Structural Characterization and Spatial Map** of Tetrodotoxins in Australian Polyclads
Abstract
:1. Introduction
2. Results
2.1. Separation and Identification of TTX and Its Analogues Using HILIC-HRMS
2.2. Optimization of Flatworm Sample Preparation for TTX MALDI-MSI
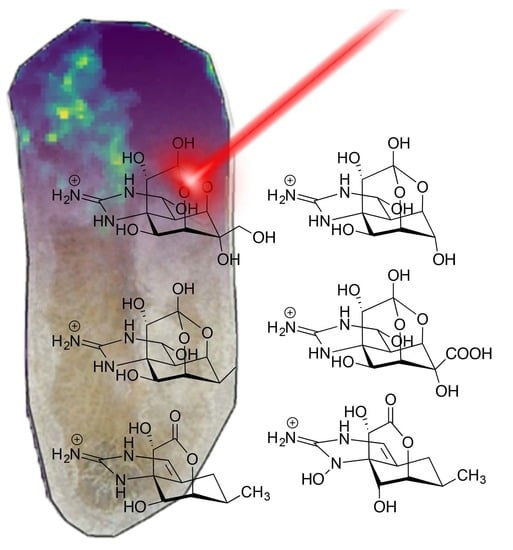
2.3. MALDI-MSI of Freeze Dried Stylochus sp. 1 Flatworms
3. Discussion
4. Materials and Methods
4.1. Flatworm Specimen Collection
4.2. Tetrodotoxin Analogue Separation and Analysis from Flatworms Using Hydrophilic Interaction Liquid Chromatography Mass Spectrometry
4.3. Spatial Map** of TTX across Flatworms Using Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry Imaging
4.4. Calibrant and Matrix Deposition
4.5. MALDI-TOF/TOF MS Data Acquisition
4.6. MALDI-TOF/TOF MS Data Analysis
4.7. Flatworm Specimen Identification
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Paul, V.J.; Puglisi, M.P. Chemical mediation of interactions among marine organisms. Nat. Prod. Rep. 2004, 21, 189–209. [Google Scholar] [CrossRef] [PubMed]
- Williamson, J.E.; De Nys, R.; Kumar, N.; Carson, D.G.; Steinberg, P.D. Induction of metamorphosis in the sea urchin Holopneustes purpurascens by a metabolite complex from the algal host Delisea pulchra. Biol. Bull. 2000, 198, 332–345. [Google Scholar] [CrossRef] [PubMed]
- Williamson, J.E.; Carson, D.G.; De Nys, R.; Steinberg, P.D. Demographic consequences of an ontogenetic shift by a sea urchin in response to host plant chemistry. Ecology 2004, 85, 1355–1371. [Google Scholar] [CrossRef]
- Karuso, P. Chemical ecology of the nudibranchs. In Bioorganic Marine Chemistry; Springer: Berlin/Heidelberg, Germany, 1987; pp. 31–60. [Google Scholar]
- Paul, V.J.; Ritson-Williams, R. Marine chemical ecology. Nat. Prod. Rep. 2008, 25, 662–695. [Google Scholar] [CrossRef] [PubMed]
- Puglisi, M.P.; Sneed, J.M.; Sharp, K.H.; Ritson-Williams, R.; Paul, V.J. Marine chemical ecology in benthic environments. Nat. Prod. Rep. 2014, 31, 1510–1553. [Google Scholar] [CrossRef] [PubMed]
- Patockaa, J.; Stredab, L. Brief review of natural nonprotein neurotoxins. ASA Newslett. 2002, 89, 16–24. [Google Scholar]
- Wang, D.Z. Neurotoxins from marine dinoflagellates: A brief review. Mar. Drugs 2008, 6, 349–371. [Google Scholar] [CrossRef] [PubMed]
- Catterall, W.A. Neurotoxins that act on voltage-sensitive sodium channels in excitable membranes. Annu. Rev. Pharmacol. Toxicol. 1980, 20, 15–43. [Google Scholar] [CrossRef]
- Bane, V.; Lehane, M.; Dikshit, M.; O’Riordan, A.; Furey, A. Tetrodotoxin: Chemistry, toxicity, source, distribution and detection. Toxins 2014, 6, 693–755. [Google Scholar] [CrossRef] [Green Version]
- Chau, R.; Kalaitzis, J.A.; Neilan, B.A. On the origins and biosynthesis of tetrodotoxin. Aquat. Toxicol. 2011, 104, 61–72. [Google Scholar] [CrossRef]
