Review on Nanoparticles and Nanostructured Materials: Bioimaging, Biosensing, Drug Delivery, Tissue Engineering, Antimicrobial, and Agro-Food Applications
Abstract
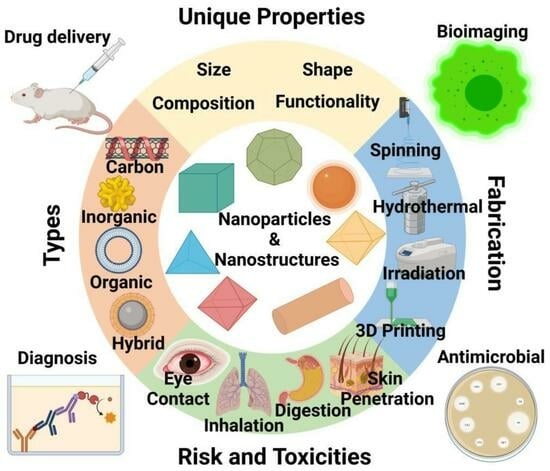
:1. Introduction
2. Fluorescent Nanomaterials for Bioimaging
3. Nano-Drug Delivery Systems
3.1. Nano-Vehicles for Anticancer Drugs
3.2. Nanostructured Materials as Drug Delivery Vehicles for Antioxidant Drugs
4. Antimicrobial Materials
5. Gene Therapy
6. Biosensors
7. Tissue Engineering
8. Agriculture and Food Industry
9. Risks of Exposure to Nanomaterials
10. Global Market and Future of Nanomaterials
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gaur, M.; Misra, C.; Yadav, A.B.; Swaroop, S.; Maolmhuaidh, F.; Bechelany, M.; Barhoum, A. Biomedical Applications of Carbon Nanomaterials: Fullerenes, Quantum Dots, Nanotubes, Nanofibers, and Graphene. Materials 2021, 14, 5978. [Google Scholar] [CrossRef] [PubMed]
- Barhoum, A.; Pal, K.; Rahier, H.; Uludag, H.; Kim, I.S.; Bechelany, M. Nanofibers as new-generation materials: From spinning and nano-spinning fabrication techniques to emerging applications. Appl. Mater. Today 2019, 17, 1–35. [Google Scholar] [CrossRef]
- Jeevanandam, J.; Barhoum, A.; Chan, Y.S.; Dufresne, A.; Danquah, M.K. Review on nanoparticles and nanostructured materials: History, sources, toxicity and regulations. Beilstein J. Nanotechnol. 2018, 9, 1050–1074. [Google Scholar] [CrossRef]
- Barhoum, A.; El-Maghrabi, H.H.; Nada, A.A.; Sayegh, S.; Roualdes, S.; Renard, A.; Iatsunskyi, I.; Coy, E.; Bechelany, M. Simultaneous hydrogen and oxygen evolution reactions using free-standing nitrogen-doped-carbon–Co/CoOx nanofiber electrodes decorated with palladium nanoparticles. J. Mater. Chem. A 2021, 9, 17724–17739. [Google Scholar] [CrossRef]
- Prasad, S.; Kumar, V.; Kirubanandam, S.; Barhoum, A. Engineered nanomaterials: Nanofabrication and surface functionalization. In Emerging Applications of Nanoparticles and Architecture Nanostructures: Current Prospects and Future Trends; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 305–340. [Google Scholar] [CrossRef]
- Cremers, V.; Rampelberg, G.; Barhoum, A.; Walters, P.; Claes, N.; de Oliveira, T.M.; Van Assche, G.; Bals, S.; Dendooven, J.; Detavernier, C. Oxidation barrier of Cu and Fe powder by Atomic Layer Deposition. Surf. Coat. Technol. 2018, 349, 1032–1041. [Google Scholar] [CrossRef]
- Hammani, S.; Moulai-Mostefa, N.; Samyn, P.; Bechelany, M.; Dufresne, A.; Barhoum, A. Morphology, Rheology and Crystallization in Relation to the Viscosity Ratio of Polystyrene/Polypropylene Polymer Blends. Materials 2020, 13, 926. [Google Scholar] [CrossRef]
- Barhoum, A.; Van Lokeren, L.; Rahier, H.; Dufresne, A.; Van Assche, G. Roles of in situ surface modification in controlling the growth and crystallization of CaCO3 nanoparticles, and their dispersion in polymeric materials. J. Mater. Sci. 2015, 50, 7908–7918. [Google Scholar] [CrossRef]
- Rehan, M.; Barhoum, A.; Khattab, T.; Gätjen, L.; Wilken, R. Colored, photocatalytic, antimicrobial and UV-protected viscose fibers decorated with Ag/Ag2CO3 and Ag/Ag3PO4 nanoparticles. Cellulose 2019, 26, 5437–5453. [Google Scholar] [CrossRef]
- Abdel-Haleem, F.M.; Salah, A.; Rizk, M.S.; Moustafa, H.; Bechelany, M.; Barhoum, A. Carbon-based Nanosensors for Salicylate Determination in Pharmaceutical Preparations. Electroanalysis 2019, 31, 778–789. [Google Scholar] [CrossRef]
- Abdel-Haleem, F.; Mahmoud, S.; Abdel-Ghani, N.; El Nashar, R.; Bechelany, M.; Barhoum, A. Polyvinyl Chloride Modified Carbon Paste Electrodes for Sensitive Determination of Levofloxacin Drug in Serum, Urine, and Pharmaceutical Formulations. Sensors 2021, 21, 3150. [Google Scholar] [CrossRef]
- Abdel-Haleem, F.M.; Gamal, E.; Rizk, M.S.; Madbouly, A.; El Nashar, R.M.; Anis, B.; Elnabawy, H.M.; Khalil, A.S.G.; Barhoum, A. Molecularly Imprinted Electrochemical Sensor-Based Fe2O3@MWCNTs for Ivabradine Drug Determination in Pharmaceutical Formulation, Serum, and Urine Samples. Front. Bioeng. Biotechnol. 2021, 9, 648704. [Google Scholar] [CrossRef] [PubMed]
- Parikha Mehrotra, Biosensors and their applications—A review. J. Oral Biol. Craniofac. Res. 2016, 6, 153–159. [CrossRef] [PubMed]
- Rasouli, R.; Barhoum, A.; Uludag, H. A review of nanostructured surfaces and materials for dental implants: Surface coating, patterning and functionalization for improved performance. Biomater. Sci. 2018, 6, 1312–1338. [Google Scholar] [CrossRef] [PubMed]
- Rasouli, R.; Barhoum, A.; Bechelany, M.; Dufresne, A. Nanofibers for Biomedical and Healthcare Applications. Macromol. Biosci. 2018, 19, e1800256. [Google Scholar] [CrossRef]
- Singh, K.R.; Nayak, V.; Singh, J.; Singh, A.K.; Singh, R.P. Potentialities of bioinspired metal and metal oxide nanoparticles in biomedical sciences. RSC Adv. 2021, 11, 24722–24746. [Google Scholar] [CrossRef]
- Tan, K.X.; Barhoum, A.; Pan, S.; Danquah, M.K. Risks and toxicity of nanoparticles and nanostructured materials. In Emerging Applications of Nanoparticles and Architecture Nanostructures: Current Prospects and Future Trends; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 121–139. [Google Scholar] [CrossRef]
- Kim, D.; Kim, J.; Park, Y.I.; Lee, N.; Hyeon, T. Recent Development of Inorganic Nanoparticles for Biomedical Imaging. ACS Central Sci. 2018, 4, 324–336. [Google Scholar] [CrossRef]
- Mihai, M.M.; Dima, M.B.; Dima, B.; Holban, A.M. Nanomaterials for Wound Healing and Infection Control. Materials 2019, 12, 2176. [Google Scholar] [CrossRef]
- Said, M.M.; Rehan, M.; El-Sheikh, S.M.; Zahran, M.K.; Abdel-Aziz, M.S.; Bechelany, M.; Barhoum, A. Multifunctional Hydroxyapatite/Silver Nanoparticles/Cotton Gauze for Antimicrobial and Biomedical Applications. Nanomaterials 2021, 11, 429. [Google Scholar] [CrossRef]
- Kumar, S.; Bhushan, P.; Bhattacharya, S. Fabrication of Nanostructures with Bottom-up Approach and Their Utility in Diagnostics, Therapeutics, and Others. In Environmental, Chemical and Medical Sensors; Springer: Berlin/Heidelberg, Germany, 2017; pp. 167–198. [Google Scholar] [CrossRef]
- Sawy, A.M.; Barhoumbb, A.; Gaber, S.A.A.; El-Hallouty, S.M.; Shousha, W.G.; Maarouf, A.A.; Khalilaf, S.G.A. Insights of doxorubicin loaded graphene quantum dots: Synthesis, DFT drug interactions, and cytotoxicity. Mater. Sci. Eng. C 2021, 122, 111921. [Google Scholar] [CrossRef]
- Barhoum, A.; Van Assche, G.; Rahier, H.; Fleisch, M.; Bals, S.; Delplancked, M.-P.; Leroux, F.; Bahnemann, D. Sol-gel hot injection synthesis of ZnO nanoparticles into a porous silica matrix and reaction mechanism. Mater. Des. 2017, 119, 270–276. [Google Scholar] [CrossRef]
- Barhoum, A.; Melcher, J.; Van Assche, G.; Rahier, H.; Bechelany, M.; Fleisch, M.; Bahnemann, D. Synthesis, growth mechanism, and photocatalytic activity of Zinc oxide nanostructures: Porous microparticles versus nonporous nanoparticles. J. Mater. Sci. 2016, 52, 2746–2762. [Google Scholar] [CrossRef]
