Advances in Analysis and Detection of Major Mycotoxins in Foods
Abstract
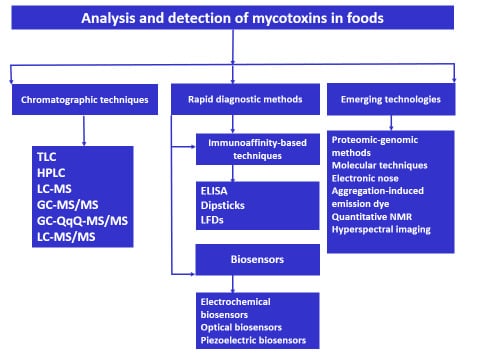
:1. Introduction
2. Extraction Solutions, Extraction Methodologies and Clean-Up Procedures of Mycotoxins
Author Contributions
Funding
Conflicts of Interest
References
- Moretti, A.; Logrieco, A.F.; Susca, A. Mycotoxins: An Underhand Food Problem. In Mycotoxigenic Fungi: Methods and Protocols, Methods in Molecular Biology; Moretti, A., Susca, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2017; Volume 1542, pp. 154–196. [Google Scholar]
- Agriopoulou, S. Enniatins: An emerging food safety issue. EC Nutr. 2016, 3, 1142–1146. [Google Scholar]
- Barac, A. Mycotoxins and Human Disease. In Clinically Relevant Mycoses; Presterl, I., Ed.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 213–225. [Google Scholar]
- Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in Occurrence, Importance, and Mycotoxin Control Strategies: Prevention and Detoxification in Foods. Foods 2020, 9, 137. [Google Scholar] [CrossRef] [PubMed]
- Al-Jaal, B.; Salama, S.; Al-Qasmi, N.; Jaganjac, M. Mycotoxin contamination of food and feed in the Gulf Cooperation Council countries and its detection. Toxicon 2019, 171, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Kluczkovski, A.M. Fungal and mycotoxin problems in the nut industry. Curr. Opin. Food Sci. 2019, 29, 56–63. [Google Scholar] [CrossRef]
- Sanzani, S.M.; Reverberi, M.; Geisen, R. Mycotoxins in harvested fruits and vegetables: Insights in producing fungi, biological role, conducive conditions, and tools to manage postharvest contamination. Postharvest Boil. Technol. 2016, 122, 95–105. [Google Scholar] [CrossRef]
- Gonçalves, B.L.; Coppa, C.C.; De Neeff, D.V.; Corassin, C.H.; De Oliveira, C.A.F. Mycotoxins in fruits and fruit-based products: Occurrence and methods for decontamination. Toxin Rev. 2018, 38, 263–272. [Google Scholar] [CrossRef]
- Varzakas, T. Quality and Safety Aspects of Cereals (Wheat) and Their Products. Crit. Rev. Food Sci. Nutr. 2015, 56, 2495–2510. [Google Scholar] [CrossRef]
- Welke, J.E. Fungal and mycotoxin problems in grape juice and wine industries. Curr. Opin. Food Sci. 2019, 29, 7–13. [Google Scholar] [CrossRef]
- Pascari, X.; Ramos, A.J.; Marín, S.; Sanchis, V. Mycotoxins and beer. Impact of beer production process on mycotoxin contamination. A review. Food Res. Int. 2018, 103, 121–129. [Google Scholar] [CrossRef] [Green Version]
- Viegas, S.; Assunção, R.; Twaruźek, M.; Kosicki, R.; Grajewski, J.; Viegas, C. Mycotoxins feed contamination in a dairy farm - potential implications for milk contamination and workers’ exposure in a One Health approach. J. Sci. Food Agric. 2019, 100, 1118–1123. [Google Scholar] [CrossRef]
- Gambacorta, L.; El Darra, N.; Fakhoury, R.; Logrieco, A.; Solfrizzo, M. Incidence and levels of Alternaria mycotoxins in spices and herbs produced worldwide and commercialized in Lebanon. Food Control 2019, 106, 106724. [Google Scholar] [CrossRef]
- Bessaire, T.; Perrin, I.; Tarres, A.; Bebius, A.; Reding, F.; Theurillat, V. Mycotoxins in green coffee: Occurrence and risk assessment. Food Control 2019, 96, 59–67. [Google Scholar] [CrossRef]
- Huertas-Pérez, J.F.; Arroyo-Manzanares, N.; García-Campaña, A.; Gámiz-Gracia, L. Solid Phase Extraction as Sample Treatment for the Determination of Ochratoxin A in Foods: A Review. Crit. Rev. Food Sci. Nutr. 2016, 57, 3405–3420. [Google Scholar] [CrossRef] [PubMed]
- Kebede, H.; Liu, X.; **, J.; **ng, F. Current status of major mycotoxins contamination in food and feed in Africa. Food Control 2020, 110, 106975. [Google Scholar] [CrossRef]
- Agriopoulou, S.; Koliadima, A.; Karaiskakis, G.; Kapolos, J. Kinetic study of aflatoxins’ degradation in the presence of ozone. Food Control 2016, 61, 221–226. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; International Agency for Research on Cancer: Lyon, France, 1993; Volume 56. [Google Scholar]
- Alshannaq, A.F.; Yu, J.-H. Occurrence, Toxicity, and Analysis of Major Mycotoxins in Food. Int. J. Environ. Res. Public Health 2017, 14, 632. [Google Scholar] [CrossRef] [Green Version]
- Somsubsin, S.; Seebunrueng, K.; Boonchiangma, S.; Srijaranai, S. A simple solvent based microextraction for high performance liquid chromatographic analysis of aflatoxins in rice samples. Talanta 2018, 176, 172–177. [Google Scholar] [CrossRef]
- Leal, T.; Abrunhosa, L.; Domingues, L.; Venâncio, A.; Oliveira, C. BSA-based sample clean-up columns for ochratoxin A determination in wine: Method development and validation. Food Chem. 2019, 300, 125204. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Dou, X.-W.; Zhang, C.; Logrieco, A.; Yang, M. A Review of Current Methods for Analysis of Mycotoxins in Herbal Medicines. Toxins 2018, 10, 65. [Google Scholar] [CrossRef] [Green Version]
- Tittlemier, S.; Cramer, B.; Dall’Asta, C.; Iha, M.; Lattanzio, V.; Malone, R.; Maragos, C.; Solfrizzo, M.; Stranska-Zachariasova, M.; Stroka, J. Developments in mycotoxin analysis: An update for 2017–2018. World Mycotoxin J. 2019, 12, 3–29. [Google Scholar] [CrossRef] [Green Version]
- Krska, R.; Molinelli, A. Rapid test strips for analysis of mycotoxins in food and feed. Anal. Bioanal. Chem. 2008, 393, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, I.S.; Junior, A.G.D.S.; De Andrade, C.A.S.; Oliveira, M.D.L. Biosensors for early detection of fungi spoilage and toxigenic and mycotoxins in food. Curr. Opin. Food Sci. 2019, 29, 64–79. [Google Scholar] [CrossRef]
- Neethirajan, S.; Ragavan, K.; Weng, X. Agro-defense: Biosensors for food from healthy crops and animals. Trends Food Sci. Technol. 2018, 73, 25–44. [Google Scholar] [CrossRef]
- Tothill, I.E. Biosensors and nanomaterials and their application for mycotoxin determination. World Mycotoxin J. 2011, 4, 361–374. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez, A.; Rodríguez, M.; Andrade, M.J.; Cordoba, M.D.G. Detection of filamentous fungi in foods. Curr. Opin. Food Sci. 2015, 5, 36–42. [Google Scholar] [CrossRef]
- El Sheikha, A.F. New Strategies for Tracing Foodstuffs: Biological Barcodes Utilising PCR-DGGE. Adv. Food Technol. Nutr. Sci. Open J. 2015, 1, S1–S7. [Google Scholar] [CrossRef]
- Liang, K.; Liu, Q.X.; Xu, J.H.; Wang, Y.Q.; Okinda, C.S.; Shena, M.X. Determination and Visualization of Different Levels of Deoxynivalenol in Bulk Wheat Kernels by Hyperspectral Imaging. J. Appl. Spectrosc. 2018, 85, 953–961. [Google Scholar] [CrossRef]