- Kaneko, Y.; Matsumoto, G.; Hanyu, Y. TTX resistivity of Na+ channel in newt retinal neuron. Biochem. Biophys. Res. Commun. 1997, 240, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, T.; Arakawa, O.; Takatani, T. TTX accumulation in pufferfish. Comp. Biochem. Physiol. Part D Genom. Proteom. 2006, 1, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Finefield, J.M.; Frisvad, J.C.; Sherman, D.H.; Williams, R.M. Fungal origins of the bicyclo[2.2.2]diazaoctane ring system of prenylated indole alkaloids. J. Nat. Prod. 2012, 75, 812–833. [Google Scholar] [CrossRef] [Green Version]
- Matsumura, K. Tetrodotoxin as a pheromone. Nature 1995, 378, 563–564. [Google Scholar] [CrossRef]
- Hwang, P.A.; Noguchi, T.; Hwang, D.F. Neurotoxin tetrodotoxin as attractant for toxic snails. Fish. Sci. 2004, 70, 1106–1112. [Google Scholar] [CrossRef]
- Tsai, Y.H.; Ho, P.H.; Hwang, C.C.; Hwang, P.A.; Cheng, C.A.; Hwang, D.F. Tetrodotoxin in several species of xanthid crabs in southern Taiwan. Food Chem. 2006, 95, 205–212. [Google Scholar] [CrossRef]
- Williams, B.L. Behavioral and chemical ecology of marine organisms with respect to tetrodotoxin. Mar. Drugs 2010, 8, 381–398. [Google Scholar] [CrossRef] [Green Version]
- Salvitti, L.R.; Wood, S.A.; Winsor, L.; Cary, S.C. Intracellular immunohistochemical detection of tetrodotoxin in Pleurobranchaea maculata (Gastropoda) and Stylochoplana sp. (Turbellaria). Mar. Drugs 2015, 13, 756–769. [Google Scholar] [CrossRef] [Green Version]
- Ferrer, R.P.; Zimmer, R.K. Molecules of Keystone Significance: Crucial Agents in Ecology and Resource Management. Bioscience 2013, 63, 428–438. [Google Scholar] [CrossRef] [Green Version]
- Zimmer, R.K.; Ferrer, R.P. Neuroecology, chemical defense, and the keystone species concept. Biol. Bull. 2007, 213, 208–225. [Google Scholar] [CrossRef]
- McNab, J.M.; Rodriguez, J.; Karuso, P.; Williamson, J.E. Natural Products in Polyclad Flatworms. Mar. Drugs 2021, 19, 47. [Google Scholar] [CrossRef] [PubMed]
- Ritson-Williams, R.; Yotsu-Yamashita, M.; Paul, V.J. Ecological functions of tetrodotoxin in a deadly polyclad flatworm. Proc. Natl. Acad. Sci. USA 2006, 103, 3176–3179. [Google Scholar] [CrossRef] [Green Version]
- Yotsu-Yamashita, M.; Abe, Y.; Kudo, Y.; Ritson-Williams, R.; Paul, V.J.; Konoki, K.; Cho, Y.; Adachi, M.; Imazu, T.; Nishikawa, T.; et al. First identification of 5,11-dideoxytetrodotoxin in marine animals, and characterization of major fragment ions of tetrodotoxin and its analogs by high resolution ESI-MS/MS. Mar. Drugs 2013, 11, 2799–2813. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kashitani, M.; Okabe, T.; Oyama, H.; Noguchi, K.; Yamazaki, H.; Suo, R.; Mori, T.; Sugita, H.; Itoi, S. Taxonomic Distribution of Tetrodotoxin in Acotylean Flatworms (Polycladida: Platyhelminthes). Mar. Biotechnol. 2020, 22, 805–811. [Google Scholar] [CrossRef] [PubMed]
- Prudhoe, S. Some polyclad turbellarians new to the fauna of the Australian coasts. Rec. Aust. Mus. 1978, 31, 586–604. [Google Scholar] [CrossRef]
- Newman, L.J.; Cannon, L.R.G. Biodiversity of tropical polyclad flatworms from the Great Barrier Reef, Australia. Mem. Queensl. Mus. 1994, 36, 159–163. [Google Scholar]
