Journal Description
Antibodies
Antibodies
is an international, peer-reviewed, open access journal on immunoglobulins, published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: CiteScore - Q2 (Drug Discovery)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 23 days after submission; acceptance to publication is undertaken in 6.6 days (median values for papers published in this journal in the first half of 2024).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
3.0 (2023);
5-Year Impact Factor:
4.7 (2023)
Latest Articles
Characterization of the Charge Heterogeneity of a Monoclonal Antibody That Binds to Both Cation Exchange and Anion Exchange Columns under the Same Binding Conditions
Antibodies 2024, 13(3), 52; https://doi.org/10.3390/antib13030052 (registering DOI) - 30 Jun 2024
Abstract
►
Show Figures
Therapeutic antibodies play an important role in the public healthcare system to treat patients with a variety of diseases. Protein characterization using an array of analytical tools provides in-depth information for drug quality, safety, efficacy, and the further understanding of the molecule. A
[...] Read more.
Therapeutic antibodies play an important role in the public healthcare system to treat patients with a variety of diseases. Protein characterization using an array of analytical tools provides in-depth information for drug quality, safety, efficacy, and the further understanding of the molecule. A therapeutic antibody candidate MAB1 exhibits unique binding properties to both cation and anion exchange columns at neutral pH. This uniqueness disrupts standard purification processes and necessitates adjustments in manufacturing. This study identifies that the charge heterogeneity of MAB1 is primarily due to the N-terminal cyclization of glutamine to pyroglutamine and, to a lesser extent, succinimide intermediate, deamidation, and C-terminal lysine. Using three approaches, i.e., deferential chemical labeling, H/D exchange, and molecular modeling, the binding to anion exchange resins is attributed to negatively charged patches on the antibody’s surface, involving specific carboxylic acid residues. The methodologies shown here can be extended to study protein binding orientation in column chromatography.
Full article
Open AccessArticle
177Lu Anti-Angiogenic Radioimmunotherapy Targeting ATP Synthase in Gastric Cancer Model
by
Bok-Nam Park, Young-Sil An, Su-Min Kim, Su-** Lee, Yong-** Park and Joon-Kee Yoon
Antibodies 2024, 13(3), 51; https://doi.org/10.3390/antib13030051 - 27 Jun 2024
Abstract
This study investigated a novel radioimmunotherapy strategy for targeting tumor angiogenesis. We developed a radiopharmaceutical complex by labeling an anti-adenosine triphosphate synthase (ATPS) monoclonal antibody (mAb) with the radioisotope 177Lu using DOTA as a chelating agent. 177Lu-DOTA-ATPS mAb demonstrated high labeling
[...] Read more.
This study investigated a novel radioimmunotherapy strategy for targeting tumor angiogenesis. We developed a radiopharmaceutical complex by labeling an anti-adenosine triphosphate synthase (ATPS) monoclonal antibody (mAb) with the radioisotope 177Lu using DOTA as a chelating agent. 177Lu-DOTA-ATPS mAb demonstrated high labeling efficiency (99.0%) and stability in serum. MKN-45 cancer cells exhibited the highest cellular uptake, which could be specifically blocked by unlabeled ATPS mAb. In mice, 177Lu-DOTA-ATPS mAb accumulated significantly in tumors, with a tumor uptake of 16.0 ± 1.5%ID/g on day 7. 177Lu-DOTA-ATPS mAb treatment significantly reduced the viability of MKN-45 cells in a dose-dependent manner. In a xenograft tumor model, this radioimmunotherapy strategy led to substantial tumor growth inhibition (82.8%). Furthermore, combining 177Lu-DOTA-ATPS mAb with sunitinib, an anti-angiogenic drug, enhanced the therapeutic efficacy of sunitinib in the mouse model. Our study successfully developed 177Lu-DOTA-ATPS mAb, a radioimmunotherapy agent targeting tumor blood vessels. This approach demonstrates significant promise for inhibiting tumor growth, both as a single therapy and in combination with other anti-cancer drugs.
Full article
(This article belongs to the Special Issue Emerging Antibody Engineering Strategies and Applications for Immunotherapy of Cancer)
►▼
Show Figures

Figure 1
Open AccessArticle
Development, Optimization and Evaluation of a Sensitive Enzyme-Linked Immunosorbent Assay (ELISA) Prototype for Detection of Chicken-Based IgY Polyclonal Antibodies against Toxins of D. polylepis Venom
by
Stephen Wilson Kpordze, Gideon Mutie Kikuvi, James Hungo Kimotho and Victor Atunga Mobegi
Antibodies 2024, 13(3), 50; https://doi.org/10.3390/antib13030050 - 21 Jun 2024
Abstract
Life-threatening medical issues can result from snakebite, and hence this is a public health concern. In many tropical and subtropical nations such as Kenya, where a wide variety of poisonous snakes are prevalent, diagnosis of snakebite in health facilities is imperative. Different antivenoms
[...] Read more.
