Sorption and Diffusion of Water Vapor and Carbon Dioxide in Sulfonated Polyaniline as Chemical Sensing Materials
Abstract
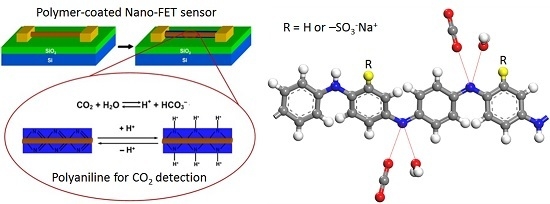
:1. Introduction
2. Materials and Methods
2.1. Atomistic Models
2.2. Simulation
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- MacDiarmid, A.G. A novel role for organic polymers (Nobel Lecture). Angew. Chem. Int. Ed. 2001, 40, 2581–2590. [Google Scholar] [CrossRef]
- Chen, X.P.; Shen, L.; Yuan, C.A.; Wong, C.K.Y.; Zhang, G.Q. Molecular model for the charge carrier density dependence of conductivity of polyaniline as chemical sensing materials. Sens. Actuators B Chem. 2013, 177, 856–861. [Google Scholar] [CrossRef]
- Freund, M.S.; Deore, B.A. Self-Doped Conducting Polymers; John Wiley & Sons: Chichester, UK, 2007. [Google Scholar]
- Pinto, N.J.; Johnson, A.T., Jr.; MacDiarmid, A.G.; Mueller, C.H.; Theofylaktos, N.; Robinson, D.C.; Miranda, F.A. Electrospun polyaniline/polyethylene oxide nanofiber field-effect transistor. Appl. Phys. Lett. 2003, 83, 4244–4246. [Google Scholar] [CrossRef]
- **, Z.; Su, Y.X.; Duan, Y.X. An improved optical pH sensor based on polyaniline. Sens. Actuators B Chem. 2000, 71, 118–122. [Google Scholar] [CrossRef]
- Gustafsson, G.; Cao, Y.; Treacy, G.M.; Klavetter, F.; Colaneri, N.; Heeger, A.J. Flexible light-emitting diodes made from soluble conducting polymers. Nature 1992, 357, 477–479. [Google Scholar] [CrossRef]
- Wang, K.; Meng, Q.H.; Zhang, Y.J.; Wei, Z.X.; Miao, M.H. High-performance two-ply yarn supercapacitors based on carbon nanotubes and polyaniline nanowire arrays. Adv. Mater. 2013, 25, 1494–1498. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.Y.; Liang, J.; Tao, Z.L.; Chen, J. Functional materials for rechargeable batteries. Adv. Mater. 2011, 23, 1695–1715. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Tan, T.; Lee, J. Corrosion protection of mild steel by electroactive polyaniline coatings. Synthetic. Met. 1997, 88, 237–242. [Google Scholar] [CrossRef]
- Wang, Y.; **g, X. Intrinsically conducting polymers for electromagnetic interference shielding. Polym. Adv. Technol. 2005, 16, 344–351. [Google Scholar] [CrossRef]
- Shen, P.; Huang, H.; Tseung, A. A Study of tungsten trioxide and polyaniline composite films I. Electrochemical and electrochromic behavior. J. Am. Chem. Soc. 1992, 139, 1840–1845. [Google Scholar]
- Ostwal, M.M.; Tsotsis, T.T.; Sahimi, M. Molecular dynamics simulation of diffusion and sorption of water in conducting polyaniline. J. Chem. Phys. 2007, 126. [Google Scholar] [CrossRef] [PubMed]
- Ostwal, M.M.; Sahimi, M.; Tsotsis, T.T. Water harvesting using a conducting polymer: A study by molecular dynamics simulation. Phys. Rev. E 2009, 79. [Google Scholar] [CrossRef] [PubMed]
- Yue, J.; Wang, Z.H.; Cromack, K.R.; Epstein, A.J.; MacDiarmid, A.G. Effect of sulfonic acid group on polyaniline backbone. J. Am. Chem. Soc. 1991, 113, 2665–2671. [Google Scholar] [CrossRef]
- Polshettiwar, V.; Varma, R.S. Aqueous microwave chemistry: A clean and green synthetic tool for rapid drug discovery. Chem. Soc. Rev. 2008, 37, 1546–1557. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Virji, S.; Weiller, B.H.; Kaner, R.B. Polyaniline nanofibers: Facile synthesis and chemical sensors. J. Am. Chem. Soc. 2003, 125, 314–315. [Google Scholar] [CrossRef] [PubMed]