- Hong, G.; Antaris, A.L.; Dai, H. Near-infrared fluorophores for biomedical imaging. Nat. Biomed. Eng. 2017, 1, 10. [Google Scholar] [CrossRef]
- Malik, N.; Arfin, T.; Khan, A.U. Graphene nanomaterials: Chemistry and pharmaceutical perspectives. In Nanomaterials for Drug Delivery and Therapy; Grumezescu, T., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 373–402. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, L.; Wan, B.; Gu, Y.; Li, X. Optically Active Nanomaterials for Bioimaging and Targeted Therapy. Front. Bioeng. Biotechnol. 2019, 7, 320. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Kang, P.M. Systemic Review of Biodegradable Nanomaterials in Nanomedicine. Nanomaterials 2020, 10, 656. [Google Scholar] [CrossRef]
- Siafaka, P.I.; Okur, N.Ü.; Karantas, I.D.; Okur, M.E.; Gündoğdu, E.A. Current update on nanoplatforms as therapeutic and diagnostic tools: A review for the materials used as nanotheranostics and imaging modalities. Asian J. Pharm. Health Sci. 2021, 16, 24–46. [Google Scholar] [CrossRef]
- Yoon, H.Y.; Jeon, S.; You, D.G.; Park, J.H.; Kwon, I.C.; Koo, H.; Kim, K. Inorganic Nanoparticles for Image-Guided Therapy. Bioconjug. Chem. 2017, 28, 124–134. [Google Scholar] [CrossRef]
- Snipstad, S.; Hak, S.; Baghirov, H.; Sulheim, E.; Mørch, Y.; Lélu, S.; Von Haartman, E.; Bäck, M.; Nilsson, K.P.R.; Klymchenko, A.S.; et al. Labeling nanoparticles: Dye leakage and altered cellular uptake. Cytom. Part A 2016, 91, 760–766. [Google Scholar] [CrossRef]
- Shandilya, P.; Sambyal, S.; Sharma, R.; Mandyal, P.; Fang, B. Properties, optimized morphologies, and advanced strategies for photocatalytic applications of WO3 based photocatalysts. J. Hazard. Mater. 2022, 428, 128218. [Google Scholar] [CrossRef]
- Rees, P.; Wills, J.W.; Brown, R.; Barnes, C.M.; Summers, H.D. The origin of heterogeneous nanoparticle uptake by cells. Nat. Commun. 2019, 10, 2341. [Google Scholar] [CrossRef]
- Sukhanova, A.; Bozrova, S.; Sokolov, P.; Berestovoy, M.; Karaulov, A.; Nabiev, I. Dependence of Nanoparticle Toxicity on Their Physical and Chemical Properties. Nanoscale Res. Lett. 2018, 13, 44. [Google Scholar] [CrossRef]
- Forest, V.; Pourchez, J. Preferential binding of positive nanoparticles on cell membranes is due to electrostatic interactions: A too simplistic explanation that does not take into account the nanoparticle protein corona. Mater. Sci. Eng. C 2017, 70, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Foroozandeh, P.; Aziz, A.A. Insight into Cellular Uptake and Intracellular Trafficking of Nanoparticles. Nanoscale Res. Lett. 2018, 13, 339. [Google Scholar] [CrossRef] [PubMed]
- Friedman, A.D.; Claypool, S.E.; Liu, R. The Smart Targeting of Nanoparticles. Curr. Pharm. Des. 2013, 19, 6315–6329. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.; Park, C.; Yi, G.; Lee, D.; Koo, H. Active Targeting Strategies Using Biological Ligands for Nanoparticle Drug Delivery Systems. Cancers 2019, 11, 640. [Google Scholar] [CrossRef]
- Spicer, C.D.; Jumeaux, C.; Gupta, B.; Stevens, M.M. Peptide and protein nanoparticle conjugates: Versatile platforms for biomedical applications. Chem. Soc. Rev. 2018, 47, 3574–3620. [Google Scholar] [CrossRef]
- Kher, G.; Trehan, S.; Misra, A. Antisense Oligonucleotides and RNA Interference. In Challenges in Delivery of Therapeutic Genomics and Proteomics; Elsevier: Amsterdam, The Netherlands, 2011; pp. 325–386. [Google Scholar] [CrossRef]
- Cremers, G.A.O.; Rosier, B.J.H.M.; Brillas, R.R.; Albertazzi, L.; de Greef, T.F.A. Efficient Small-Scale Conjugation of DNA to Primary Antibodies for Multiplexed Cellular Targeting. Bioconjug. Chem. 2019, 30, 2384–2392. [Google Scholar] [CrossRef]
- Gao, J.; Yao, X.; Chen, Y.; Gao, Z.; Zhang, J. Near-Infrared Light-Induced Self-Powered Aptasensing Platform for Aflatoxin B1 Based on Upconversion Nanoparticles-Doped Bi2S3 Nanorods. Anal. Chem. 2020, 93, 677–682. [Google Scholar] [CrossRef]
- Yu, Z.; Eich, C.; Cruz, L.J. Recent Advances in Rare-Earth-Doped Nanoparticles for NIR-II Imaging and Cancer Theranostics. Front. Chem. 2020, 8, 496. [Google Scholar] [CrossRef]
- Chinnathambi, S.; Shirahata, N. Recent advances on fluorescent biomarkers of near-infrared quantum dots for in vitro and in vivo imaging. Sci. Technol. Adv. Mater. 2019, 20, 337–355. [Google Scholar] [CrossRef]
- Arvizo, R.; Bhattacharya, R.; Mukherjee, P. Gold nanoparticles: Opportunities and challenges in nanomedicine. Expert Opin. Drug Deliv. 2010, 7, 753–763. [Google Scholar] [CrossRef]
- Dong, H.; Sun, L.-D.; Yan, C.-H. Lanthanide-Doped Upconversion Nanoparticles for Super-Resolution Microscopy. Front. Chem. 2021, 8, 619377. [Google Scholar] [CrossRef] [PubMed]
- El-Sheikh, S.M.; Barhoum, A.; El-Sherbiny, S.; Morsy, F.; El-Midany, A.A.-H.; Rahier, H. Preparation of superhydrophobic nanocalcite crystals using Box–Behnken design. Arab. J. Chem. 2019, 12, 1479–1486. [Google Scholar] [CrossRef]
- Rehan, M.; Khattab, T.A.; Barohum, A.; Gätjen, L.; Wilken, R. Development of Ag/AgX (X = Cl, I) nanoparticles toward antimicrobial, UV-protected and self-cleanable viscose fibers. Carbohydr. Polym. 2018, 197, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Wahajuddin; Arora, S. Superparamagnetic iron oxide nanoparticles: Magnetic nanoplatforms as drug carriers. Int. J. Nanomed. 2012, 7, 3445–3471. [Google Scholar] [CrossRef]
- Lim, W.Q.; Phua, S.Z.F.; Xu, H.V.; Sreejith, S.; Zhao, Y. Recent advances in multifunctional silica-based hybrid nanocarriers for bioimaging and cancer therapy. Nanoscale 2015, 8, 12510–12519. [Google Scholar] [CrossRef] [PubMed]
- Liang, R.; Wei, M.; Evans, D.G.; Duan, X. Inorganic nanomaterials for bioimaging, targeted drug delivery and therapeutics. Chem. Commun. 2014, 50, 14071–14081. [Google Scholar] [CrossRef] [PubMed]
- Bhunia, S.K.; Saha, A.; Maity, A.; Ray, S.C.; Jana, N.R. Carbon Nanoparticle-based Fluorescent Bioimaging Probes. Sci. Rep. 2013, 3, srep01473. [Google Scholar] [CrossRef] [PubMed]
- Karatutlu, A.; Barhoum, A.; Sapelkin, A. Theories of nanoparticle and nanostructure formation in liquid phase. In Emerging Applications of Nanoparticles and Architecture Nanostructures: Current Prospects and Future Trends; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 597–619. [Google Scholar] [CrossRef]
- Barhoum, A.; García-Betancourt, M.L. Physicochemical characterization of nanomaterials: Size, morphology, optical, magnetic, and electrical properties. In Emerging Applications of Nanoparticles and Architecture Nanostructures: Current Prospects and Future Trends; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 279–304. [Google Scholar] [CrossRef]
- Karatutlu, A.; Barhoum, A.; Sapelkin, A. Liquid-phase synthesis of nanoparticles and nanostructured materials. In Emerging Applications of Nanoparticles and Architecture Nanostructures: Current Prospects and Future Trends; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 1–28. [Google Scholar] [CrossRef]