Life-threatening medical issues can result from snakebite, and hence this is a public health concern. In many tropical and subtropical nations such as Kenya, where a wide variety of poisonous snakes are prevalent, diagnosis of snakebite in health facilities is imperative. Different antivenoms are needed to treat the venom of different snake species. Nonetheless, it might be difficult for medical professionals to identify the exact snake species that envenomated a patient due to the similarities of several snake envenomations’ clinical symptoms. Therefore, the necessity for an assay or technique for identifying venomous species is critical. The current study sought to develop a sensitive ELISA prototype for the detection of D. polylepis venom in Kenya using generated chicken-based IgY polyclonal antibodies. Serum samples containing specific chicken-based IgY antibodies previously raised against D. polylepis venom toxins were used in the assay development. ELISA parameters were optimized, and the developed assay was assessed for applicability. The limit of detection (LoD) of the ELISA for neurotoxic venoms was determined to be 0.01 µg/mL. Successful discrimination between neurotoxic and cytotoxic venoms was achieved by the ensuing inhibition ELISA assay. The developed assay showed the capability of identifying venoms in blood samples (from spiked and venom-challenged blood samples) of BALB/c mice, providing compelling evidence of the strategy’s usefulness. This assay could help physicians diagnose and manage victims of snakebites through the evaluation of clinical samples.
Full article
(This article belongs to the Section Antibody-Based Diagnostics)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Screening for TORCH Antibodies in Croatian Childbearing-Aged Women, 2014–2023
by
Tatjana Vilibic-Cavlek, Branko Kolaric, Marko Belamaric, Mario Sviben, Thomas Ferenc, Dan Navolan, Viktor Bekic, Ljiljana Milasincic, Ljiljana Antolasic, Maja Vilibic, Mateja Vujica Ferenc, Ema Reicher, Tadej Jezek, Ioana Ciohat, Raluca Catalina Parvanescu, Matea Kos and Maja Bogdanic
Antibodies 2024, 13(2), 49; https://doi.org/10.3390/antib13020049 - 18 Jun 2024
Abstract
►▼
Show Figures
TORCH infections usually result in mild maternal morbidity, but may cause severe congenital abnormalities. Therefore, it is important to detect maternal infections, monitor the fetus after the disease has been recognized, and define the seronegative women who are at risk of primary infection
[...] Read more.
TORCH infections usually result in mild maternal morbidity, but may cause severe congenital abnormalities. Therefore, it is important to detect maternal infections, monitor the fetus after the disease has been recognized, and define the seronegative women who are at risk of primary infection during pregnancy. From 2014 to 2023, serum samples from 1032 childbearing-aged and pregnant women (16–45 years) were tested for IgM/IgG antibodies to the most common TORCH pathogens: Toxoplasma gondii, rubella virus (RUBV), cytomegalovirus (CMV), and herpes simplex viruses (HSV-1 and HSV-2). The overall IgG seroprevalence rates were 20.1% for T. gondii, 91.3% for RUBV, 70.5% for CMV, 66.8% for HSV-1, and 3.5% for HSV-2. Only HSV-2 seroprevalence was age-related, with a significant progressive increase in seropositivity from 0% in those aged less than 26 years to 9.3% in those older than 40 years. The seroprevalence of T. gondii was higher in residents of suburban/rural areas than in residents of urban areas (27.4% vs. 17.1%). In addition, participants from continental regions were more often toxoplasma-seropositive than those from coastal regions (22.2% vs. 15.3%). HSV-1 seroprevalence was also higher in suburban/rural areas (71.7% vs. 64.7%). Obstetric history was not associated with TORCH seropositivity. Univariate and multivariate risk analysis showed that suburban/rural areas of residence and continental geographic regions were significant risk factors for T. gondii seroprevalence. Furthermore, suburban/rural area of residence was a significant risk factor for HSV-1 seroprevalence, while older age was a significant risk factor for HSV-2 seroprevalence. A declining trend in the seroprevalence of all TORCH pathogens was observed compared to previous Croatian studies (2005–2011). Similarly, the proportion of women simultaneously IgG-seropositive to two or three pathogens decreased over time. The maternal serology before pregnancy could potentially reduce the burden of congenital TORCH infections.