- Femenias, A.; Gatius, F.; Ramos, A.J.; Sanchis, V.; Marín, S. Use of hyperspectral imaging as a tool for Fusarium and deoxynivalenol risk management in cereals: A review. Food Control 2020, 108, 106819. [Google Scholar] [CrossRef]
- Leite, M.; Freitas, A.; Silva, A.S.; Barbosa, J.; Ramos, F. Maize (Zea mays L.) and mycotoxins: A review on optimization and validation of analytical methods by liquid chromatography coupled to mass spectrometry. Trends Food Sci. Technol. 2020. [Google Scholar] [CrossRef]
- Valenzano, S.; Lippolis, V.; Pascale, M.; De Marco, A.; Maragos, C.M.; Suman, M.; Visconti, A. Determination of Deoxynivalenol in Wheat Bran and Whole-Wheat Flour by Fluorescence Polarization Immunoassay. Food Anal. Methods 2013, 7, 806–813. [Google Scholar] [CrossRef]
- Spanjer, M.C.; Rensen, P.M.; Scholten, J.M. LC–MS/MS multi-method for mycotoxins after single extraction, with validation data for peanut, pistachio, wheat, maize, cornflakes, raisins and figs. Food Addit. Contam. Part A 2008, 25, 472–489. [Google Scholar] [CrossRef]
- Di Mavungu, J.D.; Monbaliu, S.; Scippo, M.-L.; Maghuin-Rogister, G.; Schneider, Y.-J.; Larondelle, Y.; Callebaut, A.; Robbens, J.; Van Peteghem, C.; De Saeger, S. LC-MS/MS multi-analyte method for mycotoxin determination in food supplements. Food Addit. Contam. Part A 2009, 26, 885–895. [Google Scholar] [CrossRef] [Green Version]
- Delmulle, B.S.; De Saeger, S.; Adams, A.; De Kimpe, N.; Van Peteghem, C. Development of a liquid chromatography/tandem mass spectrometry method for the simultaneous determination of 16 mycotoxins on cellulose filters and in fungal cultures. Rapid Commun. Mass Spectrom. 2006, 20, 771–776. [Google Scholar] [CrossRef]
- Razzazi-Fazeli, E.; Reiter, E. Sample preparation and clean up in mycotoxin analysis: Principles, applications and recent developments. In Determining Mycotoxins and Mycotoxigenic Fungi in Food and Feed; De Saeger, S., Ed.; Woodhead Publishing: Cambridge, UK, 2011; pp. 37–70. [Google Scholar]
- He, T.; Zhou, T.; Wan, Y.; Tan, T. A Simple Strategy Based on Deep Eutectic Solvent for Determination of Aflatoxins in Rice Samples. Food Anal. Methods 2019, 13, 542–550. [Google Scholar] [CrossRef]
- Anastassiades, M.; Lehotay, S.; Stajnbaher, D. Quick, easy, cheap, effective, rugged, and safe (QuEChERS) approach for the determination of pesticide residues VC. In Proceedings of the 18th Annual Waste Testing and Quality Assurance Symposium Proceedings, Arlington, VA, USA, 10–15 August 2002; pp. 231–241. [Google Scholar]
- Pereira, V.; Fernandes, J.O.; Cunha, S.C. Comparative assessment of three cleanup procedures after QuEChERS extraction for determination of trichothecenes (type A and type B) in processed cereal-based baby foods by GC–MS. Food Chem. 2015, 182, 143–149. [Google Scholar] [CrossRef]
- Carballo, D.; Font, G.; Ferrer, E.; Berrada, H. Evaluation of Mycotoxin Residues on Ready-to-Eat Food by Chromatographic Methods Coupled to Mass Spectrometry in Tandem. Toxins 2018, 10, 243. [Google Scholar] [CrossRef] [Green Version]
- Koesukwiwat, U.; Sanguankaew, K.; Leepipatpiboon, N. Evaluation of a modified QuEChERS method for analysis of mycotoxins in rice. Food Chem. 2014, 153, 44–51. [Google Scholar] [CrossRef]
- Yang, J.; Li, J.; Jiang, Y.; Duan, X.; Qu, H.; Yang, B.; Chen, F.; Sivakumar, D. Natural Occurrence, Analysis, and Prevention of Mycotoxins in Fruits and their Processed Products. Crit. Rev. Food Sci. Nutr. 2013, 54, 64–83. [Google Scholar] [CrossRef]
- Du, L.-J.; Chu, C.; Warner, E.; Wang, Q.-Y.; Hu, Y.-H.; Chai, K.-J.; Cao, J.; Peng, L.-Q.; Chen, Y.-B.; Yang, J.; et al. Rapid microwave-assisted dispersive micro-solid phase extraction of mycotoxins in food using zirconia nanoparticles. J. Chromatogr. A 2018, 1561, 1–12. [Google Scholar] [CrossRef]
- Turner, N.; Subrahmanyam, S.; Piletsky, S.A. Analytical methods for determination of mycotoxins: A review. Anal. Chim. Acta 2009, 632, 168–180. [Google Scholar] [CrossRef]
- D’Arco, G.; Franzón, M.F.; Font, G.; Damiani, P.; Mañes, J. Analysis of fumonisins B1, B2 and B3 in corn-based baby food by pressurized liquid extraction and liquid chromatography/tandem mass spectrometry. J. Chromatogr. A 2008, 1209, 188–194. [Google Scholar] [CrossRef]
- Kou, D.; Mitra, S. Sample Preparation Techniques in Analytical Chemistry; Mitra, S., Ed.; Wiley: New Jersey, NJ, USA, 2003; Chapter 3; pp. 139–183. [Google Scholar]
- Eskilsson, C.S.; Björklund, E. Analytical-scale microwave-assisted extraction. J. Chromatogr. A 2000, 902, 227–250. [Google Scholar] [CrossRef]
- Woo, S.Y.; Ryu, S.; Tian, F.; Lee, S.Y.; Park, S.; Chun, H.S. Simultaneous Determination of Twenty Mycotoxins in the Korean Soybean Paste Doenjang by LC-MS/MS with Immunoaffinity Cleanup. Toxins 2019, 11, 594. [Google Scholar] [CrossRef] [Green Version]
- Ran, C.; Chen, D.; Ma, H.; Jiang, Y. Graphene oxide adsorbent based dispersive solid phase extraction coupled with multi-pretreatment clean-up for analysis of trace aflatoxins in traditional proprietary Chinese medicines. J. Chromatogr. B 2017, 1044, 120–126. [Google Scholar] [CrossRef]
- Dong, M.; Si, W.; Jiang, K.; Nie, D.; Wu, Y.; Zhao, Z.; De Saeger, S.; Han, Z. Multi-walled carbon nanotubes as solid-phase extraction sorbents for simultaneous determination of type A trichothecenes in maize, wheat and rice by ultra-high performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2015, 1423, 177–182. [Google Scholar] [CrossRef]
- Lattanzio, V.M.T.; Ciasca, B.; Powers, S.; Visconti, A. Improved method for the simultaneous determination of aflatoxins, ochratoxin A and Fusarium toxins in cereals and derived products by liquid chromatography–tandem mass spectrometry after multi-toxin immunoaffinity clean up. J. Chromatogr. A 2014, 1354, 139–143. [Google Scholar] [CrossRef]
- Irakli, M.; Skendi, A.; Papageorgiou, M. HPLC-DAD-FLD Method for Simultaneous Determination of Mycotoxins in Wheat Bran. J. Chromatogr. Sci. 2017, 55, 690–696. [Google Scholar] [CrossRef]
- Golge, O.; Hepsag, F.; Kabak, B. Determination of aflatoxins in walnut sujuk and Turkish delight by HPLC-FLD method. Food Control 2016, 59, 731–736. [Google Scholar] [CrossRef]
- Castegnaro, M.; Tozlovanu, M.; Wild, C.; Molinié, A.; Sylla, A.; Pfohl-Leszkowicz, A. Advantages and drawbacks of immunoaffinity columns in analysis of mycotoxins in food. Mol. Nutr. Food Res. 2006, 50, 480–487. [Google Scholar] [CrossRef]
- Ringot, D.; Chango, A.; Schneider, Y.-J.; Larondelle, Y. Toxicokinetics and toxicodynamics of ochratoxin A, an update. Chem. Interact. 2006, 159, 18–46. [Google Scholar] [CrossRef]