- Tian, P.; Tang, L.; Teng, K.; Lau, S. Graphene quantum dots from chemistry to applications. Mater. Today Chem. 2018, 10, 221–258. [Google Scholar] [CrossRef]
- Singh, I.; Arora, R.; Dhiman, H.; Pahwa, R. Carbon Quantum Dots: Synthesis, Characterization and Biomedical Applications. Turk. J. Pharm. Sci. 2018, 15, 219–230. [Google Scholar] [CrossRef]
- Jhonsi, M.A. Carbon Quantum Dots for Bioimaging. In State of the Art in Nano-Bioimaging; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Ravichandiran, P.; Subramaniyan, S.A.; Bella, A.P.; Johnson, P.M.; Kim, A.R.; Shim, K.S.; Yoo, D.J. Simple Fluorescence Turn-On Chemosensor for Selective Detection of Ba2+ Ion and Its Live Cell Imaging. Anal. Chem. 2019, 91, 10095–10101. [Google Scholar] [CrossRef]
- Hubbs, A.F.; Sargent, L.M.; Porter, D.W.; Sager, T.M.; Chen, B.T.; Frazer, D.G.; Castranova, V.; Sriram, K.; Nurkiewicz, T.R.; Reynolds, S.H.; et al. Nanotechnology: Toxicologic Pathology. Toxicol. Pathol. 2013, 41, 395–409. [Google Scholar] [CrossRef] [PubMed]
- Maldiney, T.; Richard, C.; Seguin, J.; Wattier, N.; Bessodes, M.; Scherman, D. Effect of Core Diameter, Surface Coating, and PEG Chain Length on the Biodistribution of Persistent Luminescence Nanoparticles in Mice. ACS Nano 2011, 5, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Heeger, A.J. Semiconducting and Metallic Polymers: The Fourth Generation of Polymeric Materials (Nobel Lecture). Angew. Chemie Int. Ed. 2001, 40, 2591–2611. [Google Scholar] [CrossRef]
- Thomas, S.W.; Joly, G.D.; Swager, T.M. Chemical Sensors Based on Amplifying Fluorescent Conjugated Polymers. Chem. Rev. 2007, 107, 1339–1386. [Google Scholar] [CrossRef]
- Feng, X.; Liu, L.; Wang, S.; Zhu, D. Water-soluble fluorescent conjugated polymers and their interactions with biomacromolecules for sensitive biosensors. Chem. Soc. Rev. 2010, 39, 2411–2419. [Google Scholar] [CrossRef]
- Khanbeigi, R.A.; Abelha, T.F.; Woods, A.; Rastoin, O.; Harvey, R.D.; Jones, M.-C.; Forbes, B.; Green, M.A.; Collins, H.; Dailey, L.A. Surface Chemistry of Photoluminescent F8BT Conjugated Polymer Nanoparticles Determines Protein Corona Formation and Internalization by Phagocytic Cells. Biomacromolecules 2015, 16, 733–742. [Google Scholar] [CrossRef]
- Tuncel, D.; Demir, H.V. Conjugated polymer nanoparticles. Nanoscale 2010, 2, 484–494. [Google Scholar] [CrossRef]
- Du, T.; Zhao, C.; Lai, L.; Luo, S.; Selke, M.; Rehman, F.U.; Li, X.; Sun, Y.; Jiang, H.; Wang, X. Rapid and multimodal in vivo bioimaging of cancer cells through in situ biosynthesis of Zn&Fe nanoclusters. Nano Res. 2017, 10, 2626–2632. [Google Scholar] [CrossRef]
- Feng, L.; Liu, L.; Lv, F.; Bazan, G.C.; Wang, S. Preparation and Biofunctionalization of Multicolor Conjugated Polymer Nanoparticles for Imaging and Detection of Tumor Cells. Adv. Mater. 2014, 26, 3926–3930. [Google Scholar] [CrossRef]
- Ravichandiran, P.; Prabakaran, D.; Maroli, N.; Kim, A.R.; Park, B.-H.; Han, M.-K.; Ramesh, T.; Ponpandian, S.; Yoo, D.J. Mitochondria-targeted acridine-based dual-channel fluorescence chemosensor for detection of Sn4+ and Cr2O72-ions in water and its application in discriminative detection of cancer cells. J. Hazard. Mater. 2021, 419, 126409. [Google Scholar] [CrossRef]
- Rhim, W.-K.; Kim, M.; Hartman, K.L.; Kang, K.W.; Nam, J.-M. Radionuclide-labeled nanostructures for In Vivo imaging of cancer. Nano Converg. 2015, 2, 10. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, C.; Jiang, T.; Ran, G.; Song, Q. Cadmium induced aggregation of orange–red emissive carbon dots with enhanced fluorescence for intracellular imaging. J. Hazard. Mater. 2021, 427, 128092. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Yu, X.; Peng, S.; Luo, Y.; Li, J.; Lu, L. Construction of nanomaterials as contrast agents or probes for glioma imaging. J. Nanobiotechnol. 2021, 19, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Pratiwi, F.; Kuo, C.W.; Chen, B.-C.; Chen, P. Recent advances in the use of fluorescent nanoparticles for bioimaging. Nanomedicine 2019, 14, 1759–1769. [Google Scholar] [CrossRef]
- Chen, S.; Wang, H.; Hong, Y.; Tang, B.Z. Fabrication of fluorescent nanoparticles based on AIE luminogens (AIE dots) and their applications in bioimaging. Mater. Horizons 2016, 3, 283–293. [Google Scholar] [CrossRef]
- Caponetti, V.; Trzcinski, J.W.; Cantelli, A.; Tavano, R.; Papini, E.; Mancin, F.; Montalti, M. Self-Assembled Biocompatible Fluorescent Nanoparticles for Bioimaging. Front. Chem. 2019, 7, 168. [Google Scholar] [CrossRef]
- Lin, J.; Chen, X.; Huang, P. Graphene-based nanomaterials for bioimaging. Adv. Drug Deliv. Rev. 2016, 105, 242–254. [Google Scholar] [CrossRef]
- Yadav, V.; Roy, S.; Singh, P.; Khan, Z.; Jaiswal, A. 2D MoS2-Based Nanomaterials for Therapeutic, Bioimaging, and Biosensing Applications. Small 2018, 15, e1803706. [Google Scholar] [CrossRef]
- Zhao, W.; Li, A.; Zhang, A.; Zheng, Y.; Liu, J. Recent Advances in Functional-Polymer-Decorated Transition-Metal Nanomaterials for Bioimaging and Cancer Therapy. ChemMedChem 2018, 13, 2134–2149. [Google Scholar] [CrossRef]
- Yi, Z.; Luo, Z.; Qin, X.; Chen, Q.; Liu, X. Lanthanide-Activated Nanoparticles: A Toolbox for Bioimaging, Therapeutics, and Neuromodulation. Accounts Chem. Res. 2020, 53, 2692–2704. [Google Scholar] [CrossRef]
- Xu, Y.; Li, P.; Cheng, D.; Wu, C.; Lu, Q.; Yang, W.; Zhu, X.; Yin, P.; Liu, M.; Li, H.; et al. Group IV nanodots: Synthesis, surface engineering and application in bioimaging and biotherapy. J. Mater. Chem. B 2020, 8, 10290–10308. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, Y.; Bidram, E.; Zarrabi, A.; Amini, A.; Cheng, C. Graphene oxide and its derivatives as promising in-vitro bio-imaging platforms. Sci. Rep. 2020, 10, 18052. [Google Scholar] [CrossRef] [PubMed]
- Das, A.K.; Gavel, P.K. Low molecular weight self-assembling peptide-based materials for cell culture, antimicrobial, anti-inflammatory, wound healing, anticancer, drug delivery, bioimaging and 3D bioprinting applications. Soft Matter 2020, 16, 10065–10095. [Google Scholar] [CrossRef] [PubMed]
- Tan, P.; Li, H.; Wang, J.; Gopinath, S.C. Silver nanoparticle in biosensor and bioimaging: Clinical perspectives. Biotechnol. Appl. Biochem. 2021, 68, 1236–1242. [Google Scholar] [CrossRef] [PubMed]
- Bao, C.; Beziere, N.; del Pino, P.; Pelaz, B.; Estrada, G.; Tian, F.; Ntziachristos, V.; de la Fuente, J.M.; Cui, D. Gold Nanoprisms as Optoacoustic Signal Nanoamplifiers for In Vivo Bioimaging of Gastrointestinal Cancers. Small 2012, 9, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.-T.; Tang, K.-C.; Chung, M.-F.; Cheng, S.-H.; Huang, C.-M.; Chu, C.-H.; Chou, P.-T.; Souris, J.S.; Chen, C.-T.; Mou, C.-Y.; et al. Enhanced Plasmonic Resonance Energy Transfer in Mesoporous Silica-Encased Gold Nanorod for Two-Photon-Activated Photodynamic Therapy. Theranostics 2014, 4, 798–807. [Google Scholar] [CrossRef]
