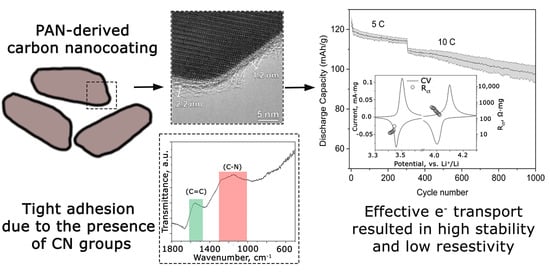
Polyacrylonitrile-Derived Carbon Nanocoating for Long-Life High-Power Phosphate Electrodes
Abstract
:1. Introduction
2. Materials and Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yang, L.; Chen, J.; Chen, L.; Yang, P.; Zhang, J.; Li, A.; Wang, Y.; Wang, Y.; Wang, R. LiFePO4 Nanoplates with {010} Exposed Active Planes Prepared by Hydrothermal Method. J. Mater. Sci. Mater. Electron. 2016, 27, 12258–12263. [Google Scholar] [CrossRef]
- Nazarov, E.E.; Dembitskiy, A.D.; Trussov, I.A.; Tyablikov, O.A.; Glazkova, I.S.; Sobolev, A.V.; Presmiakov, I.A.; Mikheev, I.A.; Morozov, A.V.; Nikitina, V.A.; et al. A Li-Rich Strategy towards Advanced Mn-Doped Triphylite Cathodes for Li-Ion Batteries. RCS Energy Adv. 2023. [Google Scholar] [CrossRef]
- Meng, Y.; Wang, Y.; Zhang, Z.; Chen, X.; Guo, Y.; **ao, D. A Phytic Acid Derived LiMn0.5Fe0.5PO4/Carbon Composite of High Energy Density for Lithium Rechargeable Batteries. Sci. Rep. 2019, 9, 6665. [Google Scholar] [CrossRef]
- Hong, L.; Yang, K.; Tang, M. A Mechanism of Defect-Enhanced Phase Transformation Kinetics in Lithium Iron Phosphate Olivine. Npj Comput. Mater. 2019, 5, 118. [Google Scholar] [CrossRef]
- Hong, L.; Li, L.; Chen-Wiegart, Y.-K.; Wang, J.; **ang, K.; Gan, L.; Li, W.; Meng, F.; Wang, F.; Wang, J.; et al. Two-Dimensional Lithium Diffusion Behavior and Probable Hybrid Phase Transformation Kinetics in Olivine Lithium Iron Phosphate. Nat. Commun. 2017, 8, 1194. [Google Scholar] [CrossRef] [PubMed]
- Park, K.-Y.; Park, I.; Kim, H.; Yoon, G.; Gwon, H.; Cho, Y.; Soo Yun, Y.; Kim, J.J.; Lee, S.; Ahn, D.; et al. Lithium-Excess Olivine Electrode for Lithium Rechargeable Batteries. Energy Environ. Sci. 2016, 9, 2902–2915. [Google Scholar] [CrossRef]
- Drozhzhin, O.A.; Sobolev, A.V.; Sumanov, V.D.; Glazkova, I.S.; Aksyonov, D.A.; Grebenshchikova, A.D.; Tyablikov, O.A.; Alekseeva, A.M.; Mikheev, I.V.; Dovgaliuk, I.; et al. Exploring the Origin of the Superior Electrochemical Performance of Hydrothermally Prepared Li-Rich Lithium Iron Phosphate Li1+δFe1−δPO4. J. Phys. Chem. C 2020, 124, 126–134. [Google Scholar] [CrossRef]
- Zhou, G.; Sun, X.; Li, Q.-H.; Wang, X.; Zhang, J.N.; Yang, W.; Yu, X.; **ao, R.; Li, H. Mn Ion Dissolution Mechanism for Lithium-Ion Battery with LiMn2O4 Cathode: In Situ Ultraviolet–Visible Spectroscopy and Ab Initio Molecular Dynamics Simulations. J. Phys. Chem. Lett. 2020, 11, 3051–3057. [Google Scholar] [CrossRef]
- Hanf, L.; Henschel, J.; Diehl, M.; Winter, M.; Nowak, S. Mn2+ or Mn3+? Investigating Transition Metal Dissolution of Manganese Species in Lithium Ion Battery Electrolytes by Capillary Electrophoresis. Electrophoresis 2020, 41, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Chi, Z.X.; Zhang, W.; Wang, X.S.; Cheng, F.Q.; Chen, J.T.; Cao, A.-M.; Wan, L.J. Accurate Surface Control of Core–Shell Structured LiMn0.5Fe0.5PO4@C for Improved Battery Performance. J. Mater. Chem. A 2014, 2, 17359–17365. [Google Scholar] [CrossRef]