- Nohria, R.; Khillan, R.K.; Su, Y.; Dikshit, R.; Lvov, Y.; Varahramyan, K. Humidity sensor based on ultrathin polyaniline film deposited using layer-by-layer nano-assembly. Sens. Actuators B Chem. 2006, 114, 218–222. [Google Scholar] [CrossRef]
- Chen, X.P.; Wong, C.K.Y.; Yuan, C.A.; Zhang, G.Q. Impact of the functional group on the working range of polyaniline as carbon dioxide sensors. Sens. Actuators B Chem. 2012, 175, 15–21. [Google Scholar] [CrossRef]
- Wei, X.L.; Wang, Y.Z.; Long, S.; Bobeczko, C.; Epstein, A.J. Synthesis and physical properties of highly sulfonated polyaniline. J. Am. Chem. Soc. 1996, 118, 2545–2555. [Google Scholar] [CrossRef]
- Banerjee, R.; Furukawa, H.; Britt, D.; Knobler, C.; O’Keeffe, M.; Yaghi, O.M. Control of pore size and functionality in isoreticular zeolitic imidazolate frameworks and their carbon dioxide selective capture properties. J. Am. Chem. Soc. 2009, 131, 3875–3877. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.; Jiang, X.; Ruan, L.-W.; Liu, B.; Wang, Y.-M.; Zhu, J.-F.; Qiu, L.-G. Post-combustion CO2 capture with the HKUST-1 and MIL-101(Cr) metal–organic frameworks: Adsorption, separation and regeneration investigations. Micropor. Mesopor. Mater. 2013, 179, 191–197. [Google Scholar] [CrossRef]
- Liu, Q.; Ning, L.; Zheng, S.; Tao, M.; Shi, Y.; He, Y. Adsorption of carbon dioxide by MIL-101 (Cr): Regeneration conditions and influence of flue gas contaminants. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Neethirajan, S.; Freund, M.S.; Jayas, D.S.; Shafai, C.; Thomson, D.J.; White, N.D.G. Development of carbon dioxide (CO2) sensor for grain quality monitoring. Biosyst. Eng. 2010, 106, 395–404. [Google Scholar] [CrossRef]
- Ostwal, M.M.; Pellegrino, J.; Norris, I.; Tsotsis, T.T.; Sahimi, M.; Mattes, B.R. Water sorption of acid-doped polyaniline solid fibers: Equilibrium and kinetic response. Ind. Eng. Chem. Res. 2005, 44, 7860–7867. [Google Scholar] [CrossRef]
- Zhang, T.; Nix, M.B.; Yoo, B.-Y.; Deshusses, M.A.; Myung, N.V. Electrochemically functionalized single-walled carbon nanotube gas sensor. Electroanalysis 2006, 18, 1153–1158. [Google Scholar] [CrossRef]
- **e, D.; Jiang, Y.; Pan, W.; Li, D.; Wu, Z.; Li, Y. Fabrication and characterization of polyaniline-based gas sensor by ultra-thin film technology. Sens. Actuators B Chem. 2002, 81, 158–164. [Google Scholar] [CrossRef]
- Sadek, A.Z.; Baker, C.O.; Powell, D.A.; Wlodarski, W.; Kaner, R.B.; Kalantar-zadeh, K. Polyaniline nanofiber based surface acoustic wave gas sensors—Effect of nanofiber diameter on H2 response. IEEE Sens. J. 2007, 7, 213–218. [Google Scholar] [CrossRef]
- Arsat, R.; Yu, X.; Li, Y.; Wlodarski, W.; Kalantar-Zadeh, K. Hydrogen gas sensor based on highly ordered polyaniline nanofibers. Sens. Actuators B Chem. 2009, 137, 529–532. [Google Scholar] [CrossRef]
- Atashbar, M.; Sadek, A.; Wlodarski, W.; Sriram, S.; Bhaskaran, M.; Cheng, C.; Kaner, R.; Kalantar-Zadeh, K. Layered SAW gas sensor based on CSA synthesized polyaniline nanofiber on AlN on 64° YX LiNbO3 for H2 sensing. Sens. Actuators B Chem. 2009, 138, 85–89. [Google Scholar] [CrossRef]
