Discovery of Novel Saponins from the Viscera of the Sea Cucumber Holothuria lessoni
Abstract
:1. Introduction
2. Results and Discussion







| [M + Na]+ m/z | MW | Formula | Compound’s Name | Novel (N)/Published (P) | References |
|---|---|---|---|---|---|
| 889.4 | 866 | C41H63NaO16S | Holothurin B3 | P | [83] |
| C42H67NaO15S | Unidentified | N | − | ||
| 905.4 | 882 | C41H63NaO17S | Holothurin B4 | P | [83] |
| Holothurin B | P | [61,84,85,86] | |||
| Nobiliside B | P | [87] | |||
| 907.4 | 884 | C41H65NaO17S | Holothurin B2 | P | [83] |
| Leucospilotaside B | P | [8] | |||
| 911.6 | 888 | C45H92O16 | Unidentified | N | − |
| 917.4 | 994 | C44H71NaO15S | Unidentified | N | − |
| 921.4 | 898 | C41H63NaO18S | Leucospilotaside A | P | [84] |
| 1034.1 | 1011 | a* | Unidentified | N | − |
| 1065.5 | 1042 | C48H82O24 | Unidentified | N | − |
| 1071.5 | 1048 | C47H93NaO21S | Unidentified | N | − |
| 1078.5 | 1055 | a * | Unidentified | N | − |
| 1083.3 | 1060 | C58H64O25 | Unidentified | N | − |
| 1087.6 | 1064 | C47H93NaO22S | Unidentified | N | − |
| 1123.5 | 1100 | C54H84O23 | Unidentified | N | − |
| 1125.5 | 1102 | C54H86O23 | Holothurinoside C Holothurinoside C1 | P | [62,69,88,89] |
| 1127.6 | 1104 | C54H88O23 | Unidentified | N | − |
| Unidentified | N | − | |||
| 1141.6 | 1118 | C54H86O24 | Desholothurin A (Nobiliside 2a), Desholothurin A1 (Arguside E) | P | >[5,62,69,88,89,90] |
| 1149.2 | 1126 | a * | Unidentified | N | − |
| 1157.5 | 1134 | C54H109O25 | Holothurinoside J1 | P | [50] |
| C49H91NaO25S | Unidentified | N | − | ||
| 1193.5 | 1170 | C55H87NaO23S | Unidentified | N | − |
| 1199.4 | 1176 | C54H64O29 | Unidentified | N | − |
| 1221.5 ** | 1198 | C56H78O28 | Unidentified | N | − |
| 1225.5 | 1202 | C54H83NaO26S | Unidentified | N | − |
| 1227.5 | 1204 | C54H85NaO26S | Fuscocineroside B/C, Scabraside A or 24-Dehydroechinoside A | P | [29,56,89,91,92] |
| 1229.5 | 1206 | C54H87NaO26S | Holothurin A2, Echinoside A | P | [7,61,91,93,94,95] |
| 1243.5 | 1220 | C54H85NaO27S | Holothurin A Scabraside B 17-Hydroxy fuscocineroside B 25-Hydroxy fuscocinerosiden B | P | [29,58,61,95,96,97] |
| 1245.5 | 1222 | C54H87NaO27S | Holothurin A1 | P | [91] |
| Holothurin A4 | [36] | ||||
| Scabraside D | [92] | ||||
| 1259.5 | 1236 | C54H85NaO28S | Holothurin A3 | P | [36] |
| Unidentified | N | − | |||
| 1265.5 | 1242 | C56H83NaO27S | Unidentified | N | − |
| 1271.6 | 1248 | C60H96O27 | Impatienside B | P | [5,98] |
| 1287.6 | 1264 | C60H96O28 | Holothurinoside E, Holothurinoside E1 | P | [62,69] |
| Unidentified | N | − | |||
| Unidentified | N | − | |||
| 17-Dehydroxyholothurinoside A | P | [5,99] | |||
| 1289.6 | 1266 | C60H98O28 | Griseaside A | P | [99] |
| 1301.6 | 1278 | C61H98O28 | Holothurinoside M | P | [65] |
| C60H94O29 | Unidentified | N | − | ||
| 1303.6 | 1280 | C60H96O29 | Holothurinoside A | P | [5,62,69,88] |
| Holothurinoside A1 | |||||
| Unidentified | N | − | |||
| Unidentified | N | − | |||
| Unidentified | N | − | |||
| Unidentified | N | − | |||
| 1305.6 | 1282 | a * | Unidentified | N | − |
| 1317.6 | 1294 | C61H98O29 | Unidentified | N | − |