- Yadav, A.; Rao, C.; Verma, N.C.; Mishra, P.M.; Nandi, C.K. Magnetofluorescent Nanoprobe for Multimodal and Multicolor Bioimaging. Mol. Imaging 2020, 19, 1–8. [Google Scholar] [CrossRef]
- Klymchenko, A.S.; Liu, F.; Collot, M.; Anton, N. Dye-Loaded Nanoemulsions: Biomimetic Fluorescent Nanocarriers for Bioimaging and Nanomedicine. Adv. Health Mater. 2020, 10, e2001289. [Google Scholar] [CrossRef]
- Xu, Y.-M.; Tan, H.W.; Zheng, W.; Liang, Z.-L.; Yu, F.-Y.; Wu, D.-D.; Yao, Y.; Zhong, Q.-H.; Yan, R.; Lau, A.T.Y. Cadmium telluride quantum dot-exposed human bronchial epithelial cells: A further study of the cellular response by proteomics. Toxicol. Res. 2019, 8, 994–1001. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; del Pilar Rodriguez-Torres, M.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef]
- Yao, Y.; Zhou, Y.; Liu, L.; Xu, Y.; Chen, Q.; Wang, Y.; Wu, S.; Deng, Y.; Zhang, J.; Shao, A. Nanoparticle-Based Drug Delivery in Cancer Therapy and Its Role in Overcoming Drug Resistance. Front. Mol. Biosci. 2020, 7, 193. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Shin, J.; Wu, J.; Omstead, D.T.; Kiziltepe, T.; Littlepage, L.E.; Bilgicer, B. Engineering peptide-targeted liposomal nanoparticles optimized for improved selectivity for HER2-positive breast cancer cells to achieve enhanced in vivo efficacy. J. Control. Release 2020, 322, 530–541. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Zhang, X.; Shen, H.; He, Q.; Wu, Z.; Liao, W.; Yuan, M. Application of the Nano-Drug Delivery System in Treatment of Cardiovascular Diseases. Front. Bioeng. Biotechnol. 2020, 7, 489. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Gigliobianco, M.R.; Censi, R.; Di Martino, P. Polymeric Nanocapsules as Nanotechnological Alternative for Drug Delivery System: Current Status, Challenges and Opportunities. Nanomaterials 2020, 10, 847. [Google Scholar] [CrossRef]
- du Toit, L.; Pillay, V.; Choonara, Y.; Pillay, S.; Harilall, S.-L. Patenting of Nanopharmaceuticals in Drug Delivery: No Small Issue. Recent Patents Drug Deliv. Formul. 2007, 1, 131–142. [Google Scholar] [CrossRef]
- Narducci, D. An Introduction to Nanotechnologies: What’s in it for Us? Veter-Res. Commun. 2007, 31, 131–137. [Google Scholar] [CrossRef]
- Navya, P.; Kaphle, A.; Srinivas, S.; Bhargava, S.K.; Rotello, V.M.; Daima, H.K. Current trends and challenges in cancer management and therapy using designer nanomaterials. Nano Converg. 2019, 6, 1–30. [Google Scholar] [CrossRef]
- Wen, H.; Jung, H.; Li, X. Drug Delivery Approaches in Addressing Clinical Pharmacology-Related Issues: Opportunities and Challenges. AAPS J. 2015, 17, 1327–1340. [Google Scholar] [CrossRef]
- Debnath, S.K.; Srivastava, R. Drug Delivery with Carbon-Based Nanomaterials as Versatile Nanocarriers: Progress and Prospects. Front. Nanotechnol. 2021, 3, 15. [Google Scholar] [CrossRef]
- Marcelo, G.; Kaplan, E.; Tarazona, M.P.; Mendicuti, F. Interaction of gold nanoparticles with Doxorubicin mediated by supramolecular chemistry. Colloids Surf. B Biointerfaces 2015, 128, 237–244. [Google Scholar] [CrossRef]
- Zhang, Y.; Walker, J.B.; Minic, Z.; Liu, F.; Goshgarian, H.; Mao, G. Transporter protein and drug-conjugated gold nanoparticles capable of bypassing the blood-brain barrier. Sci. Rep. 2016, 6, 25794. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, Z.; Raza, A.; Ghafoor, S.; Naeem, A.; Naz, S.S.; Riaz, S.; Ahmed, W.; Rana, N.F. PEG capped methotrexate silver nanoparticles for efficient anticancer activity and biocompatibility. Eur. J. Pharm. Sci. 2016, 91, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Prabha, G.; Raj, V. Sodium alginate–polyvinyl alcohol–bovin serum albumin coated Fe3O4 nanoparticles as anticancer drug delivery vehicle: Doxorubicin loading and in vitro release study and cytotoxicity to HepG2 and L02 cells. Mater. Sci. Eng. C 2017, 79, 410–422. [Google Scholar] [CrossRef] [PubMed]
- Prabha, G.; Raj, V. Formation and characterization of β-cyclodextrin (β-CD)-polyethyleneglycol (PEG)-polyethyleneimine (PEI) coated Fe3O4 nanoparticles for loading and releasing 5-Fluorouracil drug. Biomed. Pharmacother. 2016, 80, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Matranga, C.; Tan, S.; Alba, N.; Cui, X.T. Carbon nanotube nanoreservior for controlled release of anti-inflammatory dexamethasone. Biomaterials 2011, 32, 6316–6323. [Google Scholar] [CrossRef] [PubMed]
- Bhirde, A.A.; Patel, S.; Sousa, A.A.; Patel, V.; Molinolo, A.A.; Ji, Y.; Leapman, R.D.; Gutkind, J.S.; Rusling, J.F. Distribution and clearance of PEG-single-walled carbon nanotube cancer drug delivery vehicles in mice. Nanomedicine 2010, 5, 1535–1546. [Google Scholar] [CrossRef] [PubMed]
- Ruzycka, M.; Kowalik, P.; Kowalczyk, A.; Bujak, P.; Nowicka, A.; Wojewódzka, M.; Kruszewski, M.; Grudzinski, I. Quantum dots as targeted doxorubicin drug delivery nanosystems in human lung cancer cells. Cancer Nanotechnol. 2021, 12, 8. [Google Scholar] [CrossRef]
- Roozbahani, M.; Kharaziha, M.; Emadi, R. pH sensitive dexamethasone encapsulated laponite nanoplatelets: Release mechanism and cytotoxicity. Int. J. Pharm. 2017, 518, 312–319. [Google Scholar] [CrossRef]
- Gurdag, S.; Khandare, J.; Stapels, S.; Matherly, L.H.; Kannan, R.M. Activity of Dendrimer−Methotrexate Conjugates on Methotrexate-Sensitive and -Resistant Cell Lines. Bioconjug. Chem. 2006, 17, 275–283. [Google Scholar] [CrossRef]
- Cirstoiu-Hapca, A.; Buchegger, F.; Lange, N.; Bossy, L.; Gurny, R.; Delie, F. Benefit of anti-HER2-coated paclitaxel-loaded immuno-nanoparticles in the treatment of disseminated ovarian cancer: Therapeutic efficacy and biodistribution in mice. J. Control. Release 2010, 144, 324–331. [Google Scholar] [CrossRef]
- Wilson, B.; Samanta, M.K.; Santhi, K.; Kumar, K.S.; Ramasamy, M.; Suresh, B. Chitosan nanoparticles as a new delivery system for the anti-Alzheimer drug tacrine. Nanomed. NanotechnoL. Biol. Med. 2010, 6, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.; Franch, A.; Castell, M.; Perez-Cano, F.J.; Bräuer, R.; Pohlers, D.; Gajda, M.; Siskos, A.P.; Katsila, T.; Tamvakopoulos, C.; et al. Liposomal encapsulation enhances and prolongs the anti-inflammatory effects of water-soluble dexamethasone phosphate in experimental adjuvant arthritis. Arthritis Res. Ther. 2010, 12, R147. [Google Scholar] [CrossRef] [PubMed]
- Zalba, S.; Contreras-Sandoval, A.; Haeri, A.; Hagen, T.L.T.; Navarro-Blasco, I.; Koning, G.; Garrido, M.J. Cetuximab-oxaliplatin-liposomes for epidermal growth factor receptor targeted chemotherapy of colorectal cancer. J. Control. Release 2015, 210, 26–38. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wu, Y.; Kuang, G.; Liu, S.; Zhou, D.; Chen, X.; **g, X.; Huang, Y. Pt(iv) prodrug-backboned micelle and DCA loaded nanofibers for enhanced local cancer treatment. J. Mater. Chem. B 2017, 5, 2115–2125. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhou, G.; Liu, D.; ** agents. Rom. J. Mater. 2021, 51, 342–352. [Google Scholar]