- Li, G.; Martinez, C.; Semancik, S. Controlled electrophoretic patterning of polyaniline from a colloidal suspension. J. Am. Chem. Soc. 2005, 127, 4903–4909. [Google Scholar] [CrossRef] [PubMed]
- Athawale, A.A.; Bhagwat, S.; Katre, P.P. Nanocomposite of Pd-polyaniline as a selective methanol sensor. Sens. Actuators B Chem. 2006, 114, 263–267. [Google Scholar] [CrossRef]
- Sharma, S.; Nirkhe, C.; Pethkar, S.; Athawale, A.A. Chloroform vapour sensor based on copper/polyaniline nanocomposite. Sens. Actuators B Chem. 2002, 85, 131–136. [Google Scholar] [CrossRef]
- Doan, T.C.; Ramaneti, R.; Baggerman, J.; van der Bent, J.F.; Marcelis, A.T.; Tong, H.D.; van Rijn, C.J. Carbon dioxide sensing with sulfonated polyaniline. Sens. Actuators B Chem. 2012, 168, 123–130. [Google Scholar] [CrossRef]
- Chen, X.P.; Jiang, J.K.; Liang, Q.H.; Yang, N.; Ye, H.Y.; Cai, M.; Shen, L.; Yang, D.G.; Ren, T.L. First-principles study of the effect of functional groups on polyaniline backbone. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Ullah, H.; Shah, A.-H.A.; Bilal, S.; Ayub, K. DFT Study of Polyaniline NH3, CO2, and CO Gas Sensors: Comparison with Recent Experimental Data. J. Phys. Chem. C 2013, 117, 23701–23711. [Google Scholar] [CrossRef]
- Chen, X.P.; Yang, N.; Jiang, J.K.; Liang, Q.H.; Yang, D.G.; Zhang, G.Q.; Ren, T.L. Ab initio Study of Temperature, Humidity and Covalent Functionalization Induced Band Gap Change of Single-Walled Carbon Nanotubes. IEEE Electr. Device Lett. 2015, 36, 606–608. [Google Scholar] [CrossRef]
- Virji, S.; Huang, J.; Kaner, R.B.; Weiller, B.H. Polyaniline nanofiber gas sensors: Examination of response mechanisms. Nano Lett. 2004, 4, 491–496. [Google Scholar] [CrossRef]
- Athawale, A.A.; Kulkarni, M.V. Polyaniline and its substituted derivatives as sensor for aliphatic alcohols. Sens. Actuators B Chem. 2000, 67, 173–177. [Google Scholar] [CrossRef]
- Chen, X.P.; Yuan, C.A.; Wong, C.K.Y.; Koh, S.W.; Zhang, G.Q. Validation of forcefields in predicting the physical and thermophysical properties of emeraldine base polyaniline. Mol. Simulat. 2011, 37, 990–996. [Google Scholar] [CrossRef]
- Chen, X.P.; Yuan, C.A.; Wong, C.K.Y.; Zhang, G.Q. Molecular modeling of temperature dependence of solubility parameters for amorphous polymers. J. Mol. Model. 2012, 18, 2333–2341. [Google Scholar] [CrossRef] [PubMed]
- Berashevich, J.; Chakraborty, T. Tunable band gap and magnetic ordering by adsorption of molecules on graphene. Phys. Rev. B 2009, 80. [Google Scholar] [CrossRef]
- Shih, Y.C.; Chen, C.S.; Wu, K.C. First-principles surface stress calculations and multiscale deformation analysis of a self-assembled monolayer adsorbed on a micro-cantilever. Sensors 2014, 14, 7435–7450. [Google Scholar] [CrossRef] [PubMed]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.P.; Liang, Q.H.; Jiang, J.K.; Wong, C.K.Y.; Leung, S.Y.Y.; Ye, H.Y.; Yang, D.G.; Ren, T.L. Functionalization-induced changes in the structural and physical properties of amorphous polyaniline: A first-principles and molecular dynamics study. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Ostwal, M.M.; Qi, B.; Pellegrino, J.; Fadeev, A.G.; Norris, I.D.; Tsotsis, T.T.; Sahimi, M.; Mattes, B.R. Water sorption of acid-doped polyaniline powders and hollow fibers: Equilibrium and kinetic response. Ind. Eng. Chem. Res. 2006, 45, 6021–6031. [Google Scholar] [CrossRef]
