therapy and autologous stem cell transplantation. Leuk. Lymphoma 2005. [Google Scholar] [CrossRef]
- Michot, J.M.; Lazarovici, J.; Ghez, D.; Danu, A.; Fermé, C.; Bigorgne, A.; Ribrag, V.; Marabelle, A.; Aspeslagh, S. Challenges and perspectives in the immunotherapy of Hodgkin lymphoma. Eur. J. Cancer. 2017. [Google Scholar] [CrossRef]




| LME-Associated Composition | Cell Types |
|---|---|
| Immune cells | Cytotoxic T cells (CTLs) |
| Follicular B helper T cells (TFH) | |
| Regulatory T cells (Treg) | |
| Natural Killer cells (NK) | |
| Bystander B cells | |
| Stromal cells | Mesenchymal stromal cells (MSCs) |
| Lymphoma associated macrophages (LAMs) | |
| Myeloid-derived suppressor cells (MDSCs) | |
| Dendritic cells | |
| Extracellular components | Extracellular matrix (ECM) |
| Cytokines/Chemokines | |
| Lymphoma exosome |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navarro-Tableros, V.; Gomez, Y.; Camussi, G.; Brizzi, M.F. Extracellular Vesicles: New Players in Lymphomas. Int. J. Mol. Sci. 2019, 20, 41. https://doi.org/10.3390/ijms20010041
Navarro-Tableros V, Gomez Y, Camussi G, Brizzi MF. Extracellular Vesicles: New Players in Lymphomas. International Journal of Molecular Sciences. 2019; 20(1):41. https://doi.org/10.3390/ijms20010041
Chicago/Turabian StyleNavarro-Tableros, Victor, Yonathan Gomez, Giovanni Camussi, and Maria Felice Brizzi. 2019. "Extracellular Vesicles: New Players in Lymphomas" International Journal of Molecular Sciences 20, no. 1: 41. https://doi.org/10.3390/ijms20010041