The Role of Apoptotic Signaling in Axon Guidance
Abstract
:1. Introduction
Drosophila Netrin-B Is a Neurotrophic Factor That Blocks Cell Death
2. The Apoptotic Machinery and Guidance Receptors
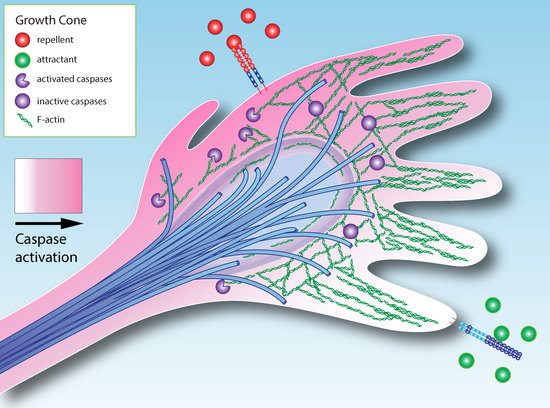
3. How Could Caspase Signaling Operate in Growth Cone Guidance?
4. Extracellular Modulation of Caspase Activity in the Growth Cone
5. Caspase Signaling at the Fly CNS Midline
6. Apoptotic Signaling in Axon Branching
7. Critical Experiments for the Activated Caspase Model
8. Conclusions
- Localized activation of caspases in the growth cone may modulate axon guidance.
- Axon attractants can promote cell survival, while repellents can promote cell death.
- Neurotrophic factor effects on axon guidance could be through caspase signaling.
- Based on an analogy with systems in which caspase signaling has non-apoptotic roles, we propose that the duration and intensity of caspase activation can modulate growth cone activity, while longer and stronger caspase activity can induce death.
- Crossing the CNS midline is associated with lower caspase activity.
- Correct wiring of the nervous system could result from the elimination of incorrectly navigating neurons due to increased activity of the cell death machinery.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lowery, L.A.; Van Vactor, D. The trip of the tip: Understanding the growth cone machinery. Nat. Rev. Mol. Cell Biol. 2009, 10, 332–343. [Google Scholar] [CrossRef] [PubMed]
- Kolodkin, A.L.; Tessier-Lavigne, M. Mechanisms and molecules of neuronal wiring: A primer. Cold Spring Harbor Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef] [PubMed]
- Dent, E.W.; Gupton, S.L.; Gertler, F.B. The growth cone cytoskeleton in axon outgrowth and guidance. Cold Spring Harbor Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef] [PubMed]
- Gomez, T.M.; Letourneau, P.C. Actin dynamics in growth cone motility and navigation. J. Neurochem. 2014, 129, 221–234. [Google Scholar] [CrossRef] [PubMed]
- Chacon, M.R.; Fazzari, P. FAK: Dynamic integration of guidance signals at the growth cone. Cell Adhes. Migr. 2011, 5, 52–55. [Google Scholar] [CrossRef]
- Dudanova, I.; Klein, R. Integration of guidance cues: Parallel signaling and crosstalk. Trends Neurosci. 2013, 36, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Varadarajan, S.G.; Kong, J.H.; Phan, K.D.; Kao, T.J.; Panaitof, S.C.; Cardin, J.; Eltzschig, H.; Kania, A.; Novitch, B.G.; Butler, S.J. Netrin1 Produced by Neural Progenitors, Not Floor Plate Cells, Is Required for Axon Guidance in the Spinal Cord. Neuron 2017, 94, 790–799. [Google Scholar] [CrossRef] [PubMed]
- Dominici, C.; Moreno-Bravo, J.A.; Puiggros, S.R.; Rappeneau, Q.; Rama, N.; Vieugue, P.; Bernet, A.; Mehlen, P.; Chedotal, A. Floor-plate-derived netrin-1 is dispensable for commissural axon guidance. Nature 2017, 545, 350–354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, X.; Wadsworth, W.G. SAX-3 (Robo) and UNC-40 (DCC) regulate a directional bias for axon guidance in response to multiple extracellular cues. PLoS ONE 2014, 9, e110031. [Google Scholar] [CrossRef] [PubMed]
- Stoeckli, E.T. Understanding axon guidance: Are we nearly there yet? Development 2018, 145. [Google Scholar] [CrossRef] [PubMed]