- Fatima, S.; Ali, K.; Ahmed, B.; Al Kheraif, A.A.; Syed, A.; Elgorban, A.M.; Musarrat, J.; Lee, J. Titanium Dioxide Nanoparticles Induce Inhibitory Effects against Planktonic Cells and Biofilms of Human Oral Cavity Isolates of Rothia mucilaginosa, Georgenia sp. and Staphylococcus saprophyticus. Pharmaceutics 2021, 13, 1564. [Google Scholar] [CrossRef]
- Al-Shabib, N.A.; Husain, F.M.; Qais, F.A.; Ahmad, N.; Khan, A.; Alyousef, A.A.; Arshad, M.; Noor, S.; Khan, J.M.; Alam, P.; et al. Phyto-Mediated Synthesis of Porous Titanium Dioxide Nanoparticles From Withania somnifera Root Extract: Broad-Spectrum Attenuation of Biofilm and Cytotoxic Properties Against HepG2 Cell Lines. Front. Microbiol. 2020, 11, 1680. [Google Scholar] [CrossRef]
- Ilyas, M.; Waris, A.; Khan, A.U.; Zamel, D.; Yar, L.; Baset, A.; Muhaymin, A.; Khan, S.; Ali, A.; Ahmad, A. Biological synthesis of titanium dioxide nanoparticles from plants and microorganisms and their potential biomedical applications. Inorg. Chem. Commun. 2021, 133, 108968. [Google Scholar] [CrossRef]
- Balaraman, R.P.; Mendel, J.; Flores, L.; Choudhary, M. Nanoparticle Biosynthesis and Interaction with the Microbial Cell, Antimicrobial and Antibiofilm Effects, and Environmental Impact. In Nanomaterial Biointeractions at the Cellular, Organismal and System Levels; Springer: Cham, Switzerland, 2021; pp. 371–405. [Google Scholar] [CrossRef]
- Jardón-Maximino, N.; Cadenas-Pliego, G.; Ávila-Orta, C.; Comparán-Padilla, V.; Lugo-Uribe, L.; Pérez-Alvarez, M.; Tavizón, S.; Santillán, G. Antimicrobial Property of Polypropylene Composites and Functionalized Copper Nanoparticles. Polymers 2021, 13, 1694. [Google Scholar] [CrossRef]
- Gharpure, S.; Akash, A.; Ankamwar, B. A Review on Antimicrobial Properties of Metal Nanoparticles. J. Nanosci. Nanotechnol. 2020, 20, 3303–3339. [Google Scholar] [CrossRef]
- El-Sheikh, S.; El-Sherbiny, S.; Barhoum, A.; Deng, Y. Effects of cationic surfactant during the precipitation of calcium carbonate nano-particles on their size, morphology, and other characteristics. Colloids Surf. A Physicochem. Eng. Asp. 2013, 422, 44–49. [Google Scholar] [CrossRef]
- Sharma, U.; Badyal, P.N.; Gupta, S. Polymeric nanoparticles drug delivery to brain: A review. Int. J. Pharmacol. 2015, 2, 60–69. [Google Scholar]
- Jun, A.S.; Larkin, D.F.P. Prospects for gene therapy in corneal disease. Eye 2003, 17, 906–911. [Google Scholar] [CrossRef] [PubMed]
- Dzau, V.J.; Mann, M.J.; Morishita, R.; Kaneda, Y. Fusigenic viral liposome for gene therapy in cardiovascular diseases. Proc. Natl. Acad. Sci. USA 1996, 93, 11421–11425. [Google Scholar] [CrossRef] [PubMed]
- Caplen, N.; Gao, X.; Hayes, P.; Elaswarapu, R.; Fisher, G.; Kinrade, E.; Chakera, A.; Schorr, J.; Hughes, B.; Dorin, J.R. Gene therapy for cystic fibrosis in humans by liposome-mediated DNA transfer: The production of resources and the regulatory process. Gene Ther. 1994, 1, 139–147. [Google Scholar] [PubMed]
- Balazs, D.A.; Godbey, W. Liposomes for Use in Gene Delivery. J. Drug Deliv. 2010, 2011, 326497. [Google Scholar] [CrossRef] [PubMed]
- Ito, I.; Ji, L.; Tanaka, F.; Saito, Y.; Gopalan, B.; Branch, C.D.; Xu, K.; Atkinson, E.N.; Bekele, B.N.; Stephens, L.C.; et al. Liposomal vector mediated delivery of the 3p FUS1 gene demonstrates potent antitumor activity against human lung cancer in vivo. Cancer Gene Ther. 2004, 11, 733–739. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.N.; Prateeksha; Gupta, V.K.; Chen, J.; Atanasov, A. Organic Nanoparticle-Based Combinatory Approaches for Gene Therapy. Trends Biotechnol. 2017, 35, 1121–1124. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Guo, Z.; Tian, H.; Chen, X. Production and clinical development of nanoparticles for gene delivery. Mol. Ther. Methods Clin. Dev. 2016, 3, 16023. [Google Scholar] [CrossRef]
- Nagamune, T. Biomolecular engineering for nanobio/bionanotechnology. Nano Converg. 2017, 4, 1–56. [Google Scholar] [CrossRef]
- Prabu, S.L.; Suriyaprakash, T.N.K.; Thirumurugan, R. Medicated Nanoparticle for Gene Delivery. In Advanced Technology for Delivering Therapeutics; IntechOpen: London, UK, 2017; Available online: https://www.intechopen.com/chapters/52818 (accessed on 11 May 2017). [CrossRef]
- Jat, S.K.; Bhattacharya, J.; Sharma, M.K. Nanomaterial based gene delivery: A promising method for plant genome engineering. J. Mater. Chem. B 2020, 8, 4165–4175. [Google Scholar] [CrossRef]
- Abdel-Haleem, F.M.; Gamal, E.; Rizk, M.S.; El Nashar, R.M.; Anis, B.; Elnabawy, H.M.; Khalil, A.S.; Barhoum, A. t-Butyl calixarene/Fe2O3@MWCNTs composite-based potentiometric sensor for determination of ivabradine hydrochloride in pharmaceutical formulations. Mater. Sci. Eng. C 2020, 116, 111110. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Haleem, F.M.; Saad, M.; Barhoum, A.; Bechelany, M.; Rizk, M.S. PVC membrane, coated-wire, and carbon-paste ion-selective electrodes for potentiometric determination of galantamine hydrobromide in physiological fluids. Mater. Sci. Eng. C 2018, 89, 140–148. [Google Scholar] [CrossRef] [PubMed]
- El Nashar, R.M.; Ghani, N.T.A.; El Gohary, N.A.; Barhoum, A.; Madbouly, A. Molecularly imprinted polymers based biomimetic sensors for mosapride citrate detection in biological fluids. Mater. Sci. Eng. C 2017, 76, 123–129. [Google Scholar] [CrossRef] [PubMed]
- El-Beshlawy, M.M.; Abdel-Haleem, F.M.; Barhoum, A. Molecularly Imprinted Potentiometric Sensor for Nanomolar Determination of Pioglitazone Hydrochloride in Pharmaceutical Formulations. Electroanalysis 2021, 33, 1244–1254. [Google Scholar] [CrossRef]
- Naresh, V.; Lee, N. A Review on Biosensors and Recent Development of Nanostructured Materials-Enabled Biosensors. Sensors 2021, 21, 1109. [Google Scholar] [CrossRef]
- Abdel-Karim, R.; Reda, Y.; Abdel-Fattah, A. Review—Nanostructured Materials-Based Nanosensors. J. Electrochem. Soc. 2020, 167, 037554. [Google Scholar] [CrossRef]
- Tang, C.K.; Vaze, A.; Shen, M.; Rusling, J.F. High-Throughput Electrochemical Microfluidic Immunoarray for Multiplexed Detection of Cancer Biomarker Proteins. ACS Sens. 2016, 1, 1036–1043. [Google Scholar] [CrossRef]
- Wang, Z.; Hu, T.; Liang, R.; Wei, M. Application of Zero-Dimensional Nanomaterials in Biosensing. Front. Chem. 2020, 8, 320. [Google Scholar] [CrossRef]
- Cotta, M.A. Quantum Dots and Their Applications: What Lies Ahead? ACS Appl. Nano Mater. 2020, 3, 4920–4924. [Google Scholar] [CrossRef]
- Abraham, J.; Arunima, R.; Nimitha, K.; George, S.C.; Thomas, S. One-dimensional (1D) nanomaterials: Nanorods and nanowires. In Nanoscale Processing; Elsevier: Amsterdam, The Netherlands, 2021; pp. 71–101. [Google Scholar] [CrossRef]