- Newman, L.J.; Cannon, L.R.G. Marine Flatworms: The world of Polyclads; CSIRO Publishing: Clayton, Australia, 2003. [Google Scholar]
- Rodrguez, J.; Hutchings, P.A.; Williamson, J.E. Biodiversity of intertidal marine flatworms (Polycladida, Platyhelminthes) in southeastern Australia. Zootaxa 2021, 5024, 1–63. [Google Scholar] [CrossRef]
- McNabb, P.; Selwood, A.I.; Munday, R.; Wood, S.A.; Taylor, D.I.; Mackenzie, L.A.; van Ginkel, R.; Rhodes, L.L.; Cornelisen, C.; Heasman, K.; et al. Detection of tetrodotoxin from the grey side-gilled sea slug—Pleurobranchaea maculata, and associated dog neurotoxicosis on beaches adjacent to the Hauraki Gulf, Auckland, New Zealand. Toxicon 2010, 56, 466–473. [Google Scholar] [CrossRef]
- Nakamura, M.; Yasumoto, T. Tetrodotoxin derivatives in puffer fish. Toxicon 1985, 23, 271–276. [Google Scholar] [CrossRef]
- Mahmud, Y.; Okada, K.; Takatani, T.; Kawatsu, K.; Hamano, Y.; Arakawa, O.; Noguchi, T. Intra-tissue distribution of tetrodotoxin in two marine puffers Takifugu vermicularis and Chelonodon patoca. Toxicon 2003, 41, 13–18. [Google Scholar] [CrossRef]
- Tanu, M.B.; Mahmud, Y.; Arakawa, O.; Takatani, T.; Kajihara, H.; Kawatsu, K.; Hamano, Y.; Asakawa, M.; Miyazawa, K.; Noguchi, T. Immunoenzymatic visualization of tetrodotoxin (TTX) in Cephalothrix species (Nemertea: Anopla: Palaeonemertea: Cephalotrichidae) and Planocera reticulata (Platyhelminthes: Turbellaria: Polycladida: Planoceridae). Toxicon 2004, 44, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Rudd, D.; Ronci, M.; Johnston, M.R.; Guinan, T.; Voelcker, N.H.; Benkendorff, K. Mass spectrometry imaging reveals new biological roles for choline esters and Tyrian purple precursors in muricid molluscs. Sci. Rep. 2015, 5, 13408. [Google Scholar] [CrossRef] [PubMed]
- Kaspar, S.; Peukert, M.; Svatos, A.; Matros, A.; Mock, H.P. MALDI-imaging mass spectrometry–an emerging technique in plant biology. Proteomics 2011, 11, 1840–1850. [Google Scholar] [CrossRef] [PubMed]
- Castellino, S.; Groseclose, M.R.; Wagner, D. MALDI imaging mass spectrometry: Bridging biology and chemistry in drug development. Bioanalysis 2011, 3, 2427–2441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dutertre, S.; **, A.-H.; Vetter, I.; Hamilton, B.; Sunagar, K.; Lavergne, V.; Dutertre, V.; Fry, B.G.; Antunes, A.; Venter, D.J. Evolution of separate predation-and defence-evoked venoms in carnivorous cone snails. Nat. Commun. 2014, 5, 3521. [Google Scholar] [CrossRef] [Green Version]
- Ronci, M.; Rudd, D.; Guinan, T.; Benkendorff, K.; Voelcker, N.H. Mass spectrometry imaging on porous silicon: Investigating the distribution of bioactives in marine mollusc tissues. Anal. Chem. 2012, 84, 8996–9001. [Google Scholar] [CrossRef]
- Bane, V.; Brosnan, B.; Barnes, P.; Lehane, M.; Furey, A. High-resolution mass spectrometry analysis of tetrodotoxin (TTX) and its analogues in puffer fish and shellfish. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2016, 33, 1468–1489. [Google Scholar] [CrossRef]