- Yaroslavtsev, A.B.; Stenina, I.A. Carbon Coating of Electrode Materials for Lithium-Ion Batteries. Surf. Innov. 2021, 9, 92–110. [Google Scholar] [CrossRef]
- Wang, J.; Gu, Y.J.; Kong, W.L.; Liu, H.Q.; Chen, Y.B.; Liu, W. Effect of Carbon Coating on the Crystal Orientation and Electrochemical Performance of Nanocrystalline LiFePO4. Solid State Ion. 2018, 327, 11–17. [Google Scholar] [CrossRef]
- Qi, M.; Liu, Y.; Xu, M.; Feng, M.; Gu, J.; Liu, Y.; Wang, L. Improved Electrochemical Performances of Carbon-Coated LiFePO4 Microspheres for Li-Ion Battery Cathode. Mater. Res. Express 2019, 6, 115520. [Google Scholar] [CrossRef]
- Luchinin, N.D.; Aksyonov, D.A.; Morozov, A.V.; Ryazantsev, S.V.; Nikitina, V.A.; Abakumov, A.M.; Antipov, E.V.; Fedotov, S.S. α-TiPO4 as a Negative Electrode Material for Lithium-Ion Batteries. Inorg. Chem. 2021, 60, 12237–12246. [Google Scholar] [CrossRef]
- Fedotov, S.S.; Luchinin, N.D.; Aksyonov, D.A.; Morozov, A.V.; Ryazantsev, S.V.; Gaboardi, M.; Plaisier, J.R.; Stevenson, K.J.; Abakumov, A.M.; Antipov, E.V. Titanium-Based Potassium-Ion Battery Positive Electrode with Extraordinarily High Redox Potential. Nat. Commun. 2020, 11, 1484. [Google Scholar] [CrossRef]
- Iarchuk, A.R.; Nikitina, V.A.; Karpushkin, E.A.; Sergeyev, V.G.; Antipov, E.V.; Stevenson, K.J.; Abakumov, A.M. Influence of Carbon Coating on Intercalation Kinetics and Transport Properties of LiFePO4. ChemElectroChem 2019, 6, 5090–5100. Available online: https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/celc.201901219 (accessed on 20 June 2022). [CrossRef]
- Lan, Y.; Wang, X.; Zhang, J.; Zhang, J.; Wu, Z.; Zhang, Z. Preparation and Characterization of Carbon-Coated LiFePO4 Cathode Materials for Lithium-Ion Batteries with Resorcinol–Formaldehyde Polymer as Carbon Precursor. Powder Technol. 2011, 212, 327–331. [Google Scholar] [CrossRef]
- Stenina, I.; Minakova, P.; Kulova, T.; Yaroslavtsev, A. Electrochemical Properties of LiFePO4 Cathodes: The Effect of Carbon Additives. Batteries 2022, 8, 111. [Google Scholar] [CrossRef]
- Qiao, M.; Kong, H.; Ding, X.; Hu, Z.; Zhang, L.; Cao, Y.; Yu, M. Study on the Changes of Structures and Properties of PAN Fibers during the Cyclic Reaction in Supercritical Carbon Dioxide. Polymers 2019, 11, 402. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Guo, X.; Wang, J.; Yuan, Y.; Sun, Q.; Tian, R.; Yang, H.; Lu, J. Restraining the Octahedron Collapse in Lithium and Manganese Rich NCM Cathode toward Suppressing Structure Transformation. Adv. Energy Mater. 2022, 12, 2201323. Available online: https://onlinelibrary.wiley.com/doi/10.1002/aenm.202201323 (accessed on 20 November 2022). [CrossRef]
- Yang, X.; Shen, L.; Wu, B.; Zuo, Z.; Mu, D.; Wu, B.; Zhou, H. Improvement of the Cycling Performance of LiCoO2 with Assistance of Cross-Linked PAN for Lithium Ion Batteries. J. Alloys Compd. 2015, 639, 458–464. [Google Scholar] [CrossRef]