| 1335.3 | 1312 | a * | Unidentified | N | − |
| 1356.4 | 1333 | a * | Unidentified | N | − |
| 1409.4 | 1386 | C61H78O36 | Unidentified | N | − |
| 1411.7 | 1388 | C62H116O33 | Unidentified | N | − |
| 1419.7 | 1396 | C66H108O31 | Unidentified | N | − |
| 1435.7 | 1412 | C66H108O32 | Unidentified | N | − |
| 1465.7 | 1442 | C67H110O33 | Arguside B | P | [5,32] |
| Arguside C | |||||
| 1475.6 | 1452 | C65H96O36 | Unidentified | N | − |
| 1477.7 ** | 1454 | C61H114O38 | Unidentified | N | − |
| 1481.7 | 1458 | C66H106O35 | Unidentified | N | − |
| 1493.7 | 1470 | C65H114O36 | Unidentified | N | − |
| 1495.7 | 1472 | C61H116O39 | Holothurinoside K1 | P | [50] |
| C72H112O31 | Unidentified | N | − | ||
| 1591.7 | 1568 | C66H120O41 | Unidentified | N | − |
2.1. MALDI-MS/MS Data of Compound Holothurin A in the Positive Ion Mode

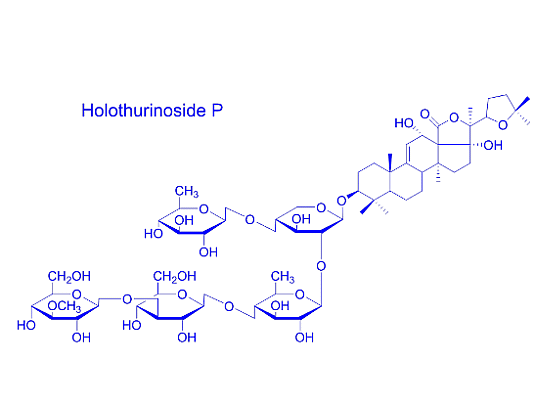
2.2. Key Fragments and Structure Elucidation of Novel Saponins
| Diagnostic ions in CID Spectra of [M + Na]+ | |||
|---|---|---|---|
| m/z Signals (Da) | |||
| 507 | 523 | 639 | |
| Chemical signatures | MeGlc-Glc-Qui + Na | MeGlc-Glc-Glc + Na | MeGlc-Glc-Qui-Xyl + Na |

2.3. Analyses of Saponins by ESI-MS


2.3.1. Molecular Mass of Saponins by ESI
2.3.2. Structure Elucidation of the Saponins by ESI-MS/MS

3. Experimental Section
3.1. Sea Cucumber Sample
3.2. Extraction Protocol
Extraction of Saponins
3.3. Purification of the Extract
3.4. Thin Layer Chromatography (TLC)
3.5. High Performance Centrifugal Partition Chromatography (HPCPC or CPC)
3.6. Mass Spectrometry
3.6.1. MALDI-MS
3.6.2. ESI-MS
4. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lovatelli, A.; Conand, C. Advances in Sea Cucumber Aquaculture and Management; FAO: Rome, Italy, 2004. [Google Scholar]
- Purcell, S.W.; Samyn, Y.; Conand, C. Commercially Important Sea Cucumbers of the World; FAO Species Catalogue for Fishery Purposes No. 6; FAO: Rome, Italy, 2012; p. 150. [Google Scholar]
- Waller, G.R.; Yamasaki, K. Saponins Used in Food and Agriculture; Plenum Press: New York, NY, USA, 1996; Volume 405. [Google Scholar]
- Hostettmann, K.; Marston, A. Saponins; Cambridge University Press: Cambridge, MA, USA, 1995. [Google Scholar]
- Elbandy, M.; Rho, J.; Afifi, R. Analysis of saponins as bioactive zoochemicals from the marine functional food sea cucumber Bohadschia cousteaui. Eur. Food Res. Technol. 2014. [Google Scholar] [CrossRef]
- Caulier, G.; van Dyck, S.; Gerbaux, P.; Eeckhaut, I.; Flammang, P. Review of saponin diversity in sea cucumbers belonging to the family Holothuriidae. SPC Beche-de-mer Inf. Bull. 2011, 31, 48–54. [Google Scholar]
- Dong, P.; Xue, C.; Du, Q. Separation of two main triterpene glycosides from sea cucumber Pearsonothuria graeffei by high-speed countercurrent chromatography. Acta Chromatogr. 2008, 20, 269–276. [Google Scholar] [CrossRef]
- Han, H.; Zhang, W.; Yi, Y.H.; Liu, B.S.; Pan, M.X.; Wang, X.H. A novel sulfated holostane glycoside from sea cucumber Holothuria leucospilota. Chem. Biodivers. 2010, 7, 1764–1769. [Google Scholar] [CrossRef]