- Vlasenko, A.E.; Magarlamov, T.Y. Tetrodotoxin and its analogues in Cephalothrix cf. simula (Nemertea: Palaeonemertea) from the sea of Japan (Peter the Great Gulf): Intrabody distribution and secretions. Toxins 2020, 12, 745. [Google Scholar] [CrossRef]
- Yotsu-Yamashita, M.; Sugimoto, A.; Takai, A.; Yasumoto, T. Effects of Specific Modifications of Several Hydroxyls of Tetrodotoxin on Its Affinity to Rat Brain Membrane. J. Pharmacol. Exp. Ther. 1999, 289, 1688. [Google Scholar]
- Lin, S.J.; Chai, T.J.; Jeng, S.S.; Hwang, D.F. Toxicity of the puffer Takifugu rubripes cultured in northern Taiwan. Fish. Sci. 1998, 64, 766–770. [Google Scholar] [CrossRef] [Green Version]
- Hyman, L.H. The Invertebrates: Platyhelminthes and Rhynchocoela. The Acoelomate Bilateria; McGraw Hill Book Company: New York, NY, USA, 1951; Volume 2, p. 550. [Google Scholar]
- Galleni, L.; Tongiorgi, P.; Ferrero, E.; Salghetti, U. Stylochus mediterraneus (Turbellaria: Polycladida), predator on the mussel Mytilus galloprovincialis. Mar. Biol. 1980, 55, 317–326. [Google Scholar] [CrossRef]
- Newman, L.J.; Cannon, L.R.G.; Govan, H. Stylochus-(Imogene)-Matatasi N-Sp (Platyhelminthes, Polycladida)—Pest of Cultured Giant Clams and Pearl Oysters from Solomon-Islands. Hydrobiologia 1993, 257, 185–189. [Google Scholar] [CrossRef]
- Lee, K.M.; Beal, M.A.; Johnston, E.L. A new predatory flatworm (Platyhelminthes, Polycladida) from Botany Bay, New South Wales, Australia. J. Nat. Hist. 2006, 39, 3987–3995. [Google Scholar] [CrossRef]
- O’Connor, W.A.; Newman, L.J. Halotolerance of the oyster predator, Imogine mcgrathi, a stylochid flatworm from Port Stephens, New South Wales, Australia. Hydrobiologia 2001, 459, 157–163. [Google Scholar] [CrossRef]
- Sluys, R.; Faubel, A.; Rajagopal, S.; Van Der Velde, G. A new and alien species of “oyster leech”(Platyhelminthes, Polycladida, Stylochidae) from the brackish North Sea Canal, The Netherlands. Helgol. Mar. Res. 2005, 59, 310–314. [Google Scholar] [CrossRef] [Green Version]
- Bane, V.; Hutchinson, S.; Sheehan, A.; Brosnan, B.; Barnes, P.; Lehane, M.; Furey, A. LC-MS/MS method for the determination of tetrodotoxin (TTX) on a triple quadruple mass spectrometer. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2016, 33, 1728–1740. [Google Scholar] [CrossRef]
- Oya, Y.; Kajihara, H. Molecular Phylogenetic Analysis of Acotylea (Platyhelminthes: Polycladida). Zoolog. Sci. 2020, 37, 271–279. [Google Scholar] [CrossRef]
- Whitelaw, B.L.; Cooke, I.R.; Finn, J.; Zenger, K.; Strugnell, J.M. The evolution and origin of tetrodotoxin acquisition in the blue-ringed octopus (genus Hapalochlaena). Aquat. Toxicol. 2019, 206, 114–122. [Google Scholar] [CrossRef]
- Kogure, K.; Do, H.K.; Thuesen, E.V.; Nanba, K.; Ohwada, K.; Simidu, U. Accumulation of Tetrodotoxin in Marine Sediment. Mar. Ecol. Prog. Ser. 1988, 45, 303–305. [Google Scholar] [CrossRef]
- Do, H.K.; Kogure, K.; Simidu, U. Identification of deep-sea-sediment bacteria which produce tetrodotoxin. Appl. Environ. Microbiol. 1990, 56, 1162–1163. [Google Scholar] [CrossRef] [Green Version]