- Baggiani, C.; Giovannoli, C.; Anfossi, L. Man-Made Synthetic Receptors for Capture and Analysis of Ochratoxin A. Toxins 2015, 7, 4083–4098. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pichon, V.; Combès, A. Selective tools for the solid-phase extraction of Ochratoxin A from various complex samples: Immunosorbents, oligosorbents, and molecularly imprinted polymers. Anal. Bioanal. Chem. 2016, 408, 6983–6999. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, Y.; Chen, X.; Duan, N.; Wu, S.; Wang, Z.; Wei, X.; Wang, Y. Selection and characterization of DNA aptamers against Staphylococcus aureus enterotoxin C1. Food Chem. 2015, 166, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yu, J.; Yang, Q.; McDermott, J.; Scott, A.; Vukovich, M.; Lagrois, R.; Gong, Q.; Greenleaf, W.; Eisenstein, M.; et al. Multiparameter Particle Display (MPPD): A Quantitative Screening Method for the Discovery of Highly Specific Aptamers. Angew. Chem. 2016, 129, 762–765. [Google Scholar] [CrossRef] [Green Version]
- Xu, L.; Zhang, Z.; Zhang, Q.; Li, P. Mycotoxin Determination in Foods Using Advanced Sensors Based on Antibodies or Aptamers. Toxins 2016, 8, 239. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Huang, Y.; Duan, N.; Wu, S.; **a, Y.; Ma, X.; Zhu, C.; Jiang, Y.; Ding, Z.; Wang, Z. Selection and characterization of single stranded DNA aptamers recognizing fumonisin B1. Microchim. Acta 2014, 181, 1317–1324. [Google Scholar] [CrossRef]
- Rhouati, A.; Bulbul, G.; Latif, U.; Hayat, A.; Li, Z.; Marty, J.L. Nano-Aptasensing in Mycotoxin Analysis: Recent Updates and Progress. Toxins 2017, 9, 349. [Google Scholar] [CrossRef] [Green Version]
- **a, X.; Wang, Y.; Yang, H.; Dong, Y.; Zhang, K.; Lu, Y.; Deng, R.; He, Q. Enzyme-free amplified and ultrafast detection of aflatoxin B1 using dual-terminal proximity aptamer probes. Food Chem. 2019, 283, 32–38. [Google Scholar] [CrossRef]
- Pereira, V.; Fernandes, J.O.; Cunha, S.C. Mycotoxins in cereals and related foodstuffs: A review on occurrence and recent methods of analysis. Trends Food Sci. Technol. 2014, 36, 96–136. [Google Scholar] [CrossRef]
- Soares, R.R.G.; Ricelli, A.; Fanelli, C.; Caputo, D.; De Cesare, G.; Chu, V.; Aires-Barros, M.R.; Conde, J.P. Advances, challenges and opportunities for point-of-need screening of mycotoxins in foods and feeds. Analyst 2018, 143, 1015–1035. [Google Scholar] [CrossRef]
- Stroka, J.; Maragos, C.M. Challenges in the analysis of multiple mycotoxins. World Mycotoxin J. 2016, 9, 847–861. [Google Scholar] [CrossRef]
- Lin, L.; Zhang, J.; Wang, P.; Wang, Y.; Chen, J. Thin-layer chromatography of mycotoxins and comparison with other chromatographic methods. J. Chromatogr. A 1998, 815, 3–20. [Google Scholar] [CrossRef]
- Aiko, V.; Mehta, A. Prevalence of toxigenic fungi in common medicinal herbs and spices in India. 3 Biotech 2016, 6, 159. [Google Scholar] [CrossRef] [Green Version]
- Ezekwesili-Ofili, J.; Onyemelukwe, N.; Agwaga, P.; Orji, I. The Bioload and Aflatoxin Content of Herbal Medicines from Selected States in Nigeria. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 143–147. [Google Scholar] [CrossRef] [Green Version]
- Andrade, P.; Homem-De-Mello, M.; França, J.; Caldas, E.D. Aflatoxins in food products consumed in Brazil: A preliminary dietary risk assessment. Food Addit. Contam. Part A 2013, 30, 127–136. [Google Scholar] [CrossRef]
- Abdel-Gawad, K.M.; Zohri, A.A. Fungal flora and mycotoxins of six kinds of nut seeds for human consumption in Saudi Arabia. Mycopathologia 1993, 124, 55–64. [Google Scholar] [CrossRef]
- Ahmad, B.; Ashiq, S.; Hussain, A.; Bashir, S.; Hussain, M. Evaluation of mycotoxins, mycobiota, and toxigenic fungi in selected medicinal plants of Khyber Pakhtunkhwa, Pakistan. Fungal Boil. 2014, 118, 776–784. [Google Scholar] [CrossRef]
- Caldas, E.D.; Silva, A.C.S. Mycotoxins in Corn-Based Food Products Consumed in Brazil: An Exposure Assessment for Fumonisins. J. Agric. Food Chem. 2007, 55, 7974–7980. [Google Scholar] [CrossRef]
- Jeleń, H.; Wasowicz, E. Determination of trichothecenes in wheat grain without sample cleanup using comprehensive two-dimensional gas chromatography–time-of-flight mass spectrometry. J. Chromatogr. A 2008, 1215, 203–207. [Google Scholar] [CrossRef]
- Rodríguez-Carrasco, Y.; Berrada, H.; Font, G.; Mañes, J. Multi-mycotoxin analysis in wheat semolina using an acetonitrile-based extraction procedure and gas chromatography–tandem mass spectrometry. J. Chromatogr. A 2012, 1270, 28–40. [Google Scholar] [CrossRef]
- Guo, W.; Fan, K.; Nie, D.; Meng, J.; Huang, Q.; Yang, J.; Shen, Y.; Tangni, E.K.; Zhao, Z.; Wu, Y.; et al. Development of a QuEChERS-Based UHPLC-MS/MS Method for Simultaneous Determination of Six Alternaria Toxins in Grapes. Toxins 2019, 11, 87. [Google Scholar] [CrossRef] [Green Version]
- Valenta, H. Chromatographic methods for the determination of ochratoxin A in animal and human tissues and fluids. J. Chromatogr. A 1998, 815, 75–92. [Google Scholar] [CrossRef]
- Lai, X.; Liu, R.; Ruan, C.; Zhang, H.; Liu, C. Occurrence of aflatoxins and ochratoxin A in rice samples from six provinces in China. Food Control 2015, 50, 401–404. [Google Scholar] [CrossRef]
- Ma, L.; Xu, W.; He, X.; Huang, K.; Wang, Y.; Luo, Y. Determination of fumonisins B1and B2in Chinese rice wine by HPLC using AQC precolumn derivatisation. J. Sci. Food Agric. 2012, 93, 1128–1133. [Google Scholar] [CrossRef] [PubMed]
- Myresiotis, C.K.; Testempasis, S.; Vryzas, Z.; Karaoglanidis, G.S.; Papadopoulou-Mourkidou, E. Determination of mycotoxins in pomegranate fruits and juices using a QuEChERS-based method. Food Chem. 2015, 182, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Pascale, M.; De Girolamo, A.; Lippolis, V.; Stroka, J.; Mol, H.G.J.; Lattanzio, V.M.T. Performance evaluation of LC-MS methods for multimycotoxin determination. J. AOAC Int. 2019, 102, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Bessaire, T.; Mujahid, C.; Mottier, P.; Desmarchelier, A. Multiple Mycotoxins Determination in Food by LC-MS/MS: An International Collaborative Study. Toxins 2019, 11, 658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Taher, F.; Cappozzo, J.; Zweigenbaum, J.; Lee, H.; Jackson, L.S.; Ryu, D. Detection and quantitation of mycotoxins in infant cereals in the U.S. market by LC-MS/MS using a stable isotope dilution assay. Food Control 2017, 72, 27–35. [Google Scholar] [CrossRef] [Green Version]
- EN 16924:2017. Foodstuffs—Determination of Zearalenone in Edible Vegetable Oils by LC-FLD or LC-MS/MS; European Committee for Standardization: Brussels, Belgium, 2017. [Google Scholar]