- Naidu, A.S. Natural Food Antimicrobial Systems; CRC Press: New York, NY, USA, 2000. [Google Scholar]
- Zhang, S.L.; Li, L.; Yi, Y.H.; Sun, P. Philinopsides E and F, two new sulfated triterpene glycosides from the sea cucumber Pentacta quadrangularis. Nat. Prod. Res. 2006, 20, 399–407. [Google Scholar] [CrossRef]
- Zhang, S.L.; Li, L.; Yi, Y.H.; Zou, Z.R.; Sun, P. Philinopgenin A, B, and C, three new triterpenoid aglycones from the sea cucumber Pentacta quadrangulasis. Mar. Drugs 2004, 2, 185–191. [Google Scholar] [CrossRef]
- Zhang, S.Y.; Yi, Y.H.; Tang, H.F.; Li, L.; Sun, P.; Wu, J. Two new bioactive triterpene glycosides from the sea cucumber Pseudocolochirus violaceus. J. Asian Nat. Prod. Res. 2006, 8, 1–8. [Google Scholar] [CrossRef]
- Chludil, H.D.; Muniain, C.C.; Seldes, A.M.; Maier, M.S. Cytotoxic and antifungal triterpene glycosides from the Patagonian sea cucumber Hemoiedema spectabilis. J. Nat. Prod. 2002, 65, 860–865. [Google Scholar] [CrossRef]
- Francis, G.; Kerem, Z.; Makkar, H.P.; Becker, K. The biological action of saponins in animal systems: A review. Br. J. Nutr. 2002, 88, 587–605. [Google Scholar] [CrossRef]
- Maier, M.S.; Roccatagliata, A.J.; Kuriss, A.; Chludil, H.; Seldes, A.M.; Pujol, C.A.; Damonte, E.B. Two new cytotoxic and virucidal trisulfated triterpene glycosides from the Antarctic sea cucumber Staurocucumis liouvillei. J. Nat. Prod. 2001, 64, 732–736. [Google Scholar] [CrossRef]
- Osbourn, A.; Goss, R.J.M.; Field, R.A. The saponins-polar isoprenoids with important and diverse biological activities. Nat. Prod. Rep. 2011, 28, 1261–1268. [Google Scholar] [CrossRef]
- Jha, R.K.; Zi-rong, X. Biomedical Compounds from Marine organisms. Mar. Drugs 2004, 2, 123–146. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Aminin, D.L.; Avilov, S.A.; Silchenko, A.S.; Stonik, V.A. Triterpene glycosides from sea cucucmbers (Holothurioidea, Echinodermata). Biological activities and functions. In Studies in Natural Products Chemistry; Atta-ur, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2008; Volume 35, pp. 135–196. [Google Scholar]
- Liu, J.; Yang, X.; He, J.; ** of saponin congeners from three tropical holothurian sea cucumbers. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2013, 166, 182–193. [Google Scholar] [CrossRef]
- Van Dyck, S.; Caulier, G.; Todesco, M.; Gerbaux, P.; Fournier, I.; Wisztorski, M.; Flammang, P. The triterpene glycosides of Holothuria forskali: Usefulness and efficiency as a chemical defense mechanism against predatory fish. J. Exp. Biol. 2011, 214, 1347–1356. [Google Scholar] [CrossRef]
- Kalyani, G.A.; Kakrani, H.K.N.; Hukkeri, V.I. Holothurin—A Review. Indian J. Nat. Prod. 1988, 4, 3–8. [Google Scholar]
- Kalinin, V.; Anisimov, M.; Prokofieva, N.; Avilov, S.; Afiyatullov, S.S.; Stonik, V. Biological activities and biological role of triterpene glycosides from holothuroids (Echinodermata). Echinoderm Stud. 1996, 5, 139–181. [Google Scholar]
- Kalinin, V.I.; Prokofieva, N.G.; Likhatskaya, G.N.; Schentsova, E.B.; Agafonova, I.G.; Avilov, S.A.; Drozdova, O.A. Hemolytic activities of triterpene glycosides from the holothurian order Dendrochirotida: Some trends in the evolution of this group of toxins. Toxicon 1996, 34, 475–483. [Google Scholar] [CrossRef]