- Do, H.K.; Kogure, K.; Imada, C.; Noguchi, T.; Ohwada, K.; Simidu, U. Tetrodotoxin production of actinomycetes isolated from marine sediment. J. Appl. Bacteriol. 1991, 70, 464–468. [Google Scholar] [CrossRef]




| Specimen | TTX (1) (Scan Channel m/z 320.1088) RT (min) | 11-Deoxy TTX (2) (Scan Channel m/z 304.1139) RT (min) | 11-norTTX-6(S)-ol (3) (Scan Channel m/z 290.0983) RT (min) | 6,11-Dideoxy TTX (4) (Scan Channel m/z 288.1190) RT (min) | Unknown Compound #1 (Scan Channel m/z 270.1085) RT (min) | 4,4a-Anhydro-5,6,11-trideoxy TTX (9) (Scan Channel m/z 254.1135) RT (min) | Unknown Compound #2 (Scan Channel m/z 334.0881) RT (min) |
|---|---|---|---|---|---|---|---|
| Stylochus sp. 1 1 | 6.11 | 7.58 | 7.55 | ||||
| Stylochus sp. 1 1 | 6.15 | 7.58 | |||||
| Stylochus cf mcgrathi 2 | 18.01 | 6.06 | 7.61 | 4.91 | 8.59 | ||
| Stylochus cf mcgrathi 2 | 17.41 | 7.55 | 4.86 | 8.56 | |||
| Stylochus cf mcgrathi 2 | 18.29 | 6.02 | 7.61 | 4.75 | 8.47 | ||
| Stylochus cf mcgrathi 2 | 18.31 | 6.19 | 7.58 | 4.82 | 8.44 | 7.57 | |
| Stylochus cf mcgrathi 2 | 18.25 | 6.15 | 7.61 | 8.61 | |||
| Stylochus cf mcgrathi 3 | 17.55 | 7.55 | 4.78 | 8.54 | 7.54 | ||
| Stylochus cf mcgrathi 3 | 7.62 | 4.89 | 8.70 | ||||
| Stylochus cf mcgrathi 3 | 7.61 | 4.89 | 8.58 | 7.57 | |||
| Stylochus cf mcgrathi 3 | 7.59 | 4.89 | 8.60 | 8.01 | 7.59 | ||
| Stylochus sp. 3 4 | |||||||
| Stylochus sp. 4 1 | |||||||
| Echinoplana cf celerrima 5 | 7.56 | 4.80 | |||||
| Echinoplana cf celerrima 1 | 7.56 | 4.84 | |||||
| Echinoplana cf celerrima 1 | 7.58 | 4.84 | 7.54 | ||||
| Echinoplana cf celerrima 1 | 6.14 | 7.63 | 4.78 | 7.55 | |||
| Notoplana cf longiducta 5 | 7.56 | 4.78 | |||||
| Pseudoceros sp. 1 1 | 7.58 | 4.91 | 8.49 | ||||
| Pseudoceros sp. 2 1 | 6.20 | ||||||
| Pseudoceros sp. 3 1 | 7.55 | 8.48 | |||||
| Pseudoceros cf velutinus 4 | 7.62 | 4.84 | 8.60 | ||||
| Pseudoceros cf velutinus 4 | 6.17 | 7.53 | |||||
| Thysanozoon brocchii4 | 7.58 | 8.48 | |||||
| Thysanozoon brocchii4 | 7.61 | 8.58 | |||||
| Eurylepta sp. 1 | 8.03 | ||||||
| Cycloporus sp. 1 | 7.60 | 4.85 | |||||
| Cestoplana cf rubocincta 5 | 7.49 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McNab, J.M.; Briggs, M.T.; Williamson, J.E.; Hoffmann, P.; Rodriguez, J.; Karuso, P. Structural Characterization and Spatial Map** of Tetrodotoxins in Australian Polyclads. Mar. Drugs 2022, 20, 788. https://doi.org/10.3390/md20120788
McNab JM, Briggs MT, Williamson JE, Hoffmann P, Rodriguez J, Karuso P. Structural Characterization and Spatial Map** of Tetrodotoxins in Australian Polyclads. Marine Drugs. 2022; 20(12):788. https://doi.org/10.3390/md20120788
Chicago/Turabian StyleMcNab, Justin M., Matthew T. Briggs, Jane E. Williamson, Peter Hoffmann, Jorge Rodriguez, and Peter Karuso. 2022. "Structural Characterization and Spatial Map** of Tetrodotoxins in Australian Polyclads" Marine Drugs 20, no. 12: 788. https://doi.org/10.3390/md20120788