- Goodhill, G.J. Can Molecular Gradients Wire the Brain? Trends Neurosci. 2016, 39, 202–211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, K.L.W.; Correia, J.P.; Kennedy, T.E. Netrins: Versatile extracellular cues with diverse functions. Development 2011, 138, 2153–2169. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.; Sabatelli, L.M.; Seeger, M.A. Guidance cues at the Drosophila CNS midline: Identification and characterization of two Drosophila Netrin/UNC-6 homologs. Neuron 1996, 17, 217–228. [Google Scholar] [CrossRef]
- Mitchell, K.J.; Doyle, J.L.; Serafini, T.; Kennedy, T.E.; Tessier-Lavigne, M.; Goodman, C.S.; Dickson, B.J. Genetic analysis of Netrin genes in Drosophila: Netrins guide CNS commissural axons and peripheral motor axons. Neuron 1996, 17, 203–215. [Google Scholar] [CrossRef]
- Newquist, G.; Drennan, J.M.; Lamanuzzi, M.; Walker, K.; Clemens, J.C.; Kidd, T. Blocking apoptotic signaling rescues axon guidance in Netrin mutants. Cell Rep. 2013, 3, 595–606. [Google Scholar] [CrossRef] [PubMed]
- Campbell, D.S.; Holt, C.E. Apoptotic pathway and MAPKs differentially regulate chemotropic responses of retinal growth cones. Neuron 2003, 37, 939–952. [Google Scholar] [CrossRef]
- Ohsawa, S.; Hamada, S.; Asou, H.; Kuida, K.; Uchiyama, Y.; Yoshida, H.; Miura, M. Caspase-9 activation revealed by semaphorin 7A cleavage is independent of apoptosis in the aged olfactory bulb. J. Neurosci. 2009, 29, 11385–11392. [Google Scholar] [CrossRef] [PubMed]
- Rotschafer, S.E.; Allen-Sharpley, M.R.; Cramer, K.S. Axonal Cleaved Caspase-3 Regulates Axon Targeting and Morphogenesis in the Develo** Auditory Brainstem. Front. Neural Circuits 2016, 10, 84. [Google Scholar] [CrossRef] [PubMed]
- Forsthoefel, D.J.; Liebl, E.C.; Kolodziej, P.A.; Seeger, M.A. The Abelson tyrosine kinase, the Trio GEF and Enabled interact with the Netrin receptor Frazzled in Drosophila. Development 2005, 132, 1983–1994. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Donnell, M.P.; Bashaw, G.J. Distinct functional domains of the Abelson tyrosine kinase control axon guidance responses to Netrin and Slit to regulate the assembly of neural circuits. Development 2013, 140, 2724–2733. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dorsten, J.N.; Varughese, B.E.; Karmo, S.; Seeger, M.A.; VanBerkum, M.F. In the absence of frazzled over-expression of Abelson tyrosine kinase disrupts commissure formation and causes axons to leave the embryonic CNS. PLoS ONE 2010, 5, e9822. [Google Scholar] [CrossRef] [PubMed]
- Muda, M.; Worby, C.A.; Simonson-Leff, N.; Clemens, J.C.; Dixon, J.E. Use of double-stranded RNA-mediated interference to determine the substrates of protein tyrosine kinases and phosphatases. Biochem. J. 2002, 366, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Schmucker, D.; Clemens, J.C.; Shu, H.; Worby, C.A.; ** sympathetic neurons. Development 2016, 143, 1560–1570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.; Park, S.; Kang, Y.S.; Park, S. EphA receptors form a complex with caspase-8 to induce apoptotic cell death. Mol. Cells 2015, 38, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, J.; Liu, J.; Luo, H.; Gou, K.; Cui, S. Prostaglandin F2alpha upregulates Slit/Robo expression in mouse corpus luteum during luteolysis. J. Endocrinol. 2013, 218, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, R.E.; Fegan, K.S.; Ren, X.; Hillier, S.G.; Duncan, W.C. Glucocorticoid regulation of SLIT/ROBO tumour suppressor genes in the ovarian surface epithelium and ovarian cancer cells. PLoS ONE 2011, 6, e27792. [Google Scholar] [CrossRef] [PubMed]
