1. Introduction
In the past few decades, develo** functional drug/gene delivery systems derived from natural resources with good biocompatibility, controllable and highly efficient delivery capability, as well as low economic cost has attracted great attention [
1,
2]. By now, many natural-based hydrophobic blocks (phospholipids, lipoid, steroids,
etc.) [
3] and amine-containing cationic groups [
4] (amino acids, polyamine,
etc.) have been widely employed to construct new biocompatible cationic lipids with the merits of a well-defined and tunable molecular structure [
5]. Yang
et al. [
6] developed 1,12-diaminododecane-polyamine cationic bolaamphiphiles with high gene transfection efficiency. Yu
et al. [
7] synthesized some natural lipid-based cyclic polyamines as efficient gene carriers. In a previous study, we designed and synthesized a series of steroid-based cationic lipids, including cholesterol-based bioreduction-responsive cationic lipids (CHOSS) [
8], “click” synthesized cholesterol and lithocholate-derived cationic lipids [
9], as well as cholesterol-based cationic lipids with versatile amino acid headgroups and chemical linkage bonds [
10]. Furthermore, the relationship between their structures and transfection activity had also been preliminarily investigated. Up to date, although many natural products-derived cationic lipids have been synthesized for gene delivery, their self-assembly properties, intracellular uptake, gene transfection efficacy,
etc., need to be further controlled and optimized [
11]. It is noteworthy for most of the lipid-based gene delivery nano-systems that the intracellular uptake and gene delivery efficiency were still far below their natural virus counterparts. Thereby, optimization of the cationic lipid by covalent and non-covalent approaches is essential for achieving high intracellular transport and efficient gene delivery capability, which needs to be continually investigated.
It had been revealed that the non-covalent binding of functional building blocks by using controlled self-assembly methods could generate new supramolecular aggregates with various structures and functions, which provided possible approaches for optimizing the cationic lipid gene carriers. In recent years, some lipid components, called “helper lipids”, had been employed for non-covalent optimization of the cationic lipids towards highly efficient gene delivery. Up to now, several electrically neutral lipids, including cholesterol [
12], phosphatidylcholine (PC) [
13], dioleoylphosphatidyl ethanolamine (DOPE) [
14], alkylacyl phosphatidylcholines (APC) [
15],
etc., had been utilized as the helper lipids for the preparation of new multi-component cationic lipid formulations as gene delivery carriers. Previous research had revealed that the addition/assembly of helper lipids could bring new functions, such as stabilizing the gene/cationic lipid payload, improving the cellular uptake, facilitating the cell-penetrating ability, and so on [
16]. Noteworthy, the co-assembly of cationic lipids with helper lipids could lead to the formation of nanostructures with a controllable and tunable size, shape and morphology [
17]. In some cases, the co-assembly of helper lipids could facilitate the formation of nanomicelles/nanoparticles [
13,
17,
18], whereas in some other cases, the introduction of helper lipids could lead to forming fluidic, highly ordered, lamellar-phased nanovesicles/liposomes [
19,
20,
21]. Moreover, the assembled nano-scale structures could convert from one to another under certain conditions. Sakurai
et al. [
22] disclosed a pH-induced structure changing from a micelle to a hexagonally-packed cylinder in a co-assembled DA-DOPE (DA:
N-(3,5-dialkylbenzyl) ethane-1,2-diamine) lipid system. It has been known that the lipid component, size, shape, morphology and surface charge of assembled cationic nano-aggregates would greatly affect the related gene transfection efficacy, cellular uptake and intracellular distribution of their gene payload. Therefore, the rational design and assembly of the cationic lipids with helper lipids to obtain a controllable, ordered and functionalized lipid formulation as gene carriers with enhanced intracellular gene transport/transfection performances have been regarded as essential issues and challenges in develo** the lipid-based gene carriers.
On the other hand, the gene substances pDNA and siRNA both possess negatively-charged phosphodiester skeletons, which make them able to interact with positively-charged cationic lipids (or liposomes and lipid nanoparticles) to form gene-loaded, condensed, nano-scale (100–400 nm) complexes for gene delivery [
23]. However, pDNA and siRNA have different molecular weights, scales and topographies. Generally, pDNA are double-stranded, high molecular weight (more than several hundreds of base pairs) biomacromolecules with coiled and supercoiled topostructures, while siRNA are also double-stranded biomacromolecules, but with a much lower molecular weight (normally 21−23 base pairs in length) and with a “rigid-rod” molecular topology [
24]. Their difference in physico-chemical properties may offer them different self-assembly capabilities. Consequently, whether a natural lipid-based gene delivery system is suitable for both pDNA and siRNA transportation is still not clear. Moreover, the comparison of the physico-chemical features, cellular uptake, intracellular transport and distribution of the natural lipid-based nano-systems serving as a pDNA and siRNA dual-functional payload was also scarcely reported in earlier literature [
25].
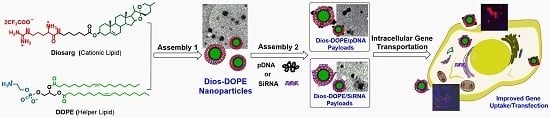
In this work, we synthesized a natural steroid diosgenin-based cationic lipid (Diosarg) with an arginine-bearing headgroup, which was then co-assembled with a membrane-fusion lipid DOPE (in various molar ratio) to prepare cationic Diosarg-DOPE nanoparticles (NPs). Then, the Diosarg- DOPE NPs were employed as templates for the preparation of pDNA and siRNA payloads (
Scheme 1). The physico-chemical properties of the as-prepared cationic lipid/NPs were studied by dynamic light scattering (DLS) instruments and transmission electron microscopy (TEM), and the gene substances’ (pDNA and siRNA) binding affinity, particle size, zeta potential, as well as the morphology of the pDNA/siRNA payloads were studied by DLS, TEM and the agarose-gel retardation assay. The cytotoxicity and gene transfection/transportation properties of the Diosarg lipid and Diosarg-DOPE NPs were measured. In addition, the cellular uptake and intracellular localization of the pDNA/siRNA payloads were also directly observed by flow cytometry (FACS) and fluorescence microscopy, respectively.