- Shraer, S.D.; Luchinin, N.D.; Trussov, I.A.; Aksyonov, D.A.; Morozov, A.V.; Ryazantsev, S.V.; Iarchuk, A.R.; Morozova, P.A.; Nikitina, V.A.; Stevenson, K.J.; et al. Development of Vanadium-Based Polyanion Positive Electrode Active Materials for High-Voltage Sodium-Based Batteries. Nat. Commun. 2022, 13, 4097. [Google Scholar] [CrossRef]
- Han, Q.; Wang, F.; Wang, Z.; Yi, Z.; Na, Z.; Wang, X.; Wang, L. PAN-Based Carbon Fiber@SnO2 for Highly Reversible Structural Lithium-Ion Battery Anode. Ionics 2018, 24, 1049–1055. [Google Scholar] [CrossRef]
- Zeng, T.; Feng, D. Conductive Carbon Networks in Surface Coating of GeP Rods toward High-Performance Lithium/Sodium-Ion Battery Anode. Surf. Interfaces 2021, 27, 101461. [Google Scholar] [CrossRef]
- Adegbola, T.A.; Agboola, O.; Fayomi, O.S.I. Review of Polyacrylonitrile Blends and Application in Manufacturing Technology: Recycling and Environmental Impact. Results Eng. 2020, 7, 100144. [Google Scholar] [CrossRef]
- Zhao, R.; Liang, Z.; Zou, R.; Xu, Q. Metal-Organic Frameworks for Batteries. Joule 2018, 2, 2235–2259. [Google Scholar] [CrossRef]
- Xu, X.; Qi, C.; Dong, Z.; Wang, H.; Jiu, J.; Liu, J.; Yan, H.; Suganuma, K. The Surface Coating of Commercial LiFePO4 by Utilizing ZIF-8 for High Electrochemical Performance Lithium Ion Battery. Nano-Micro Lett. 2018, 10, 1. Available online: https://link.springer.com/article/10.1007/s40820-017-0154-4 (accessed on 10 January 2023). [CrossRef]
- Lin, J.; Sun, Y.-H.; Lin, X. Metal-Organic Framework-Derived LiFePO4 Cathode Encapsulated in O,F-Codoped Carbon Matrix towards Superior Lithium Storage. Nano Energy 2022, 91, 106655. [Google Scholar] [CrossRef]
- Caskey, S.R.; Wong-Foy, A.G.; Matzger, A.J. Dramatic Tuning of Carbon Dioxide Uptake via Metal Substitution in a Coordination Polymer with Cylindrical Pores. J. Am. Chem. Soc. 2008, 130, 10870–10871. [Google Scholar] [CrossRef]
- Toby, B.; Von Dreele, R.B. GSAS-II: The Genesis of a Modern Open-Source All-Purpose Crystallography Software Package. J. Appl. Crystallogr. 2013, 46, 544–549. Available online: https://www.researchgate.net/publication/245022847_GSAS-II_The_Genesis_of_a_Modern_Open-Source_All-Purpose_Crystallography_Software_Package (accessed on 9 June 2022). [CrossRef]
- Harrington, D. Multiple Electrochemical Impedance Spectra Parameterization (MEISP+). Version 2.0 Kumho Petrochemical Co. Ltd., Kumho Chemical Laboratories, P.O. Box 64, Yuseong, Taejeon, 305-600, Korea. Fax: 82 42 862 5651. Http://Powergraphy.Com. Contact Kumho for Price. J. Am. Chem. Soc. 2002, 124, 1554–1555. [Google Scholar] [CrossRef]
- A Comprehensive Study of Effects of Carbon Coating on Li4Ti5O12 Anode Material for Lithium-Ion Batteries-IOPscience. Available online: https://iopscience.iop.org/article/10.1149/1.3519070 (accessed on 10 January 2023).