- Winberg, M.L.; Mitchell, K.J.; Goodman, C.S. Genetic analysis of the mechanisms controlling target selection: Complementary and combinatorial functions of netrins, semaphorins, and IgCAMs. Cell 1998, 93, 581–591. [Google Scholar] [CrossRef]
- Jiao, S.; Li, Z. Nonapoptotic function of BAD and BAX in long-term depression of synaptic transmission. Neuron 2011, 70, 758–772. [Google Scholar] [CrossRef] [PubMed]
- Gu, Z.; Serradj, N.; Ueno, M.; Liang, M.; Li, J.; Baccei, M.L.; Martin, J.H.; Yoshida, Y. Skilled Movements Require Non-apoptotic Bax/Bak Pathway-Mediated Corticospinal Circuit Reorganization. Neuron 2017, 94, 626–641. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.W.; Truman, J.W. Cellular mechanisms of dendrite pruning in Drosophila: Insights from in vivo time-lapse of remodeling dendritic arborizing sensory neurons. Development 2005, 132, 3631–3642. [Google Scholar] [CrossRef] [PubMed]
- Rogulja-Ortmann, A.; Luer, K.; Seibert, J.; Rickert, C.; Technau, G.M. Programmed cell death in the embryonic central nervous system of Drosophila melanogaster. Development 2007, 134, 105–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mukherjee, A.; Williams, D.W. More alive than dead: Non-apoptotic roles for caspases in neuronal development, plasticity and disease. Cell Death Differ. 2017, 24, 1411–1421. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Bergmann, A. The cleaved-Caspase-3 antibody is a marker of Caspase-9-like DRONC activity in Drosophila. Cell Death Differ. 2010, 17, 534–539. [Google Scholar] [CrossRef] [PubMed]
- Kidd, T.; Brose, K.; Mitchell, K.J.; Fetter, R.D.; Tessier-Lavigne, M.; Goodman, C.S.; Tear, G. Roundabout controls axon crossing of the CNS midline and defines a novel subfamily of evolutionarily conserved guidance receptors. Cell 1998, 92, 205–215. [Google Scholar] [CrossRef]
- Brankatschk, M.; Dickson, B.J. Netrins guide Drosophila commissural axons at short range. Nat. Neurosci. 2006, 9, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Pennack, J.A.; McQuilton, P.; Forero, M.G.; Mizuguchi, K.; Sutcliffe, B.; Gu, C.J.; Fenton, J.C.; Hidalgo, A. Drosophila neurotrophins reveal a common mechanism for nervous system formation. PLoS Biol. 2008, 6, e284. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Tessier-Lavigne, M. En passant neurotrophic action of an intermediate axonal target in the develo** mammalian CNS. Nature 1999, 401, 765–769. [Google Scholar] [CrossRef] [PubMed]
- Usui, N.; Watanabe, K.; Ono, K.; Tomita, K.; Tamamaki, N.; Ikenaka, K.; Takebayashi, H. Role of motoneuron-derived neurotrophin 3 in survival and axonal projection of sensory neurons during neural circuit formation. Development 2012, 139, 1125–1132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuruvilla, R.; Zweifel, L.S.; Glebova, N.O.; Lonze, B.E.; Valdez, G.; Ye, H.; Ginty, D.D. A neurotrophin signaling cascade coordinates sympathetic neuron development through differential control of TrkA trafficking and retrograde signaling. Cell 2004, 118, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Sutcliffe, B.; Forero, M.G.; Zhu, B.; Robinson, I.M.; Hidalgo, A. Neuron-type specific functions of DNT1, DNT2 and Spz at the Drosophila neuromuscular junction. PLoS ONE 2013, 8, e75902. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, A.; Tugentman, M.; Shilo, B.Z.; Steller, H. Regulation of cell number by MAPK-dependent control of apoptosis: A mechanism for trophic survival signaling. Dev. Cell 2002, 2, 159–170. [Google Scholar] [CrossRef]