- Erol, O.; Uyan, I.; Hatip, M.; Yilmaz, C.; Tekinay, A.B.; Guler, M.O. Recent advances in bioactive 1D and 2D carbon nanomaterials for biomedical applications. Nanomed. Nanotechnol. Biol. Med. 2017, 14, 2433–2454. [Google Scholar] [CrossRef]
- Dvir, T.; Timko, B.P.; Kohane, D.S.; Langer, R. Nanotechnological strategies for engineering complex tissues. Nat. Nanotechnol. 2011, 6, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Votruba, A.R.; Farokhzad, O.; Langer, R. Nanotechnology in Drug Delivery and Tissue Engineering: From Discovery to Applications. Nano Lett. 2010, 10, 3223–3230. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Barhoum, A.; ** zebrafish embryos. Nanotoxicology 2010, 5, 43–54. [Google Scholar] [CrossRef]
- Tao, C. Antimicrobial activity and toxicity of gold nanoparticles: Research progress, challenges and prospects. Lett. Appl. Microbiol. 2018, 67, 537–543. [Google Scholar] [CrossRef]
- Boisselier, E.; Astruc, D. Gold nanoparticles in nanomedicine: Preparations, imaging, diagnostics, therapies and toxicity. Chem. Soc. Rev. 2009, 38, 1759–1782. [Google Scholar] [CrossRef]
- Ahamed, M.; Siddiqui, M.; Akhtar, M.; Ahmad, I.; Pant, A.B.; Alhadlaq, H. Genotoxic potential of copper oxide nanoparticles in human lung epithelial cells. Biochem. Biophys. Res. Commun. 2010, 396, 578–583. [Google Scholar] [CrossRef]
- Fahmy, B.; Cormier, S.A. Copper oxide nanoparticles induce oxidative stress and cytotoxicity in airway epithelial cells. Toxicol. Vitro 2009, 23, 1365–1371. [Google Scholar] [CrossRef]
- Karlsson, H.L.; Gustafsson, J.; Cronholm, P.; Möller, L. Size-dependent toxicity of metal oxide particles—A comparison between nano- and micrometer size. Toxicol. Lett. 2009, 188, 112–118. [Google Scholar] [CrossRef]
- Lei, R.; Wu, C.; Yang, B.; Ma, H.; Shi, C.; Wang, Q.; Wang, Q.; Yuan, Y.; Liao, M. Integrated metabolomic analysis of the nano-sized copper particle-induced hepatotoxicity and nephrotoxicity in rats: A rapid in vivo screening method for nanotoxicity. Toxicol. Appl. Pharmacol. 2008, 232, 292–301. [Google Scholar] [CrossRef] [PubMed]
- de Lima, R.; Seabra, A.B.; Durán, N. Silver nanoparticles: A brief review of cytotoxicity and genotoxicity of chemically and biogenically synthesized nanoparticles. J. Appl. Toxicol. 2012, 32, 867–879. [Google Scholar] [CrossRef] [PubMed]
- Hadrup, N.; Sharma, A.K.; Loeschner, K.; Jacobsen, N.R. Pulmonary toxicity of silver vapours, nanoparticles and fine dusts: A review. Regul. Toxicol. Pharmacol. 2020, 115, 104690. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, C.A.; Seckler, M.; Ingle, A.P.; Gupta, I.; Galdiero, S.; Galdiero, M.; Gade, A.; Rai, M. Silver Nanoparticles: Therapeutical Uses, Toxicity, and Safety Issues. J. Pharm. Sci. 2014, 103, 1931–1944. [Google Scholar] [CrossRef]
- Cao, Y.; Gong, Y.; Liao, W.; Luo, Y.; Wu, C.; Wang, M.; Yang, Q. A review of cardiovascular toxicity of TiO2, ZnO and Ag nanoparticles (NPs). BioMetals 2018, 31, 457–476. [Google Scholar] [CrossRef]
- Kumar, A.; Pandey, A.K.; Singh, S.S.; Shanker, R.; Dhawan, A. Engineered ZnO and TiO2 nanoparticles induce oxidative stress and DNA damage leading to reduced viability of Escherichia coli. Free Radic. Biol. Med. 2011, 51, 1872–1881. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, V.D.; Prasad, S.V.; Banerjee, A.; Gopinath, M.; Murugesan, R.; Marotta, F.; Sun, X.-F.; Pathak, S. Health hazards of nanoparticles: Understanding the toxicity mechanism of nanosized ZnO in cosmetic products. Drug Chem. Toxicol. 2018, 42, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Singh, S. Zinc oxide nanoparticles impacts: Cytotoxicity, genotoxicity, developmental toxicity, and neurotoxicity. Toxicol. Mech. Methods 2019, 29, 300–311. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Yathindranath, V.; Worden, M.; Thliveris, J.A.; Chu, S.; Parkinson, F.E.; Hegmann, T.; Miller, D.W. Characterization of cellular uptake and toxicity of aminosilane-coated iron oxide nanoparticles with different charges in central nervous system-relevant cell culture models. Int. J. Nanomed. 2013, 8, 961–970. [Google Scholar] [CrossRef]
- Sohaebuddin, S.K.; Thevenot, P.T.; Baker, D.; Eaton, J.W.; Tang, L. Nanomaterial cytotoxicity is composition, size, and cell type dependent. Part. Fibre Toxicol. 2010, 7, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Raghunathan, V.K.; Devey, M.; Hawkins, S.; Hails, L.; Davis, S.A.; Mann, S.; Chang, I.T.; Ingham, E.; Malhas, A.; Vaux, D.J.; et al. Influence of particle size and reactive oxygen species on cobalt chrome nanoparticle-mediated genotoxicity. Biomaterials 2013, 34, 3559–3570. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, Y.; Horie, M.; Kobayashi, N.; Shinohara, N.; Shimada, M. Inhalation Toxicity Assessment of Carbon-Based Nanoparticles. Acc. Chem. Res. 2012, 46, 770–781. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Petibone, D.; Xu, Y.; Mahmood, M.; Karmakar, A.; Casciano, D.; Ali, S.; Biris, A.S. Toxicity and efficacy of carbon nanotubes and graphene: The utility of carbon-based nanoparticles in nanomedicine. Drug Metab. Rev. 2014, 46, 232–246. [Google Scholar] [CrossRef] [PubMed]
- Fröhlich, E. Role of omics techniques in the toxicity testing of nanoparticles. J. Nanobiotechnol. 2017, 15, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Hamimed, S.; Abdeljelil, N.; Landoulsi, A.; Chatti, A.; Aljabali, A.A.A.; Barhoum, A. Bacterial Cellulose Nanofibers. In Handbook of Nanocelluloses; Barhoum, A., Ed.; Springer: Cham, Switzerland, 2022. [Google Scholar] [CrossRef]
- Moodley, K.G.; Arumugam, V.; Barhoum, A. Nanocellulose-Based Materials for Wastewater Treatment. In Handbook of Nanocelluloses; Barhoum, A., Ed.; Springer: Cham, Switzerland, 2021. [Google Scholar] [CrossRef]













| Nanomaterial | Functionalization | Cell Lines | Refs |
|---|---|---|---|
| Graphene-based nanosheets | Surface functionalization by bio-compatible targeting ligands and coatings | MDA-MB-468 (MCF-7) | [70] |
| Molybdenum disulfide nanosheets | Chitosan; PLGA, PEG functionalization | Breast cancer cells (MDA-MB-468), HeLa uterine cancer cells, human lung cancer cells | [71] |
| Transition metal nanoparticles decorated with polymers | Polymer functionalization | Mice bearing 4T1 breast cancer cell xenografts | [72] |
| Lanthanide-activated nanoparticles | Do** with lanthanide | Cancer cells xenografted in mice | [73] |
| Group IV quantum dots | Surface functionalization | Various cancer cell types | [74] |
| Graphene oxide nanosheets | Surface functionalization | Tumor cells | [75] |
| Peptide-based nanoparticles | Chemical functionalization | Peptide-treated HeLa cells preloaded with Hg2+ | [76] |
| Silver nanoparticles | Aptamer conjugation | Leukemia cells, neural stem cells, kidney tissue, renal carcinoma cells | [77] |
| Gold nanoprisms | Conjugation with polyethylene glycol | Gastrointestinal carcinoma cells (HT 29) | [78] |
