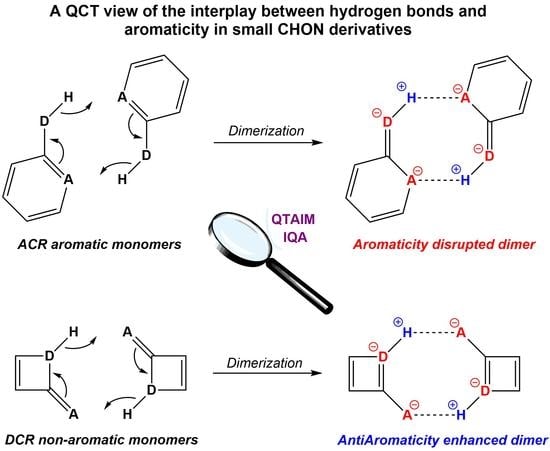
A QCT View of the Interplay between Hydrogen Bonds and Aromaticity in Small CHON Derivatives
Abstract
:1. Introduction
2. Theoretical Framework
2.1. Real Space Wavefunction Analyses
2.2. Aromaticity
3. Computational Details
4. Results and Discussion
4.1. General Energetic Changes Induced by HB Formation
4.2. Quantum Chemical Topology Analyses
4.3. Perturbation of the Aromaticity of the π Skeleton
4.4. The Peculiar Case of the AZH (DCR) Dimer
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Moore, T.S.; Winmill, T.F. CLXXVII—The state of amines in aqueous solution. J. Chem. Soc. Trans. 1912, 101, 1635–1676. [Google Scholar] [CrossRef]
- Luo, X.Y.; Fan, X.; Shi, G.L.; Li, H.R.; Wang, C.M. Decreasing the Viscosity in CO2 Capture by Amino-Functionalized Ionic Liquids through the Formation of Intramolecular Hydrogen Bond. J. Phys. Chem. B 2016, 120, 2807–2813. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, D.; Cantu, D.C.; Koech, P.K.; Heldebrant, D.J.; Karkamkar, A.; Zheng, F.; Bearden, M.D.; Rousseau, R.; Glezakou, V.A. Directed Hydrogen Bond Placement: Low Viscosity Amine Solvents for CO2 Capture. ACS Sustain. Chem. Eng. 2019, 7, 7535–7542. [Google Scholar] [CrossRef]
- Altamash, T.; Amhamed, A.; Aparicio, S.; Atilhan, M. Effect of Hydrogen Bond Donors and Acceptors on CO2 Absorption by Deep Eutectic Solvents. Processes 2020, 8, 1533. [Google Scholar] [CrossRef]
- Saeed, U.; Khan, A.L.; Gilani, M.A.; Bilad, M.R.; Khan, A.U. Supported liquid membranes comprising of choline chloride based deep eutectic solvents for CO2 capture: Influence of organic acids as hydrogen bond donor. J. Mol. Liq. 2021, 335, 116155. [Google Scholar] [CrossRef]
- Cao, L.; Li, D.; Soto, F.A.; Ponce, V.; Zhang, B.; Ma, L.; Deng, T.; Seminario, J.M.; Hu, E.; Yang, X.Q.; et al. Highly Reversible Aqueous Zinc Batteries enabled by Zincophilic–Zincophobic Interfacial Layers and Interrupted Hydrogen-Bond Electrolytes. Angew. Chem. Int. Ed. 2021, 60, 18845–18851. [Google Scholar] [CrossRef]
- Sun, T.; Zheng, S.; Nian, Q.; Tao, Z. Hydrogen Bond Shielding Effect for High-Performance Aqueous Zinc Ion Batteries. Small 2022, 18, 2107115. [Google Scholar] [CrossRef]
- ** function in dispersion corrected density functional theory. J. Comput. Chem. 2011, 32, 1456–1465. [Google Scholar] [CrossRef]
- Keith, A.T. TK Gristmill Software; AIMALL (Version 19.02.13); AIMALL: Overland Park, KS, USA, 2019. [Google Scholar]
- Martín Pendás, A.; Francisco, E. Promolden. A QTAIM/IQA code (Avaliable from the authors upon request).