- EN 16923:2017. Foodstuffs—Determination ofT-2 Toxin and HT-2 Toxin in Cereals and Cereal Products for Infants and Young Children by LC-MS/MS after SPE Cleanup; European Committee for Standardization: Brussels, Belgium, 2017. [Google Scholar]
- Habler, K.; Gotthardt, M.; Schüler, J.; Rychlik, M. Multi-mycotoxin stable isotope dilution LC—MS/MS method for Fusarium toxins in beer. Food Chem. 2017, 218, 447–454. [Google Scholar] [CrossRef] [Green Version]
- Bolechová, M.; Benešová, K.; Běláková, S.; Čáslavský, J.; Pospíchalová, M.; Mikulíková, R. Determination of seventeen mycotoxins in barley and malt in the Czech Republic. Food Control 2015, 47, 108–113. [Google Scholar] [CrossRef]
- Han, Z.; Ren, Y.; Zhu, J.; Cai, Z.; Chen, Y.; Luan, L.; Wu, Y. Multianalysis of 35 Mycotoxins in Traditional Chinese Medicines by Ultra-High-Performance Liquid Chromatography–Tandem Mass Spectrometry Coupled with Accelerated Solvent Extraction. J. Agric. Food Chem. 2012, 60, 8233–8247. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-P.; Zhao, Y.-G.; Chen, X.-H.; Pan, S.-D.; **, M.-C. Simultaneous determination of four aflatoxins in walnut kernel using dispersive solid-phase extraction combined with ultra fast liquid chromatography-tandem mass spectrometry. Chin. J. Health Lab. Technol. 2014, 24, 2647–2650. [Google Scholar]
- Tölgyesi, Á.; Stroka, J.; Tamošiūnas, V.; Zwickel, T. Simultaneous analysis of Alternaria toxins and citrinin in tomato: An optimised method using liquid chromatography-tandem mass spectrometry. Food Addit. Contam. Part A 2015, 32, 1512–1522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, W.; Han, X.; Li, F.; Zhang, L. Natural Occurrence of Alternaria Toxins in the 2015 Wheat from Anhui Province, China. Toxins 2016, 8, 308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hidalgo-Ruiz, J.L.; Romero-González, R.; Vidal, J.L.M.; Frenich, A.G. A rapid method for the determination of mycotoxins in edible vegetable oils by ultra-high performance liquid chromatography-tandem mass spectrometry. Food Chem. 2019, 288, 22–28. [Google Scholar] [CrossRef]
- Flores-Flores, M.E.; González-Peñas, E. An LC–MS/MS method for multi-mycotoxin quantification in cow milk. Food Chem. 2017, 218, 378–385. [Google Scholar] [CrossRef]
- Zhao, H.; Chen, X.; Shen, C.; Qu, B. Determination of 16 mycotoxins in vegetable oils using a QuEChERS method combined with high performance liquid chromatography tandem mass spectrometry. Food Addit. Contam. Part A 2016, 34, 1–10. [Google Scholar] [CrossRef]
- De Santis, B.; Debegnach, F.; Gregori, E.; Russo, S.; Marchegiani, F.; Moracci, G.; Brera, C. Development of a LC-MS/MS Method for the Multi-Mycotoxin Determination in Composite Cereal-Based Samples. Toxins 2017, 9, 169. [Google Scholar] [CrossRef] [Green Version]
- Kim, N.-H.; Hong, S.-Y.; Kang, J.W.; Cho, S.M.; Lee, K.R.; An, T.K.; Lee, C.; Chung, S.H. Simultaneous Determination of Multi-Mycotoxins in Cereal Grains Collected from South Korea by LC/MS/MS. Toxins 2017, 9, 106. [Google Scholar] [CrossRef] [Green Version]
- Urusov, A.; Zherdev, A.V.; Petrakova, A.V.; Sadykhov, E.G.; Koroleva, O.V.; Dzantiev, B.B. Rapid Multiple Immunoenzyme Assay of Mycotoxins. Toxins 2015, 7, 238–254. [Google Scholar] [CrossRef]
- Nolan, P.; Auer, S.; Spehar, A.; Elliott, C.T.; Campbell, K. Current trends in rapid tests for mycotoxins. Food Addit. Contam. Part A 2019, 36, 800–814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oplatowska-Stachowiak, M.; Sajic, N.; Xu, Y.; Haughey, S.A.; Mooney, M.H.; Gong, Y.Y.; Verheijen, R.; Elliott, C.T. Fast and sensitive aflatoxin B1 and total aflatoxins ELISAs for analysis of peanuts, maize and feed ingredients. Food Control 2016, 63, 239–245. [Google Scholar] [CrossRef]
- Tripathi, P.; Upadhyay, N.; Nara, S. Recent advancements in lateral flow immunoassays: A journey for toxin detection in food. Crit. Rev. Food Sci. Nutr. 2017, 58, 1715–1734. [Google Scholar] [CrossRef] [PubMed]
- Anfossi, L.; Di Nardo, F.; Cavalera, S.; Giovannoli, C.; Spano, G.; Speranskaya, E.S.; Goryacheva, I.; Baggiani, C. A lateral flow immunoassay for straightforward determination of fumonisin mycotoxins based on the quenching of the fluorescence of CdSe/ZnS quantum dots by gold and silver nanoparticles. Microchim. Acta 2018, 185, 94. [Google Scholar] [CrossRef] [PubMed]
- Anfossi, L.; Di Nardo, F.; Russo, A.; Cavalera, S.; Giovannoli, C.; Spano, G.; Baumgartner, S.; Lauter, K.; Baggiani, C. Silver and gold nanoparticles as multi-chromatic lateral flow assay probes for the detection of food allergens. Anal. Bioanal. Chem. 2018, 411, 1905–1913. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Xu, H.; Wei, M.; Gu, H.; Xu, Q.; Zhu, W. Study of superparamagnetic nanoparticles as labels in the quantitative lateral flow immunoassay. Mater. Sci. Eng. C 2009, 29, 714–718. [Google Scholar] [CrossRef]
- Zhang, X.; Yu, X.; Wen, K.; Li, C.; Mari, G.M.; Jiang, H.; Shi, W.; Shen, J.; Wang, Z. Multiplex Lateral Flow Immunoassays Based on Amorphous Carbon Nanoparticles for Detecting Three Fusarium Mycotoxins in Maize. J. Agric. Food Chem. 2017, 65, 8063–8071. [Google Scholar] [CrossRef]
- Foubert, A.; Beloglazova, N.V.; Gordienko, A.; Tessier, M.; Drijvers, E.; Hens, Z.; De Saeger, S. Development of a Rainbow Lateral Flow Immunoassay for the Simultaneous Detection of Four Mycotoxins. J. Agric. Food Chem. 2016, 65, 7121–7130. [Google Scholar] [CrossRef]
- Song, S.; Liu, N.; Wu, A.-B.; Ediage, E.N.; Wu, S.; Sun, C.; De Saeger, S.; Wu, A. Multiplex Lateral Flow Immunoassay for Mycotoxin Determination. Anal. Chem. 2014, 86, 4995–5001. [Google Scholar] [CrossRef]
- Yu, Q.; Li, H.; Li, C.; Zhang, S.; Shen, J.; Wang, Z. Gold nanoparticles-based lateral flow immunoassay with silver staining for simultaneous detection of fumonisin B1 and deoxynivalenol. Food Control 2015, 54, 347–352. [Google Scholar] [CrossRef]
- Li, R.; Meng, C.; Wen, Y.; Fu, W.; He, P. Fluorometric lateral flow immunoassay for simultaneous determination of three mycotoxins (aflatoxin B1, zearalenone and deoxynivalenol) using quantum dot microbeads. Microchim. Acta 2019, 186, 748. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.; Chen, X.; Xu, W.; Fu, J.; **ong, Y.; Wang, A. Quantum-DoT submicrobead-based immunochromatographic assay for quantitative and sensitive detection of zearalenone. Talanta 2015, 132, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Pirinçci, Ş.; Ertekin, Ö.; Laguna, D.E.; Özen, F.Ş.; Ozturk, Z.Z.; Öztürk, S. Label-Free QCM Immunosensor for the Detection of Ochratoxin A. Sensors 2018, 18, 1161. [Google Scholar] [CrossRef] [Green Version]