- Szymanski, H.A.; Erickson, R.E. Infrared Band Handbook; Springer: Boston, MA, USA, 1970; ISBN 978-1-4684-6071-1. [Google Scholar]
- Wang, K.; Hou, M.; Yuan, S.; Yu, H.; Wang, Y.; Wang, C.; **a, Y. An Additional Discharge Plateau of Mn3+ in LiFe0.5Mn0.5PO4 at High Current Rates. Electrochem. Commun. 2015, 55, 6–9. [Google Scholar] [CrossRef]
- Nikitina, V.A. Charge Transfer Processes in the Course of Metal-Ion Electrochemical Intercalation. Curr. Opin. Electrochem. 2020, 19, 71–77. [Google Scholar] [CrossRef]
- Yonemura, M.; Yamada, A.; Takei, Y.; Sonoyama, N.; Kanno, R. Comparative Kinetic Study of Olivine LixMPO4 (M = Fe, Mn). J. Electrochem. Soc. 2004, 151, A1352. [Google Scholar] [CrossRef]
- Nai, J.; Zhao, X.; Yuan, H.; Tao, X.; Guo, L. Amorphous Carbon-Based Materials as Platform for Advanced High-Performance Anodes in Lithium Secondary Batteries. Nano Res. 2021, 14, 2053–2066. Available online: https://link.springer.com/article/10.1007/s12274-021-3506-9 (accessed on 12 December 2022). [CrossRef]







| Space Group | Pnma |
|---|---|
| a, Å | 10.3847(4) |
| b, Å | 6.0502(6) |
| c, Å | 4.7196(7) |
| V, Å3 | 296.538(5) |
| Z | 4 |
| GOF | 0.11 |
| Rexp, % | 0.94 |
| Sample | Mass Change, % | Carbon Content, % Experimental | Carbon Content, % Expected | Graphitization Degree |
|---|---|---|---|---|
| Li-rich LFMP pristine | +2.36 | - | - | - |
| PAN-2 | −0.3 | 2.4 | 2 | 1.4 |
| PAN-4 | −1.94 | 4.2 | 4 | 1.3 |
| PAN-8 | −5.8 | 7.9 | 8 | 1.2 |
| Glu | −1.37 | 3.8 | 4 | 0.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nazarov, E.E.; Tyablikov, O.A.; Nikitina, V.A.; Antipov, E.V.; Fedotov, S.S. Polyacrylonitrile-Derived Carbon Nanocoating for Long-Life High-Power Phosphate Electrodes. Appl. Nano 2023, 4, 25-37. https://doi.org/10.3390/applnano4010002
Nazarov EE, Tyablikov OA, Nikitina VA, Antipov EV, Fedotov SS. Polyacrylonitrile-Derived Carbon Nanocoating for Long-Life High-Power Phosphate Electrodes. Applied Nano. 2023; 4(1):25-37. https://doi.org/10.3390/applnano4010002
Chicago/Turabian StyleNazarov, Eugene E., Oleg A. Tyablikov, Victoria A. Nikitina, Evgeny V. Antipov, and Stanislav S. Fedotov. 2023. "Polyacrylonitrile-Derived Carbon Nanocoating for Long-Life High-Power Phosphate Electrodes" Applied Nano 4, no. 1: 25-37. https://doi.org/10.3390/applnano4010002