| Gold nanorods | Encasing by mesoporous silica | Carcinoma cells | [79] |
| Magnetofluroscentnanoprobe | Surface functionalization | Human Breast Cancer (MCF-7), HeLa cells | [80] |
| Dye-loaded nanoemulsions | Lipids conjugation with polyethylene glycol | Human colon cancer (HCT116), HeLa cells | [81] |
| Cadmium telluride quantum dots | Cap** by shells | Human bronchial epithelial cells | [82] |
| Nanocarrier | Loaded Drug | Therapeutic Action | Ref |
|---|---|---|---|
| Metal-based nanoparticles | |||
| Gold nanoparticles | Doxorubicin | Anticancer effect in HeLa cells | [93] |
| Gold nanoparticles | Theophylline (THP), 1,3-dipropyl-8- cyclopentylxanthine (DPCPX) | Neuron reconstruction in vivo | [94] |
| Silver nanoparticles | Methotrexate-coated PEG | Anticancer effect in MCF-7 cells | [95] |
| Metal oxide-based nanoparticles | |||
| Fe3O4 nanoparticles | Doxorubicin | Anticancer effect in HeGP2 and Lo2 cells. | [96] |
| Fe3O4 nanoparticles | Fluorouracil | Anticancer effect in MCF-7 cells | [97] |
| Carbon-based nanoparticles | |||
| Multilayer carbon nanotubes | Dexamethasone | Anti-inflammatory effect in Highly-Aggressively Proliferating Immortalized cells (HAPI) | [98] |
| Single-layer carbon nanotubes | Cisplatin | Anticancer effect in head and neck squamous carcinoma in vivo and in vitro | [99] |
| Quantum dots | |||
| Ag–In–Zn–S quantum dots modified with 11-mercaptoundecanoic acid, L-cysteine, lipoic acid, and decorated with folic acid | Doxorubicin | Anticancer effect in A549 cells (human alveolar basal epithelial cells) | [100] |
| Nano-clays | |||
| Laponite nanoplates | Anionic dexamethasone | Anti-inflammatory effect in MG-63 osteoblast-like cells | [101] |
| Dendrimers | |||
| Poly-amido-amine dendrimers | Methotrexate | Anticancer effect in methotrexate -sensitive and resistant human acute lymphoblastoid leukemia (CCRF-CEM) and Chinese hamster ovary (CHO) cells | [102] |
| Polymeric nanoparticles | |||
| Poly-lactic acid | Paclitaxel | Anticancer effect in a mouse model of ovarian cancer in vivo. | [103] |
| Chitosan | Tacrine | Therapeutic effect in a rat model of Alzheimer’s disease in vivo (preclinical study) | [104] |
| Liposomes | |||
| Liposomes | Dexamethasone phosphate | Anti-inflammatory effect in a rat model of adjuvant-induced arthritis in vivo. | [105] |
| Liposomes | Cetuximab and oxaliplatin | Anticancer effect in mice xenografted with colon cancer cells in vivo | [106] |
| Nanofibers | |||
| Polyvinyl alcohol | PEG2000-Pt(IV) micelles and dichloroacetate | Anticancer effect in mice xenografted with cervical cancer cells in vivo | [107] |
| Polylactic acid electrospun nanofibers | Doxorubicin | Anticancer effect in mice with secondary hepatic carcinoma in vivo | [108] |
| Nanomaterial | Anticancer Drug | Targeted Cancer Cells | Refs |
|---|---|---|---|
| Silver nanoparticles | Terminaliachebula | Breast cancer cells (MCF-7) | [126] |
| Glycerylmonooleate nanostructures | Doxorubicin hydrochloride | Breast cancer cells (MCF-7, MDA-MB-231) | [127] |
| Poly (3HB-co-4HB) biodegradable nanoparticles | Docetaxel | Breast and prostate cancer cells | [128] |
| Carbon nanodots | Irinotecan | Breast cancer cells (MCF-7, MDA-MB-231) | [129] |
| Polysaccharide nanoparticles | Lapatinib | Breast cancer cells (MCF-7/ADR) | [130] |
| Fe3O4 nanoparticles | Doxorubicin | HepGP2 liver cancer cells and LO2 liver cells | [96] |
| Fe3O4 nanoparticles | Fluorouracil | Tumor cells and in vitro analysis | [97] |
| Porous silicon nanoparticles | Doxorubicin and siRNA | Prostate cancer cells | [131] |
| Thermosensitiveliposomes coated with cetuximab | Doxorubicin | EGFR-expressing breast cancer cells | [132] |
| Iron oxide nanoparticles | Cetuximab | A431 (epidermoid carcinoma) cell lines | [133] |
| Nanomaterial | Antioxidant Agent | Applications | Refs |
|---|---|---|---|
| Conjugates | Superoxide dismutase | Superoxide conversion to hydrogen peroxide | [150] |
| Conjugates | Superoxide dismutase | Enhancing drug delivery to the brain | [151] |
| Conjugates | Catalases | Hydrogen peroxide conversion to water | [152] |
| Nanozymes | Catalases | Hydrogen peroxide conversion to water | [153] |
| GSH-PEGDA oligomer nanoparticle | Glutathione peroxidase | Reduction of lipid hydroperoxides and conversion of hydrogen peroxides to water | [154] |
| Liposomes | Vitamins | ROS scavenging and upregulation of antioxidant molecules | [155] |
| Solid lipid nanoparticles | Carotenoids | Singlet oxygen quenching, formation of provitamin A carotenoids (free radical scavengers) | [156] |
| Liposomes | Lycopene | Singlet oxygen quenching, formation of provitamin A carotenoids (free radical scavengers) | [155] |
| Liposomes | Polyphenol flavonoid catechins | Free-radical scavengers, carcinogenic activity, inhibition of proinflammatory kinases | [157] |
| Quercetin nanosuspensions | Quecetin | Protection against LDL oxidation | [157] |
| Silica nanoparticles | Gallic acid | Rapid H-atom transfer to diphenyl picryl hydrazine | [158] |
| Silica nanoparticles | 3,4-di-tert-butyl-4- hydroxybenzoic acid | Improved thermal and oxidative stability of low-density polyethylene (LDPE) composites | [159] |
| PEG-coated silver nanoparticles | Salvianolic acid | Improved reactive oxygen species (ROS) scavenging and antioxidant activity in living cells | [160] |
| Mesoporous silica nanoparticles | Poly-tannic acid | Good antioxidant activity | [161] |
| Mesoporous silica nanoparticles | Morin | Potent quencher of singlet molecular oxygen (1O2), HO· scavenger | [162] |
| Ceria nanoparticles | Polyethylene glycol (PEG)-dendron phospholipids | Biocompatibility, reduction of cytotoxicity and oxidative stress | [163] |
| PLGA-PEG | Curcumin | Neuroprotection | [164] |
| Function | Mode of Action | Nanomaterial | Target Microorganism | Ref |
|---|---|---|---|---|
| Antibacterial | Interaction with DNA, resulting in DNA replication inhibition ROS production Interaction with sulfur-containing proteins, leading to the inhibition of the activity of several enzymes | Silver nanoparticles (Ag NPs) | Bacillus subtilis | [171] |
| Staphylococcus aureus | [172] | |||
| Methicillin-resistant coagulase-negative | [173] | |||
| staphylococci | [174] | |||
| Titanium oxide nanoparticles (TiO2 NPs) | Escherichia coli 0157:H7 | [175] | ||
| Staphylococcus aureus | [176] | |||
| Pseudomonas fluorescens | [177] | |||
| Copper oxide nanoparticles (CuO NPs) | Bacillus subtilis | [178] | ||
| Listeria monocytogenes | [179] | |||
| Antifungal | Disruption of the cell membrane integrity | Titanium oxide nanoparticles (TiO2 NPs) | Candida spp. | [180] |
| Penicillium expansum | [181] | |||
| Aspergillus niger spp. | [182] | |||
| Penicillium oxalicum | [183] | |||
| Siver nanoparticles (Ag NPs) | Candida spp. | [184] | ||