- Van Dyck, S.; Gerbaux, P.; Flammang, P. Elucidation of molecular diversity and body distribution of saponins in the sea cucumber Holothuria forskali (Echinodermata) by mass spectrometry. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2009, 152, 124–134. [Google Scholar] [CrossRef]
- Elyakov, G.B.; Stonik, V.A.; Levina, E.V.; Slanke, V.P.; Kuznetsova, T.A.; Levin, V.S. Glycosides of marine invertebrates—I. A comparative study of the glycoside fractions of pacific sea cucumbers. Comp. Biochem. Physiol. B Comp. Biochem. 1973, 44, 325–336. [Google Scholar] [CrossRef]
- Cai, Z.; Liu, S.; Asakawa, D. Applications of MALDI-TOF Spectroscopy; Springer: Berlin, Germany, 2013. [Google Scholar]
- Du, Q.; Jerz, G.; Waibel, R.; Winterhalter, P. Isolation of dammarane saponins from Panax notoginseng by high-speed counter-current chromatography. J. Chromatogr. 2003, 1008, 173–180. [Google Scholar] [CrossRef]
- Cui, M.; Song, F.; Zhou, Y.; Liu, Z.; Liu, S. Rapid identification of saponins in plant extracts by electrospray ionization multi-stage tandem mass spectrometry and liquid chromatography/tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2000, 14, 1280–1286. [Google Scholar] [CrossRef]
- Schöpke, T.; Thiele, H.; Wray, V.; Nimtz, M.; Hiller, K. Structure elucidation of a glycoside of 2β, 3β, t23-trihydroxy-16-oxoolean-12-en-28-oic acid from Bellis bernardii using mass spectrometry for the sugar sequence determination. J. Nat. Prod. 1995, 58, 152–155. [Google Scholar] [CrossRef]
- Bankefors, J.; Broberg, S.; Nord, L.I.; Kenne, L. Electrospray ionization ion-trap multiple-stage mass spectrometry of Quillaja saponins. J. Mass Spectrom. 2011, 46, 658–665. [Google Scholar] [CrossRef]
- Liu, S.; Cui, M.; Liu, Z.; Song, F.; Mo, W. Structural analysis of saponins from medicinal herbs using electrospray ionization tandem mass spectrometry. J. Am. Soc. Mass Spectrom. 2004, 15, 133–141. [Google Scholar] [CrossRef]
- Wang, X.; Sakuma, T.; Asafu-Adjaye, E.; Shiu, G.K. Determination of ginsenosides in plant extracts from Panax ginseng and Panax quinquefolius L. by LC/MS/MS. Anal. Chem. 1999, 71, 1579–1584. [Google Scholar] [CrossRef]
- Wolfender, J.L.; Rodriguez, S.; Hostettmann, K. Liquid chromatography coupled to mass spectrometry and nuclear magnetic resonance spectroscopy for the screening of plant constituents. J. Chromatogr. 1998, 794, 299–316. [Google Scholar] [CrossRef]
- Zheng, Z.; Zhang, W.; Kong, L.; Liang, M.; Li, H.; Lin, M.; Liu, R.; Zhang, C. Rapid identification of C21 steroidal saponins in Cynanchum versicolor Bunge by electrospray ionization multi-stage tandem mass spectrometry and liquid chromatography/tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2007, 21, 279–285. [Google Scholar]
- Cui, M.; Song, F.; Liu, Z.; Liu, S. Metal ion adducts in the structural analysis of ginsenosides by electrospray ionization with multi-stage mass spectrometry. Rapid Commun. Mass Spectrom. 2001, 15, 586–595. [Google Scholar] [CrossRef]