- Slaughter, G. Current Advances in Biosensor Design and Fabrication. In Encyclopedia of Analytical Chemistry; Wiley: Hoboken, NJ, USA, 2018; pp. 1–25. [Google Scholar]
- Schulz, K.; Pöhlmann, C.; Dietrich, R.; Märtlbauer, E.; Elßner, T. Electrochemical Biochip Assays Based on Anti-idiotypic Antibodies for Rapid and Automated On-Site Detection of Low Molecular Weight Toxins. Front. Chem. 2019, 7, 31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malekzad, H.; Zangabad, P.S.; Mirshekari, H.; Karimi, M.; Hamblin, M.R. Noble metal nanoparticles in biosensors: Recent studies and applications. Nanotechnol. Rev. 2017, 6, 301–329. [Google Scholar] [CrossRef] [PubMed]
- Doria, G.; Conde, J.; Veigas, B.; Giestas, L.; Almeida, C.; Assunção, M.; Rosa, J.; Baptista, P.V. Noble Metal Nanoparticles for Biosensing Applications. Sensors 2012, 12, 1657–1687. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Wang, Y.; Wu, X.; Pan, M.; Hu, N.; Wang, J.; Wang, S. Quartz crystal microbalance sensor based on covalent organic framework composite and molecularly imprinted polymer of poly(o-aminothiophenol) with gold nanoparticles for the determination of aflatoxin B1. Sens. Actuators B Chem. 2019, 291, 293–297. [Google Scholar] [CrossRef]
- Yagati, A.K.; Chavan, S.G.; Baek, C.; Lee, M.-H.; Min, J. Label-Free Impedance Sensing of Aflatoxin B1 with Polyaniline Nanofibers/Au Nanoparticle Electrode Array. Sensors 2018, 18, 1320. [Google Scholar] [CrossRef] [Green Version]
- Chauhan, R.; Singh, J.; Solanki, P.R.; Manaka, T.; Iwamoto, M.; Basu, T.; Malhotra, B.D. Label-free piezoelectric immunosensor decorated with gold nanoparticles: Kinetic analysis and biosensing application. Sens. Actuators B Chem. 2016, 222, 804–814. [Google Scholar] [CrossRef]
- Tang, Y.; Tang, D.-Y.; Zhang, J.; Tang, D. Novel quartz crystal microbalance immunodetection of aflatoxin B1 coupling cargo-encapsulated liposome with indicator-triggered displacement assay. Anal. Chim. Acta 2018, 1031, 161–168. [Google Scholar] [CrossRef]
- Zhang, X.; Li, C.-R.; Wang, W.-C.; Xue, J.; Huang, Y.-L.; Yang, X.-X.; Tan, B.; Zhou, X.-P.; Shao, C.; Ding, S.-J.; et al. A novel electrochemical immunosensor for highly sensitive detection of aflatoxin B1 in corn using single-walled carbon nanotubes/chitosan. Food Chem. 2016, 192, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Hossain, Z.; Busman, M.; Maragos, C.M. Immunoassay utilizing imaging surface plasmon resonance for the detection of cyclopiazonic acid (CPA) in maize and cheese. Anal. Bioanal. Chem. 2019, 411, 3543–3552. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.Z.; Maragos, C.M. Gold nanoparticle-enhanced multiplexed imaging surface plasmon resonance (iSPR) detection of Fusarium mycotoxins in wheat. Biosens. Bioelectron. 2018, 101, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Hossain, Z.; McCormick, S.; Maragos, C.M. An Imaging Surface Plasmon Resonance Biosensor Assay for the Detection of T-2 Toxin and Masked T-2 Toxin-3-Glucoside in Wheat. Toxins 2018, 10, 119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nan, M.; Bi, Y.; Xue, H.; Xue, S.; Long, H.; Pu, L.; Fu, G. Rapid Determination of Ochratoxin A in Grape and Its Commodities Based on a Label-Free Impedimetric Aptasensor Constructed by Layer-by-Layer Self-Assembly. Toxins 2019, 11, 71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, Z.; Feng, M.; Zuo, L.; Zhu, Z.; Wang, F.; Chen, L.; Li, J.; Shan, G.; Luo, S.-Z. An aptamer based surface plasmon resonance biosensor for the detection of ochratoxin A in wine and peanut oil. Biosens. Bioelectron. 2015, 65, 320–326. [Google Scholar] [CrossRef]
- Tang, J.; Huang, Y.; Cheng, Y.; Huang, L.; Zhuang, J.; Tang, D. Two-dimensional MoS2 as a nano-binder for ssDNA: Ultrasensitive aptamer based amperometric detection of Ochratoxin A. Microchim. Acta 2018, 185, 162. [Google Scholar] [CrossRef]
- Karczmarczyk, A.; Haupt, K.; Feller, K.-H. Development of a QCM-D biosensor for Ochratoxin A detection in red wine. Talanta 2017, 166, 193–197. [Google Scholar] [CrossRef]
- Rehmat, Z.; Mohammed, W.; Sadiq, M.B.; Somarapalli, M.; Anal, A.K. Ochratoxin A detection in coffee by competitive inhibition assay using chitosan-based surface plasmon resonance compact system. Colloids Surf. B Biointerfaces 2019, 174, 569–574. [Google Scholar] [CrossRef]
- **ang, Y.; Camarada, M.B.; Wen, Y.; Wu, H.; Chen, J.; Li, M.; Liao, X. Simple voltammetric analyses of ochratoxin A in food samples using highly-stable and anti-fouling black phosphorene nanosensor. Electrochim. Acta 2018, 282, 490–498. [Google Scholar] [CrossRef]
- Karczmarczyk, A.; Baeumner, A.J.; Feller, K.-H. Rapid and sensitive inhibition-based assay for the electrochemical detection of Ochratoxin A and Aflatoxin M1 in red wine and milk. Electrochim. Acta 2017, 243, 82–89. [Google Scholar] [CrossRef]
- Karczmarczyk, A.; Dubiak-Szepietowska, M.; Vorobii, M.; Rodriguez-Emmenegger, C.; Dostalek, J.; Feller, K.-H. Sensitive and rapid detection of aflatoxin M1 in milk utilizing enhanced SPR and p(HEMA) brushes. Biosens. Bioelectron. 2016, 81, 159–165. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Dong, X. Aptamer based voltammetric patulin assay based on the use of ZnO nanorods. Microchim. Acta 2018, 185, 462. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.; Ben Aissa, S.; Sherazi, T.A.; Catanante, G.; Hayat, A.; Marty, J.L. Development of an Impedimetric Aptasensor for Label Free Detection of Patulin in Apple Juice. Molcules 2019, 24, 1017. [Google Scholar] [CrossRef] [Green Version]
- He, B.; Yan, X. An amperometric zearalenone aptasensor based on signal amplification by using a composite prepared from porous platinum nanotubes, gold nanoparticles and thionine-labelled graphene oxide. Microchim. Acta 2019, 186, 383. [Google Scholar] [CrossRef]
- Liu, N.; Nie, D.; Tan, Y.; Wu, A.-B.; Liao, Y.; Wang, H.; Sun, C.; Wu, A. An ultrasensitive amperometric immunosensor for zearalenones based on oriented antibody immobilization on a glassy carbon electrode modified with MWCNTs and AuPt nanoparticles. Microchim. Acta 2016, 184, 147–153. [Google Scholar] [CrossRef]
- He, B.; Yan, X. A “signal-on” voltammetric aptasensor fabricated by hcPt@AuNFs/PEI-rGO and Fe3O4NRs/rGO for the detection of zearalenone. Sens. Actuators B Chem. 2019, 290, 477–483. [Google Scholar] [CrossRef]
- Joshi, S.; Segarra-Fas, A.; Peters, J.; Zuilhof, H.; Van Beek, T.A.; Nielen, M.W.F. Multiplex surface plasmon resonance biosensing and its transferability towards imaging nanoplasmonics for detection of mycotoxins in barley. Analyst 2016, 141, 1307–1318. [Google Scholar] [CrossRef]