- Maxwell, P.; Martín Pendás, Á.; Popelier, P.L.A. Extension of the interacting quantum atoms (IQA) approach to B3LYP level density functional theory (DFT). Phys. Chem. Chem. Phys. 2016, 18, 20986–21000. [Google Scholar] [CrossRef] [PubMed]
- Matito, E. ESI-3D: Electron Sharing Indexes Program for 3D Molecular Space Partitioning. 2020. [Google Scholar]
- Feixas, F.; Matito, E.; Solà, M.; Poater, J. Patterns of π-electron delocalization in aromatic and antiaromatic organic compounds in the light of Hückel’s 4n + 2 rule. Phys. Chem. Chem. Phys. 2010, 12, 7126–7137. [Google Scholar] [CrossRef] [PubMed] [Green Version]









| System | System | ||||
|---|---|---|---|---|---|
| 2HP (ACR) | 5.06 | 3.01 | 2HP (DCR) | −7.11 | −7.03 |
| NCO (ACR) | 0.00 | 0.00 | NCO (DCR) | 0.00 | 0.00 |
| AZH (ACR) | −2.82 | −3.34 | AZH (DCR) | 6.40 | 5.38 |
| 2AP (ACR) | 3.22 | 2.58 | 2AP (DCR) | −8.16 | −7.50 |
| NCN (ACR) | 0.00 | 0.00 | NCN (DCR) | 0.00 | 0.00 |
| AZA (ACR) | −10.05 | −9.58 | AZA (DCR) | 2.13 | 0.89 |
| ACR | ||||
| System | (D) | (H) | (A) | (C) |
| 2HP | −0.087 | +0.049 | −0.067 | +0.069 |
| NCO | −0.085 | +0.041 | −0.072 | +0.085 |
| AZH | −0.089 | +0.065 | −0.077 | +0.045 |
| 2AP | −0.040 | +0.070 | −0.021 | +0.019 |
| NCN | −0.051 | +0.088 | −0.024 | +0.008 |
| AZA | −0.061 | +0.113 | −0.063 | −0.002 |
| DCR | ||||
| System | (D) | (H) | (A) | (C) |
| 2HP | −0.070 | +0.105 | −0.024 | −0.040 |
| NCO | −0.052 | +0.085 | −0.025 | −0.007 |
| AZH | −0.037 | +0.064 | −0.024 | −0.011 |
| 2AP | −0.063 | +0.101 | −0.031 | −0.012 |
| NCN | −0.051 | +0.088 | −0.024 | +0.008 |
| AZA | −0.030 | +0.074 | −0.034 | −0.007 |
| ACR | ||||
| System | DI(D–H) | DI(D–C) | DI(C–A) | DI(H–A) |
| 2HP | −0.207 | +0.056 | −0.064 | +0.152 |
| NCO | −0.246 | +0.100 | −0.171 | +0.197 |
| AZH | −0.216 | +0.079 | −0.136 | +0.134 |
| 2AP | −0.151 | +0.051 | −0.034 | +0.103 |
| NCN | −0.181 | +0.073 | −0.076 | +0.117 |
| AZA | −0.262 | +0.131 | −0.167 | +0.165 |
| DCR | ||||
| System | DI(D–H) | DI(D–C) | DI(C–A) | DI(H–A) |
| 2HP | −0.209 | +0.065 | −0.091 | +0.132 |
| NCO | −0.162 | +0.069 | −0.070 | +0.099 |
| AZH | −0.103 | +0.034 | −0.043 | +0.066 |
| 2AP | −0.234 | +0.067 | −0.106 | +0.163 |
| NCN | −0.181 | +0.073 | −0.076 | +0.117 |
| AZA | −0.152 | +0.043 | −0.053 | +0.099 |
| System | MCI (a.u.) | FLU (a.u.) | System | MCI (a.u.) | FLU (a.u.) | ||
|---|---|---|---|---|---|---|---|
| 2HP (ACR) | −0.008 | +0.001 | − | 2HP (DCR) | +0.007 | −0.010 | + |
| AZH (ACR) | +0.001 | −0.017 | + | AZH (DCR) | −0.001 | −0.005 | − |
| 2AP (ACR) | −0.004 | +0.001 | − | 2AP (DCR) | +0.007 | −0.010 | + |
| AZA (ACR) | +0.004 | −0.021 | + | AZA (DCR) | −0.001 | −0.007 | − |
| Isomer | (kcal/mol) | FLU (a.u.) | MCI (a.u.) |
|---|---|---|---|
| bent–trans | +0.48 | +0.0006 | −0.0001 |
| bent–cis | 0.00 | 0.0000 | 0.0000 |
| planar | +7.38 | +0.0094 | −0.0038 |
| QTAIM charges | ||||
| System | Q(D) | (H) | (A) | (C) |
| bent–trans | −0.039 | +0.063 | −0.024 | −0.007 |
| bent–cis | −0.037 | +0.064 | −0.024 | −0.011 |
| planar | −0.185 | +0.127 | −0.030 | +0.065 |
| Delocalisation indices | ||||
| System | DI(D–H) | DI(D–C) | DI(C–A) | DI(H–A) |
| bent–trans | −0.099 | +0.035 | −0.043 | +0.064 |
| bent–cis | −0.103 | +0.034 | −0.043 | +0.066 |
| planar | −0.169 | +0.070 | −0.068 | +0.074 |
| IQA energy partition | ||||
| System | ||||
| bent–trans | −12.80 | −21.21 | +11.99 | −87.41 |
| bent–cis | −12.87 | −18.43 | +13.47 | −88.66 |
| planar | −40.98 | −121.17 | −1.07 | −106.99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gallegos, M.; Barrena-Espés, D.; Guevara-Vela, J.M.; Rocha-Rinza, T.; Pendás, Á.M. A QCT View of the Interplay between Hydrogen Bonds and Aromaticity in Small CHON Derivatives. Molecules 2022, 27, 6039. https://doi.org/10.3390/molecules27186039
Gallegos M, Barrena-Espés D, Guevara-Vela JM, Rocha-Rinza T, Pendás ÁM. A QCT View of the Interplay between Hydrogen Bonds and Aromaticity in Small CHON Derivatives. Molecules. 2022; 27(18):6039. https://doi.org/10.3390/molecules27186039
Chicago/Turabian StyleGallegos, Miguel, Daniel Barrena-Espés, José Manuel Guevara-Vela, Tomás Rocha-Rinza, and Ángel Martín Pendás. 2022. "A QCT View of the Interplay between Hydrogen Bonds and Aromaticity in Small CHON Derivatives" Molecules 27, no. 18: 6039. https://doi.org/10.3390/molecules27186039