- Fang, S.; Hao, C.; Sun, W.; Liu, Z.; Liu, S. Rapid analysis of steroidal saponin mixture using electrospray ionization mass spectrometry combined with sequential tandem mass spectrometry. Rapid Commun. Mass Spectrom. 1998, 12, 589–594. [Google Scholar]
- Li, L. MALDI Mass Spectrometry for Synthetic Polymer Analysis; Wiley & Sons: Hoboken, NJ, USA, 2009; Volume 175. [Google Scholar]
- Silchenko, A.S.; Stonik, V.A.; Avilov, S.A.; Kalinin, V.I.; Kalinovsky, A.I.; Zaharenko, A.M.; Smirnov, A.V.; Mollo, E.; Cimino, G. Holothurins B2, B3, and B4, new triterpene glycosides from mediterranean sea cucumbers of the genus holothuria. J. Nat. Prod. 2005, 68, 564–567. [Google Scholar] [CrossRef]
- Han, H.; Yi, Y.H.; Li, L.; Wang, X.H.; Liu, B.S.; Sun, P.; Pan, M.X. A new triterpene glycoside from sea cucumber Holothuria leucospilota. Chin. Chem. Lett. 2007, 18, 161–164. [Google Scholar] [CrossRef]
- Kitagawa, I.; Nishino, T.; Matsuno, T.; Akutsu, H.; Kyogoku, Y. Structure of holothurin B a pharmacologically active triterpene-oligoglycoside from the sea cucumber Holothuria leucospilota Brandt. Tetrahedron Lett. 1978, 19, 985–988. [Google Scholar] [CrossRef]
- Han, H.; Yi, Y.H.; Liu, B.S.; Wang, X.H.; Pan, M.X. Leucospilotaside C, a new sulfated triterpene glycoside from sea cucumber Holothuria leucospilota. Chin. Chem. Lett. 2008, 19, 1462–1464. [Google Scholar] [CrossRef]
- Wu, J.; Yi, Y.H.; Tang, H.F.; Wu, H.M.; Zou, Z.R.; Lin, H.W. Nobilisides A–C, three new triterpene glycosides from the sea cucumber Holothuria nobilis. Planta Med. 2006, 72, 932–935. [Google Scholar] [CrossRef]
- Rodriguez, J.; Castro, R.; Riguera, R. Holothurinosides: New antitumour non sulphated triterpenoid glycosides from the sea cucumber Holothuria forskalii. Tetrahedron 1991, 47, 4753–4762. [Google Scholar] [CrossRef]
- Kitagawa, I.; Kobayashi, M.; Kyogoku, Y. Marine natural products. IX. Structural elucidation of triterpenoidal oligoglycosides from the Bahamean sea cucumber Actinopyga agassizi Selenka. Chem. Pharm. Bull. (Tokyo) 1982, 30, 2045–2050. [Google Scholar] [CrossRef]
- Liu, B.S.; Yi, Y.H.; Li, L.; Sun, P.; Han, H.; Sun, G.Q.; Wang, X.H.; Wang, Z.L. Argusides D and E, two new cytotoxic triterpene glycosides from the sea cucumber Bohadschia argus Jaeger. Chem. Biodivers. 2008, 5, 1425–1433. [Google Scholar] [CrossRef]
- Han, H.; Yi, Y.H.; Li, L.; Liu, B.S.; La, M.P.; Zhang, H.W. Antifungal active triterpene glycosides from sea cucumber Holothuria scabra. Acta Pharm. Sin. 2009, 44, 620–624. [Google Scholar]
- Han, H.; Li, L.; Yi, Y.-H.; Wang, X.-H.; Pan, M.-X. Triterpene glycosides from sea cucumber Holothuria scabra with cytotoxic activity. Chin. Herbal Med. 2012, 4, 183–188. [Google Scholar]
- Kalinin, V.; Stonik, V. Glycosides of marine invertebrates. Structure of Holothurin A2 from the holothurian Holothuria edulis. Chem. Nat. Compd. 1982, 18, 196–200. [Google Scholar] [CrossRef]
- Kitagawa, I.; Kobayashi, M.; Inamoto, T.; Fuchida, M.; Kyogoku, Y. Marine natural products. XIV. Structures of echinosides A and B, antifungal lanostane-oligosides from the sea cucumber Actinopyga echinites (Jaeger). Chem. Pharm. Bull. (Tokyo) 1985, 33, 5214–5224. [Google Scholar] [CrossRef]