- Ram, Y.; Yoetz-Kopelman, T.; Dror, Y.; Freeman, A.; Shacham-Diamand, Y. Impact of Molecular Surface Charge on Biosensing by Electrochemical Impedance Spectroscopy. Electrochim. Acta 2016, 200, 161–167. [Google Scholar] [CrossRef]
- Perumal, V.; Hashim, U. Advances in biosensors: Principle, architecture and applications. J. Appl. Biomed. 2014, 12, 1–15. [Google Scholar] [CrossRef]
- Hammond, J.L.; Formisano, N.; Carrara, S.; Tkac, J. Electrochemical biosensors and nanobiosensors. Essays Biochem. 2016, 60, 69–80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grieshaber, D.; MacKenzie, R.; Vörös, J.; Reimhult, E. Electrochemical Biosensors—Sensor Principles and Architectures. Sensors 2008, 8, 1400–1458. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.A.P.; Venturini, C.; Castilho, M.D.S.; Canaverolo, N.; Vinicius, M.; Dos Santos, G.P.; Sadao, C.; Vicente, A.; Yamanak, H. Amperometric Biosensor for Diagnosis of Disease; InTech: Rijeka, Croatia, 2013; pp. 253–289. [Google Scholar]
- Ricci, F.; Volpe, G.; Micheli, L.; Palleschi, G. A review on novel developments and applications of immunosensors in food analysis. Anal. Chim. Acta 2007, 605, 111–129. [Google Scholar] [CrossRef] [PubMed]
- Damborský, P.; Švitel, J.; Katrlík, J.; Vitel, J. Optical biosensors. Essays Biochem. 2016, 60, 91–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hill, R.T. Plasmonic biosensors. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2014, 7, 152–168. [Google Scholar] [CrossRef] [Green Version]
- Montagut, Y.; Garcia, J.; Jiménez, Y.; March, C.; Montoya, A.; Arnau, A. QCM Technology in Biosensors. In Biosensors—Emerging Materials and Applications; IntechOpen: Rijeka, Croatia, 2011; pp. 153–178. [Google Scholar]
- Vidal, J.C.; Bonel, L.; Ezquerra, A.; Hernandez, S.; Bertolín, J.R.; Cubel, C.; Castillo, J.R. Electrochemical affinity biosensors for detection of mycotoxins: A review. Biosens. Bioelectron. 2013, 49, 146–158. [Google Scholar] [CrossRef]
- Rameil, S.; Schubert, P.; Grundmann, P.; Dietrich, R.; Märtlbauer, E. Use of 3-(4-hydroxyphenyl)propionic acid as electron donating compound in a potentiometric aflatoxin M1-immunosensor. Anal. Chim. Acta 2010, 661, 122–127. [Google Scholar] [CrossRef]
- Spinella, K.; Mosiello, L.; Palleschi, G.; Vitali, F. Development of a QCM (Quartz Crystal Microbalance) Biosensor to Detection of Mycotoxins. Lect. Notes Electr. Eng. 2013, 268, 195–198. [Google Scholar] [CrossRef]
- Rodrigues, P.; Santos, C.; Venâncio, A.; Lima, N. Species identification of Aspergillus section Flavi isolates from Portuguese almonds using phenotypic, including MALDI-TOF ICMS, and molecular approaches. J. Appl. Microbiol. 2011, 111, 877–892. [Google Scholar] [CrossRef]
- Panda, A.; Ghosh, A.K.; Mirdha, B.R.; Xess, I.; Paul, S.; Samantaray, J.C.; Srinivasan, A.; Khalil, S.; Rastogi, N.; Dabas, Y. MALDI-TOF mass spectrometry for rapid identification of clinical fungal isolates based on ribosomal protein biomarkers. J. Microbiol. Methods 2015, 109, 93–105. [Google Scholar] [CrossRef]
- Hleba, L.; Císarová, M.; Shariati, M.A.; Tancinová, D. Detection of mycotoxins using MALDI-TOF Mass SpectrometryJ. Microbiol. Biotechnol. Food Sci. 2017, 7, 181–185. [Google Scholar]
- Sivagnanam, K.; Komatsu, E.; Rampitsch, C.; Perreault, H.; Gräfenhan, T. Rapid screening of Alternaria mycotoxins using MALDI-TOF mass spectrometry. J. Sci. Food Agric. 2016, 97, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Bintvihok, A.; Treebonmuang, S.; Srisakwattana, K.; Nuanchun, W.; Patthanachai, K.; Usawang, S. A Rapid and Sensitive Detection of Aflatoxin-producing Fungus Using an Optimized Polymerase Chain Reaction (PCR). Toxicol. Res. 2016, 32, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Hayat, A.; Paniel, N.; Rhouati, A.; Marty, J.-L.; Barthelmebs, L. Recent advances in ochratoxin A-producing fungi detection based on PCR methods and ochratoxin A analysis in food matrices. Food Control 2012, 26, 401–415. [Google Scholar] [CrossRef]
- Midorikawa, G.E.O.; Miller, R.N.G.; Bittencourt, D.M.d.C. Molecular Techniques in Food Biology: Safety, Biotechnology, Authenticity and Traceability, 1st ed.; El Sheikha, A.F., Levin, R., Xu, J., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2018; Volume 3, pp. 385–407. [Google Scholar]
- Luo, J.; Vogel, R.F.; Niessen, L. Development and application of a loop-mediated isothermal amplification assay for rapid identification of aflatoxigenic molds and their detection in food samples. Int. J. Food Microbiol. 2012, 159, 214–224. [Google Scholar] [CrossRef]
- Falasconi, M.; Concina, I.; Gobbi, E.; Sberveglieri, V.; Pulvirenti, A.; Sberveglieri, G. Electronic Nose for Microbiological Quality Control of Food Products. Int. J. Electrochem. 2012, 2012, 1–12. [Google Scholar] [CrossRef]
- Casalinuovo, I.A.; Di Pierro, N.; Coletta, M.; Di Francesco, P. Application of Electronic Noses for Disease Diagnosis and Food Spoilage Detection. Sensors 2006, 6, 1428–1439. [Google Scholar] [CrossRef] [Green Version]
- Sanaeifar, A.; Zakidizaji, H.; Jafari, A.; De La Guardia, M. Early detection of contamination and defect in foodstuffs by electronic nose: A review. TrAC Trends Anal. Chem. 2017, 97, 257–271. [Google Scholar] [CrossRef]
- Jia, W.; Liang, G.; Tian, H.; Sun, J.; Wan, C. Electronic Nose-Based Technique for Rapid Detection and Recognition of Moldy Apples. Sensors 2019, 19, 1526. [Google Scholar] [CrossRef] [Green Version]
- Gruber, J.; Nascimento, H.M.; Yamauchi, E.Y.; Li, R.W.; Esteves, C.H.; Rehder, G.P.; Gaylarde, C.; Shirakawa, M.A. A conductive polymer based electronic nose for early detection of Penicillium digitatum in post-harvest oranges. Mater. Sci. Eng. C 2013, 33, 2766–2769. [Google Scholar] [CrossRef]
- Liu, Q.; Sun, K.; Zhao, N.; Yang, J.; Zhang, Y.; Ma, C.; Pan, L.; Tu, K. Information fusion of hyperspectral imaging and electronic nose for evaluation of fungal contamination in strawberries during decay. Postharvest Boil. Technol. 2019, 153, 152–160. [Google Scholar] [CrossRef]
- Liu, Q.; Zhao, N.; Zhou, D.; Sun, Y.; Sun, K.; Pan, L.; Tu, K. Discrimination and growth tracking of fungi contamination in peaches using electronic nose. Food Chem. 2018, 262, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission. Chem. Soc. Rev. 2011, 40, 5361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Liu, G. Advances in luminescent materials with aggregation-induced emission (AIE) properties for biomedical applications. J. Mater. Chem. B 2018, 6, 4029–4042. [Google Scholar] [CrossRef]