- Goldberg, J.L. How does an axon grow? Genes Dev. 2003, 17, 941–958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, K.H.; Brose, K.; Arnott, D.; Kidd, T.; Goodman, C.S.; Henzel, W.; Tessier-Lavigne, M. Biochemical purification of a mammalian slit protein as a positive regulator of sensory axon elongation and branching. Cell 1999, 96, 771–784. [Google Scholar] [CrossRef]
- Nguyen Ba-Charvet, K.T.; Brose, K.; Ma, L.; Wang, K.H.; Marillat, V.; Sotelo, C.; Tessier-Lavigne, M.; Chedotal, A. Diversity and specificity of actions of Slit2 proteolytic fragments in axon guidance. J. Neurosci. 2001, 21, 4281–4289. [Google Scholar] [CrossRef] [PubMed]
- Dascenco, D.; Erfurth, M.-L.; Izadifar, A.; Song, M.; Sachse, S.; Bortnick, R.; Urwyler, O.; Petrovic, M.; Ayaz, D.; He, H.; et al. Slit and Receptor Tyrosine Phosphatase 69D Confer Spatial Specificity to Axon Branching via Dscam1. Cell 2015. [Google Scholar] [CrossRef] [PubMed]
- Piper, M.; Nurcombe, V.; Reid, K.; Bartlett, P.; Little, M. N-terminal Slit2 promotes survival and neurite extension in cultured peripheral neurons. Neuroreport 2002, 13, 2375–2378. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Zheng, B. Axon plasticity in the mammalian central nervous system after injury. Trends Neurosci. 2014, 37, 583–593. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baba, K.; Yoshida, W.; Toriyama, M.; Shimada, T.; Manning, C.F.; Saito, M.; Kohno, K.; Trimmer, J.S.; Watanabe, R.; Inagaki, N. Gradient-reading and mechano-effector machinery for netrin-1-induced axon guidance. eLife 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Kamiyama, D.; Sekine, S.; Barsi-Rhyne, B.; Hu, J.; Chen, B.; Gilbert, L.A.; Ishikawa, H.; Leonetti, M.D.; Marshall, W.F.; Weissman, J.S.; et al. Versatile protein tagging in cells with split fluorescent protein. Nat. Commun. 2016, 7, 11046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamiyama, D.; McGorty, R.; Kamiyama, R.; Kim, M.D.; Chiba, A.; Huang, B. Specification of Dendritogenesis Site in Drosophila aCC Motoneuron by Membrane Enrichment of Pak1 through Dscam1. Dev. Cell 2015, 35, 93–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geden, M.J.; Deshmukh, M. Axon degeneration: Context defines distinct pathways. Curr. Opin. Neurobiol. 2016, 39, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Bardet, P.L.; Kolahgar, G.; Mynett, A.; Miguel-Aliaga, I.; Briscoe, J.; Meier, P.; Vincent, J.P. A fluorescent reporter of caspase activity for live imaging. Proc. Natl. Acad. Sci. USA 2008, 105, 13901–13905. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melzer, J.; Broemer, M. Nerve-racking—Apoptotic and non-apoptotic roles of caspases in the nervous system of Drosophila. Eur. J. Neurosci. 2016, 44, 1683–1690. [Google Scholar] [CrossRef] [PubMed]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kellermeyer, R.; Heydman, L.M.; Mastick, G.S.; Kidd, T. The Role of Apoptotic Signaling in Axon Guidance. J. Dev. Biol. 2018, 6, 24. https://doi.org/10.3390/jdb6040024
Kellermeyer R, Heydman LM, Mastick GS, Kidd T. The Role of Apoptotic Signaling in Axon Guidance. Journal of Developmental Biology. 2018; 6(4):24. https://doi.org/10.3390/jdb6040024
Chicago/Turabian StyleKellermeyer, Riley, Leah M. Heydman, Grant S. Mastick, and Thomas Kidd. 2018. "The Role of Apoptotic Signaling in Axon Guidance" Journal of Developmental Biology 6, no. 4: 24. https://doi.org/10.3390/jdb6040024