- Meunier, M. Diffusion coefficients of small gas molecules in amorphous cis-1,4-polybutadiene estimated by molecular dynamics simulations. J. Chem. Phys. 2005, 123, 134906. [Google Scholar] [CrossRef] [PubMed]







| Geometry Optimization | Equilibration Process of Structure | ||
|---|---|---|---|
| Forefield: COMPASS | Step | Simulation conditions | Time (ps) |
| Quality: Fine | |||
| Summation method: Ewald for electrostatic and atom base for van der Waals (vdW) | 1 | NPT, 1 GPa, 298 K | 20 |
| Cutoff distance: 10.5 Å for sorption and 15.5 Å for diffusion | 2 | NPT, 0.5 GPa, 298 K | 50 |
| Algorithm: smart | 3 | NPT, 0.0001 GPa, 298 K | 200 |
| ‘Fine’ convergence tolerance | 4 | A stepwise procedure of NVT of heating from 298 K to 698 K and cooling from 698 K down to 298 K by a step of 50 K | 50 ps/stepwise |
| 5 cycles | |||
| Energy (kcal/mol): 1 × 10−4 | 5 | NPT, 1 atm, 298 K | 1000 |
| Buffer width: 0.5 Å | The total simulation time for the equilibration process is 5.27 ns | ||
| Spline width: 1 Å | MD simulation for CO2 diffusion in the polymer system | ||
| Displacement (Å): 5 × 10−5 | NVT, 298 K | 7000 | |
| Max. iterations: 50,000 | |||
| Species | ΔEad (eV) | Q (|e|) | B (eV) |
|---|---|---|---|
| EB-PANI | 1.44 | ||
| Na-SPANI (I) | 1.44 | ||
| Na-SPANI (II) | 1.30 | ||
| Na-SPANI (III) | 1.29 | ||
| EB-PANI CO2_1 | −0.054 | −0.001 | 1.43 |
| EB-PANI CO2_2 | −0.082 | −0.001 | 1.45 |
| Na-SPANI (I) CO2_1 | −0.115 | −0.006 | 1.42 |
| Na-SPANI (I) CO2_2 | −0.111 | −0.005 | 1.42 |
| Na-SPANI (II) CO2_1 | −0.112 | −0.005 | 1.33 |
| Na-SPANI (II) CO2_2 | −0.32 | 0.013 | 1.33 |
| Na-SPANI (III) CO2_1 | −0.189 | −0.006 | 1.34 |
| Na-SPANI (III) CO2_2 | −0.564 | 0.014 | 1.34 |
| EB-PANI H2O_1 | −0.435 | −0.017 | 1.46 |
| EB-PANI H2O_2 | −0.463 | −0.014 | 1.42 |
| Na-SPANI (I) H2O_1 | −0.501 | −0.015 | 1.42 |
| Na-SPANI (I) H2O_2 | −0.587 | −0.014 | 1.42 |
| Na-SPANI (II) H2O_1 | −0.491 | −0.011 | 1.33 |
| Na-SPANI (II) H2O_2 | −0.899 | 0.021 | 1.32 |
| Na-SPANI (III) H2O_1 | −1.06 | −0.013 | 1.24 |
| Na-SPANI (III) H2O_2 | −1.188 | 0.014 | 1.36 |
| Polymers | Sincreasing |
|---|---|
| Na-SPANI (I)/EB-PANI | 41.4 |
| Na-SPANI (II)/EB-PANI | 171.3 |
| Na-SPANI (III)/EB-PANI | 355.7 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, Q.; Jiang, J.; Ye, H.; Yang, N.; Cai, M.; **ao, J.; Chen, X. Sorption and Diffusion of Water Vapor and Carbon Dioxide in Sulfonated Polyaniline as Chemical Sensing Materials. Sensors 2016, 16, 606. https://doi.org/10.3390/s16050606
Liang Q, Jiang J, Ye H, Yang N, Cai M, **ao J, Chen X. Sorption and Diffusion of Water Vapor and Carbon Dioxide in Sulfonated Polyaniline as Chemical Sensing Materials. Sensors. 2016; 16(5):606. https://doi.org/10.3390/s16050606
Chicago/Turabian StyleLiang, Qiuhua, Junke Jiang, Huaiyu Ye, Ning Yang, Miao Cai, **g ** Chen. 2016. "Sorption and Diffusion of Water Vapor and Carbon Dioxide in Sulfonated Polyaniline as Chemical Sensing Materials" Sensors 16, no. 5: 606. https://doi.org/10.3390/s16050606