- Wang, H.; Liu, G.; Gao, H.; Wang, Y. A pH-responsive drug delivery system with an aggregation-induced emission feature for cell imaging and intracellular drug delivery. Polym. Chem. 2015, 6, 4715–4718. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, X.; Yang, B.; Deng, F.; Huang, Z.; Yang, Y.; Li, Z.; Zhang, X.; Wei, Y. Ultrabright and biocompatible AIE dye based zwitterionic polymeric nanoparticles for biological imaging. RSC Adv. 2014, 4, 35137–35143. [Google Scholar] [CrossRef]
- Zhu, Y.; **a, X.; Deng, S.; Yan, B.; Dong, Y.; Zhang, K.; Deng, R.; He, Q. Label-free fluorescent aptasensing of mycotoxins via aggregation-induced emission dye. Dye. Pigment. 2019, 170, 107572. [Google Scholar] [CrossRef]
- Xu, Y.-J.; Wang, C.; Ho, W.E.; Ong, C.N. Recent developments and applications of metabolomics in microbiological investigations. TrAC Trends Anal. Chem. 2014, 56, 37–48. [Google Scholar] [CrossRef]
- Serkova, N.J.; Brown, M.S. Quantitative analysis in magnetic resonance spectroscopy: From metabolic profiling to in vivo biomarkers. Bioanalysis 2012, 4, 321–341. [Google Scholar] [CrossRef]
- Kleigrewe, K.; Aydin, F.; Hogrefe, K.; Piecuch, P.; Bergander, K.; Würthwein, E.-U.; Humpf, H.-U. Structure Elucidation of New Fusarins Revealing Insights in the Rearrangement Mechanisms of the Fusarium Mycotoxin Fusarin C. J. Agric. Food Chem. 2012, 60, 5497–5505. [Google Scholar] [CrossRef]
- López-Ruiz, R.; Romero-González, R.; Frenich, A.G. Metabolomics approaches for the determination of multiple contaminants in food. Curr. Opin. Food Sci. 2019, 28, 49–57. [Google Scholar] [CrossRef]
- ElMasry, G.; Sun, D.W. Principles of hyperspectral imaging technology. In Hyperspectral Imaging for Food Quality Analysis and Control; Sun, D.W., Ed.; Elsevier: London, UK, 2010; pp. 3–43. [Google Scholar]
- Qin, J.; Kim, M.; Chao, K.; Chan, D.E.; Delwiche, S.R.; Cho, B.-K. Line-Scan Hyperspectral Imaging Techniques for Food Safety and Quality Applications. Appl. Sci. 2017, 7, 125. [Google Scholar] [CrossRef] [Green Version]
- Elmasry, G.; Kamruzzaman, M.; Sun, D.-W.; Allen, P. Principles and Applications of Hyperspectral Imaging in Quality Evaluation of Agro-Food Products: A Review. Crit. Rev. Food Sci. Nutr. 2012, 52, 999–1023. [Google Scholar] [CrossRef] [PubMed]
- Tekle, S.; Måge, I.; Segtnan, V.H.; Bjørnstad, A. Near-Infrared Hyperspectral Imaging of Fusarium-Damaged Oats (Avena sativa L.). Cereal Chem. J. 2015, 92, 73–80. [Google Scholar] [CrossRef] [Green Version]
- Barbedo, J.G.; Tibola, C.S.; Lima, M.I.P. Deoxynivalenol screening in wheat kernels using hyperspectral imaging. Biosyst. Eng. 2017, 155, 24–32. [Google Scholar] [CrossRef]
- Ropelewska, E.; Zapotoczny, P. Classification of Fusarium-infected and healthy wheat kernels based on features from hyperspectral images and flatbed scanner images: A comparative analysis. Eur. Food Res. Technol. 2018, 244, 1453–1462. [Google Scholar] [CrossRef] [Green Version]
| Extraction Methods | Extraction Solvents | Limits | Benefits | Reference |
|---|---|---|---|---|
| QuEChERS | Organic solvents or mixtures (CH3CN, MeOH, MeOH/CH3CN) | Modifications of the original procedure, need of an additional enrichment step | Economical, fast, simple, detection of low ppb levels, better reproducibility and accuracy | [5] |
| LLE | Mixture of organic solvents (hexane, cyclohexane) with diluted acids or water | Time consuming, the sample can be absorbed by the glass equipment depending on the matrix and the determined compounds | Effective, for small-scale preparations | [40] |
| SLE | Mixture of organic solvents with diluted acids or water | Matrix effects | Smaller volumes of solvent | [32,45] |
| ASE or PLE | Mixture of organic solvents (MeOH/CH3CN, CH3CN/water) | Expensive instruments, matrix components excessively coextracted | Fully automated, faster extraction compared to the conventional ones, minimal solvents, higher extraction efficiency | [22,46] |
| SFE | Supercritical fluids (CO2), MeOH, ethanol, acetone | Need for specialized and very expensive equipment, not suitable for routine analysis | Fast technique, small solvent volumes, preconcentration effect, extraction of temperature sensible analytes | [22,45,47] |
| MAE | Aqueous solution | Only applicable for thermally stable compounds, costly instruments | Reduced extraction time and extraction solvent | [48] |
| VALDS–ME | Mixture of organic solvents dispersive solvent and water | Optimization after control a lot of parameters | Use of low density solvents, simple, fast, effective | [20] |
| Sample | Origin | Number of Samples | Mycotoxins | LOD | LOQ | References |
|---|---|---|---|---|---|---|
| Herbs and herbal products | India | 63 | AFB1, AFB2, AFG1, AFG2, CIT | 10 ng/mL for AFB1 NA for others | NA | [69] |
| Herbal medicines | Nigeria | 210 | AFB1, AFB2, AFG1, AFG2 | NA | NA | [70] |
| Brazil nuts | Brazil | 67 | AFB1, AFB2, AFG1, AFG2 | NA | 2 mg/kg | [71] |
| Almonds, cashew nuts, chestnuts, hazelnuts, pistachio nuts, walnuts | Saudi Arabia | 5 | AFB1, AFB2, AFG1, AFG2, CIT, OTs, PAT, T-2, ZEA, ST, DAS | NA | 5 μg/kg (for AFs), NA for others | [72] |
| Medicinal plants | Pakistan | 30 | AFB1, AFB2, AFG1, AFG2, OTA | NA | NA | [73] |
| Corn-based food products | Brazil | 208 | AFB1, AFB2, AFG1, AFG2 | NA | 2 µg/kg | [74] |
| Mycotoxin | Year of Publication | Country | Sample | Extraction Solution | Extraction Method | Clean-Up | LOD | LOQ * | Reference |
|---|---|---|---|---|---|---|---|---|---|
| AFs | 2014 | China | Walnut kernel | Methanol–water (70:30, v/v) | Sonicating | Self-made amino-function nanometer Fe3O4 magnetic polymer SPE | 0.004–0.013 µg kg−1 | 0.012–0.042 µg kg−1 | [90] |
| AFs, OTA Fusarium mycotoxins | 2014 | Italy | Cereals and derived products | Methanol–water (60:40, v/v) | Blending | IAC | 1 µg kg−1 for AFs and OTA 5–30 µg kg−1 for Fusarium toxins | Nd | [52] |
| 5 Alternaria mycotoxins, CIT | 2015 | Belgium | Tomato and tomato juice | Methanol 2,4-dinitrophenylhydrazine | Vortex | SPE cartridge | 1–20 µg kg−1 | 2–50 µg kg−1 | [91] |
| 4 Alternaria mycotoxins | 2016 | China | Wheat kernel | Acetonitrile–water–methanol (45:45:10, v/v/v) | Sonicating | SPE cartridge | 0.04–1.3 µg kg−1 | 0.1–4.2 | [92] |
| AFs, FB1, FB2, DON, OTA, ZEA | 2016 | Thailand | Brown rice | Acetonitrile with 10% (v/v) acetic acid | Vortex | QuEChERS | 1.4–25 µg kg−1 | 4.1–75 µg kg−1 | [93] |
| 15 mycotoxins | 2016 | Spain | Cow milk | Acetonitrile (2% formic acid) | Shaking | Sodium acetate | 0.02−10.14 ng mL−1 | Nd | [94] |