| Magnesium oxide nanoparticles (MgO NPs) | Saccharomyces cerevisiae | [185] | ||
| Candida albicans | [186] | |||
| Antiviral | Inhibition of virus attachment to the host cell membrane | Gold nanoparticles (Au NPs) | Human immunodeficiency virus | [187] |
| Influenza virus | [188] | |||
| Silver nanoparticles (Ag NPs) | Herpes simplex virus | [189] | ||
| Respiratory syncytial virus | [190] | |||
| Titanium oxide nanoparticles (TiO2 NPs) | Inactivation of bacteriophages | [191] | ||
| Inactivation of Qβ and T4 bacteriophages | [192] | |||
| Antiparasitic | Inhibition of promastigote proliferation and metabolic activity Generation of ROS that may inhibit parasitic infection | Silver nanoparticles (Ag NPs) | Leishmania tropica | [193] |
| Leishmania infantum | [194] | |||
| Entamoeba histolytica | [195] | |||
| Copper oxide nanoparticles (CuO NPs) | Entamoeba histolytica | [196] | ||
| Cryptosporidium parvum | [197] |
| Nanomaterial | Dimentionality | Key Benefits | Ref |
|---|---|---|---|
| Spherical metallic nanoparticles | Zero-dimensional (0D) | Immobilization of bio-receptors Improved analyte loading Strong catalytic characteristics | [226] |
| Spherical quantum dots | Zero-dimensional (0D) | Excellent fluorescence, Charge carrier quantum confinement size-adjusted band energy | [227] |
| Nanorods | One-dimensional (1D) | Excellent plasmonic materials Size-adjustable energy regulation to produce specific field responses | [228] |
| Nanowires (1D) | One-dimensional (1D) | Superior charge conduction Strong sensing characteristics | [228] |
| Carbon nanomaterials (1D and 2D) | One and two-dimensional (1D and 2D) | Superior charge conduction High functionalization potential | [229] |
| Agriculture | Food Processing | Food Packaging | Supplements | References |
|---|---|---|---|---|
| Detection of the specific molecule to estimate the enzyme-substrate interaction | Nanoencapsulation for bioavailability enhancement of nutraceuticals | Detection of foodborne chemicals and pathogens by fluorescent nanoparticles attached to antibodies | Nutrient absorption enhancement by nanosized powders | [274,275,276,277] |
| Delivery of pesticides and fertilizers through nanocapsules | Flavor enhancement using nanoencapsulation | Monitoring of temperature, moisture, and time using nanosensors | Cellulose nanocrystals function as drug carrier | [278,279,280,281] |
| Controlled delivery of growth hormones | Nanoparticles used as viscosifying agents | Ethylene detection by electrochemical nanosensors | Nutraceutical nanoencapsulation for enhancement of absorption and stability | [83,281,282,283] |
| Crop growth and soil condition monitoring using nanosensors | Replacement of meat cholesterol by plant-based steroid containing nanocapsules | Surface coated nanoparticles for antifungal and antimicrobial effect | Coiled nanoparticles (nano-cochleate) for cellular delivery of nutrients | [83,265,284,285] |
| Nanosensors for detection of plant and animal pathogens Vaccine delivery using nanocapsules | Removal of pathogens by selective binding of nanoparticles from food | Heat resistant films with silicate nanoparticles | Improvement of absorption by dispersing vitamin sprays to nanodroplets | [286,287,288,289] |
| Nanoparticle Type | Toxicity Mechanism | Applications | Refs |
|---|---|---|---|
| Aluminum oxide nanoparticles | Genotoxicity, changes in protein expression, oxidative stress, cell viability, mitochondrial function | Polymers, biomaterials, fuel cells, paints, textiles, and coatings | [290,291,292,293] |
| Gold nanoparticles | Non-toxic spherical core, relatively safe; lipid peroxidation, autophagy in lung fibroblasts | Contrast agents and drug carriers | [294,295] |
| Copper oxide nanoparticles | Oxidative damage (stress), cytotoxicity (cell membrane integrity), nephrotoxicity, genotoxicity, hepatotoxicity, and spleen toxicity | Antibacterial, semiconductors, heat transfer fluids, and contraceptive devices | [296,297,298,299] |
| Silver nanoparticles | Oxidative stress, genotoxicity, cell viability decrease, nephrotoxicity, cell membrane integrity, lung toxicity, and cardiovascular toxicity | Wound dressing, prostheses, coating for surgical instruments, and antibacterial agents | [300,301,302,303] |
| Zinc oxide nanoparticles | Mitochondrial dysfunction, genotoxicity, oxidative stress, hepatotoxicity, cell membrane integrity, cell viability, cardiovascular toxicity, inflammation, neurotoxicity, cytotoxicity, and reactive oxygen species production | Sunscreens, gas filters, UV detectors, wave filters, and body care products | [303,304,305,306] |
| Iron oxide nanoparticles | Neurotoxicity, mitochondrial function alterations, genotoxicity, lung toxicity, hepatotoxicity, reactive oxygen species production, cell viability, and endothelial permeability | Diagnostic agents and drug carriers | [307,308,309] |
| Titanium nanoparticles | Reactive oxygen species production, nephrotoxicity, genotoxicity, hepatotoxicity, immune function changes, lung toxicity, spleen toxicity, and cardiovascular toxicity | Coloring and pigment agents | [303,304] |
| Carbon-based nanoparticles and fullerenes | Cell membrane integrity, cell viability, bone toxicity, genotoxicity, hepatotoxicity, nephrotoxicity, spleen toxicity, cardiotoxicity, epigenetic toxicity, skin toxicity, carcinogenesis, neurotoxicity, and immunotoxicity | Drug carriers | [310,311,312,313] |
| Polymeric nanoparticles | Non-toxic, relatively safe, non-inflammatory, non-immunologic, and least toxic | Drug carriers | [314,315] |
| Nickel oxide nanoparticles | Apoptosis and lipid peroxidation increase | Antibacterial, antifungal, and cytotoxic | [316,317,318,319,320] |
| Cerium oxide nanoparticles | Apoptosis, cell membrane damage, p38-NRF2 signaling, and inflammation | Antimicrobial, corrosion protection, polishing, and solar cells | [321,322,323,324] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harish, V.; Tewari, D.; Gaur, M.; Yadav, A.B.; Swaroop, S.; Bechelany, M.; Barhoum, A. Review on Nanoparticles and Nanostructured Materials: Bioimaging, Biosensing, Drug Delivery, Tissue Engineering, Antimicrobial, and Agro-Food Applications. Nanomaterials 2022, 12, 457. https://doi.org/10.3390/nano12030457
Harish V, Tewari D, Gaur M, Yadav AB, Swaroop S, Bechelany M, Barhoum A. Review on Nanoparticles and Nanostructured Materials: Bioimaging, Biosensing, Drug Delivery, Tissue Engineering, Antimicrobial, and Agro-Food Applications. Nanomaterials. 2022; 12(3):457. https://doi.org/10.3390/nano12030457
Chicago/Turabian StyleHarish, Vancha, Devesh Tewari, Manish Gaur, Awadh Bihari Yadav, Shiv Swaroop, Mikhael Bechelany, and Ahmed Barhoum. 2022. "Review on Nanoparticles and Nanostructured Materials: Bioimaging, Biosensing, Drug Delivery, Tissue Engineering, Antimicrobial, and Agro-Food Applications" Nanomaterials 12, no. 3: 457. https://doi.org/10.3390/nano12030457