- Thanh, N.V.; Dang, N.H.; Kiem, P.V.; Cuong, N.X.; Huong, H.T.; Minh, C.V. A new triterpene glycoside from the sea cucumber Holothuria scabra collected in Vietnam. ASEAN J. Sci. Technol. Dev. 2006, 23, 253–259. [Google Scholar]
- Kitagawa, I.; Nishino, T.; Kyogoku, Y. Structure of holothurin A a biologically active triterpene-oligoglycoside from the sea cucumber Holothuria leucospilota Brandt. Tetrahedron Lett. 1979, 20, 1419–1422. [Google Scholar] [CrossRef]
- Yuan, W.; Yi, Y.; Tang, H.; Xue, M.; Wang, Z.; Sun, G.; Zhang, W.; Liu, B.; Li, L.; Sun, P. Two new holostan-type triterpene glycosides from the sea cucumber Bohadschia marmorata JAEGER. Chem. Pharm. Bull. (Tokyo) 2008, 56, 1207–1211. [Google Scholar] [CrossRef]
- Yuan, W.H.; Yi, Y.H.; Tan, R.X.; Wang, Z.L.; Sun, G.Q.; Xue, M.; Zhang, H.W.; Tang, H.F. Antifungal triterpene glycosides from the sea cucumber Holothuria (Microthele) axiloga. Planta Med. 2009, 75, 647–653. [Google Scholar] [CrossRef]
- Sun, G.Q.; Li, L.; Yi, Y.H.; Yuan, W.H.; Liu, B.S.; Weng, Y.Y.; Zhang, S.L.; Sun, P.; Wang, Z.L. Two new cytotoxic nonsulfated pentasaccharide holostane (=20-hydroxylanostan-18-oic acid γ-lactone) glycosides from the sea cucumber Holothuria grisea. Helv. Chim. Acta 2008, 91, 1453–1460. [Google Scholar] [CrossRef]
- Song, F.; Cui, M.; Liu, Z.; Yu, B.; Liu, S. Multiple-stage tandem mass spectrometry for differentiation of isomeric saponins. Rapid Commun. Mass Spectrom. 2004, 18, 2241–2248. [Google Scholar] [CrossRef]
- Van Setten, D.C.; Jan ten Hove, G.; Wiertz, E.J.H.J.; Kamerling, J.P.; van de Werken, G. Multiple-stage tandem mass spectrometry for structural characterization of saponins. Anal. Chem. 1998, 70, 4401–4409. [Google Scholar] [CrossRef]
- Garneau, F.X.; Simard, J.; Harvey, O.; ApSimon, J.; Girard, M. The structure of psoluthurin A, the major triterpene glycoside of the sea cucumber Psolus fabricii. Can. J. Chem. 1983, 61, 1465–1471. [Google Scholar] [CrossRef]
- Grassia, A.; Bruno, I.; Debitus, C.; Marzocco, S.; Pinto, A.; Gomez-Paloma, L.; Riccio, R. Spongidepsin, a new cytotoxic macrolide from Spongia sp. Tetrahedron 2001, 57, 6257–6260. [Google Scholar] [CrossRef]
- Kupchan, S.M.; Britton, R.W.; Ziegler, M.F.; Sigel, C.W. Bruceantin, a new potent antileukemic simaroubolide from Brucea antidysenterica. J. Org. Chem. 1973, 38, 178–179. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bahrami, Y.; Zhang, W.; Franco, C. Discovery of Novel Saponins from the Viscera of the Sea Cucumber Holothuria lessoni. Mar. Drugs 2014, 12, 2633-2667. https://doi.org/10.3390/md12052633
Bahrami Y, Zhang W, Franco C. Discovery of Novel Saponins from the Viscera of the Sea Cucumber Holothuria lessoni. Marine Drugs. 2014; 12(5):2633-2667. https://doi.org/10.3390/md12052633
Chicago/Turabian StyleBahrami, Yadollah, Wei Zhang, and Chris Franco. 2014. "Discovery of Novel Saponins from the Viscera of the Sea Cucumber Holothuria lessoni" Marine Drugs 12, no. 5: 2633-2667. https://doi.org/10.3390/md12052633