| 16 mycotoxins | 2017 | China | Vegetable oils | 85% Acetonitrile | Shaking | QuEChERS | 0.04–2.9 ng g−1 | Nd | [95] |
| 11 mycotoxins | 2017 | USA | Infant cereals | Acetonitrile/water/formic acid, (80:19.9:0.1, v/v/v) | Shaking | Nd | 0.01−10.0 ng g−1 | 0.05–50 ng g–1 | [84] |
| 12 Fusarium mycotoxins | 2017 | Germany | Beer | Acetonitrile/water (70:30, v/v) Acetonitrile/water (84:16, v/v) | Vortex | SPE cartridge | 0.05–6.9 µg L−1 | 0.15–20 µg L−1 | [87] |
| AFB1 OTA FB1 DON T2 HT-2 ZEA | 2017 | Italy | Cereal-based samples | Acetonitrile–water–acetic acid (79:20:1, v/v/v) | Shaking | Nd | 0.06–0.13 µg kg−1fo r AFB1 0.4–0.8 µg kg−1 for OTA 8−16 µg kg−1 for FB1 20 µg kg−1 for DON 4–8 µg kg−1 for T–2 20 µg kg−1 for HT–2 1.6–3.2 µg kg−1 for ZEA | Nd | [96] |
| 13 mycotoxins | 2017 | Korea | Cereal grains | Methanol 80%, containing 0.5% acetic acid | Shaking | IAC | 0.1−18.1 ng/g | 0.4–54.8 ng/g | [97] |
| 20 mycotoxins | 2019 | Korea | Soybean Paste | Methanol–water (60:40, v/v) and PBS | Blending | IAC | 0.06–4.68 µg kg−1 | 0.17−13.24 | [49] |
| 6 Alternaria toxins | 2019 | China | Grapes | Acetonitrile and dispersive solid phase extraction | Shaking | QuEChERS | 0.03–0.21 µg kg−1 | 0.09–0.48 µg kg−1 | [77] |
| AFs, ZEA, α-ZOL | 2019 | Spain | Vegetable oils | Acetonitrile | Shaking | QuEChERS | Nd | 0.5 μg kg−1 for AFs 1 μg kg−1 for ZEA and α-ZOL | [93] |
| Mycotoxin | Label Used | Commodity | Sensitivity | Reference |
|---|---|---|---|---|
| Deoxynivalenol (DON) Zearalenone (ZEA) T-2/H-T2-toxin | Epoxy-functionalized silica coated QDs | Barley | 1000 µg/kg 80 µg/kg 80 µg/kg | [106] |
| Aflatoxin B1 (AFB1) Zearalenone (ZEA) Deoxynivalenol (DON) | Monoclonal antibodies (mAbs) with the conjugates bovine serum albumin (BSA) | Wheat and maize | 0.05 μg/kg 1 μg/kg 3 μg/kg | [107] |
| Fumonisin B1 (FB1) Deoxynivalenol (DON) | Gold nanoparticles (AuNPs) | Maize | 2.0 ng mL−1 40 ng mL−1 | [108] |
| Deoxynivalenol (DON) T-2 toxin (T-2) Zearalenone (ZEN) | Amorphous carbon nanoparticles (ACNPs) | Maize | 20 µg/kg 13 µg/kg 1 µg/kg | [109] |
| Aflatoxin B1 (AFB1) Zearalenone (ZEN) Deoxynivalenol (DON) | CdSe/SiO2 quantum dot microbeads | Feedstuff | 10 pg mL−1 80 pg mL−1 500 pg mL−1 | [110] |
| Zearalenone (ZEN) | Antibody-labeled quantum dot sumicro beads | Corn | 3.6 mg mL−1 | [111] |
| Fumonisins (FUs) | CdSe/ZnS QD + GNP | Maize | 62.5 μg/kg | [102] |
| Mycotoxin | Recognition Element | Transducer/Technique | Food | Detection Limit | Reference |
|---|---|---|---|---|---|
| AFB1 | Organic framework composite | Piezoelectric (QCM) | Peanut, pistachio, rice, and wheat | 2.8 pg mL−1 | [116] |
| AFB1 | Antibody | Impedimetric (EIS) | Corn | 0.05 ng mL−1 | [117] |
| AFB1 | Antibody | Piezoelectric (EQCM) | Cereal | 8 pg mL−1 | [118] |
| AFB1 | Antibody | Piezoelectric (QCM) | Peanut | 0.83 ng kg−1 | [119] |
| AFB1 | Antibody | Potentiometric (DPV) | Corn powder | 3.5 pg mL−1 | [120] |
| Cyclopiazonic acid | Antibody | Optical (SPR) | Maize and cheese | 0.29 mg mL−1 | [121] |
| DON, ZEN, T-2toxin | Antibody | Optical (SPR) | Wheat | 15μg/kg−1 24 μg/kg−1 12 μg/kg−1 | [122] |
| HT-2 toxin, T-2 toxin, AFM1 | Antibody | Amperometric (CV) | Human urine | 0.4 ng mL−1 1 ng mL−1 0.3 ng mL−1 | [113] |
| T-2 toxin, T-2 toxin-3-glucoside (T2-G) | Antibody | Optical (iSPR) | Wheat | 1.2 ng mL−1 | [123] |
| OTA | Aptamer | Impedimetric (EIS) | Grape and commodities | 0.030 ng mL−1 | [124] |
| OTA | Aptamer | SPR | Wine and peanut oil | 0.005 ng mL−1 | [125] |
| OTA | Antibody | Piezoelectric (QCM) | Buffer | 17.2 ng mL−1 | [111] |
| OTA | Aptamer | Amperometric (CV) | Red wine | 0.23 pg mL−1 | [126] |
| OTA | Antibody | Piezoelectric (QCM) | Red wine | 0.16 ng mL−1 | [127] |
| OTA | Antibody | Optical (SPR) | Coffee | 3.8 ng mL−1 | [128] |
| OTA | Black phosphorene | Potentiometric (DPV) | Grape juice and red wine | 0.18 μg mL−1 | [129] |
| OTA | Antibody | Piezoelectric (QCM-D) | Red wine | 0.16 ng mL−1 | [127] |
| OTA, AFM1 | Antibody | Potentiometric (CV) | Red wine and milk | 0.15 ng mL−1 3.04 ng mL−1 | [130] |
| AFM1 | Antibody | Optical (SPR) | Milk | 18 pg mL−1 | [131] |
| PAT | Aptamer | Potentiometric (EIS/DPV) | Juice | 0.27 pg mL−1 | [132] |
| PAT | Aptamer | Impedimetric (EIS) | Apple juice | 2.8 ng L−1 | [133] |
| ZEN | Aptamer | Amperometric (CV/DPV) | Maize | 0.17 pg mL−1 | [134] |
| ZEN | Antibody | Amperometric (CV/DPV) | Corn and corn products | 1.5 pg mL−1 | [135] |
| ZEN | Aptamer | Potentiometric (CV/DPV) | Maize | 0.105 pg mL−1 | [136] |
| DON, T-2, ZEA, FB1 | Antibody | Optical (iSPR) | Barley | 64 µg kg−1, 26 µg kg−1, 96 µg kg−1, 13 µg kg−1 | [137] |
| Biosensors | Advantages | Limitations | Reference |
|---|---|---|---|
| Impedimetric | High sensitivity and selectivity, time-efficient, simple operation, fast response, mobility due to portable instrumentation, miniaturization | Complex construction, expensive labeling markers | [25,124,147] |
| Potentiometric | Reduced analysis time, mobility due to portable instrumentation, miniaturization, high sensitivity and selectivity, use without sample treatment | The sensitivity and lifetime are seriously influenced by factors such as temperature, pH, immobilization support, and immunological cross-reaction | [129,147,148] |
| Amperometric | Mobility due to portable instrumentation, miniaturization, high sensitivity and selectivity | Regeneration between measurements | [147] |
| Surface plasmon resonance | High specificity and sensitivity, small size and cost-efficiency, direct, real-time analysis and detection without label, development of portable devices | The broad practical application is still under development | [25,144] |
| Quartz crystal microbalance | Low cost with high sensitivity, selectivity, and possibility of reuse, real-time output, and label- or radiation-free entities, development of portable devices | Requirement of a relatively high background signal relative to the signal-on assay formation | [119,149] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in Analysis and Detection of Major Mycotoxins in Foods. Foods 2020, 9, 518. https://doi.org/10.3390/foods9040518
Agriopoulou S, Stamatelopoulou E, Varzakas T. Advances in Analysis and Detection of Major Mycotoxins in Foods. Foods. 2020; 9(4):518. https://doi.org/10.3390/foods9040518
Chicago/Turabian StyleAgriopoulou, Sofia, Eygenia Stamatelopoulou, and Theodoros Varzakas. 2020. "Advances in Analysis and Detection of Major Mycotoxins in Foods" Foods 9, no. 4: 518. https://doi.org/10.3390/foods9040518