Full article

Figure 1
Open AccessArticle
Adoptive Cell Therapy in Mice Sensitized to a Grass Pollen Allergen
by
Anna Marianne Weijler, Lisa Prickler, Verena Kainz, Eva Bergmann, Barbara Bohle, Heinz Regele, Rudolf Valenta, Birgit Linhart and Thomas Wekerle
Antibodies 2024, 13(2), 48; https://doi.org/10.3390/antib13020048 - 18 Jun 2024
Abstract
The proportion of patients with type I allergy in the world population has been increasing and with it the number of people suffering from allergic symptoms. Recently we showed that prophylactic cell therapy employing allergen-expressing bone marrow (BM) cells or splenic B cells
[...] Read more.
The proportion of patients with type I allergy in the world population has been increasing and with it the number of people suffering from allergic symptoms. Recently we showed that prophylactic cell therapy employing allergen-expressing bone marrow (BM) cells or splenic B cells induced allergen-specific tolerance in naïve mice. Here we investigated if cell therapy can modulate an established secondary allergen-specific immune response in pre-immunized mice. We sensitized mice against the grass pollen allergen Phl p 5 and an unrelated control allergen, Bet v 1, from birch pollen before the transfer of Phl p 5-expressing BM cells. Mice were conditioned with several combinations of low-dose irradiation, costimulation blockade, rapamycin and T cell-depleting anti-thymocyte globulin (ATG). Levels of allergen-specific IgE and IgG1 in serum after cell transfer were measured via ELISA and alterations in cellular responses were measured via an in vitro proliferation assay and transplantation of Phl p 5+ skin grafts. None of the tested treatment protocols impacted Phl p 5-specific antibody levels. Transient low-level chimerism of Phl p 5+ leukocytes as well as a markedly prolonged skin graft survival were observed in mice conditioned with high numbers of Phl p 5+ BMC or no sensitization events between the day of cell therapy and skin grafting. The data presented herein demonstrate that a pre-existing secondary allergen-specific immune response poses a substantial hurdle opposing tolerization through cell therapy and underscore the importance of prophylactic approaches for the prevention of IgE-mediated allergy.
Full article
(This article belongs to the Section Humoral Immunity)
►▼
Show Figures

Figure 1
Open AccessArticle
Elevated Immunoglobulin E Serum Levels: Possible Underlying Factors That Can Cause an Inborn Error of Immunity in the Pediatric Population with Recurrent Infections
by
Sînziana Oprițescu, Gabriela Viorela Nițescu, Daniela Cîrnațu, Svetlana Trifunschi, Melania Munteanu, Mihaela Golumbeanu, Dora Boghițoiu, Adriana Maria Dărăban, Elena Iuliana Ilie and Elena Moroșan
Antibodies 2024, 13(2), 47; https://doi.org/10.3390/antib13020047 - 17 Jun 2024
Abstract
►▼
Show Figures
Elevated immunoglobulin E (IgE) levels are commonly associated with allergies. However, high IgE levels are also found in several other infectious and non-infectious disorders. Elevated IgE levels typically suggest allergies, eczema, or recurrent skin infections. Hyperimmunoglobulin E (hyper-IgE) levels typically reflect a monogenic
[...] Read more.
Elevated immunoglobulin E (IgE) levels are commonly associated with allergies. However, high IgE levels are also found in several other infectious and non-infectious disorders. Elevated IgE levels typically suggest allergies, eczema, or recurrent skin infections. Hyperimmunoglobulin E (hyper-IgE) levels typically reflect a monogenic atopic condition or inborn immune defects with an atopic phenotype. The aim of our research is to investigate and observe the clinical characteristics of children with increased IgE levels who have previously manifested infectious diseases. Furthermore, the retrospective study considers other factors, such as demographic characteristics (sex, area/environment, and age), and their effect on IgE serum levels. To answer this question, we conducted a one-year hospital-based retrospective study that included 200 hospitalized children who had at least two viral or bacterial infections in the six months preceding hospitalization. Measurements of IgE and allergen panels (respiratory and digestive) using blood samples revealed that individuals who tested positive for the body’s synthesis of hyper-IgE were not observably allergic to any potential allergens despite having higher total serum IgE. According to the results, there was a strong correlation between elevated IgE serum levels and a history of infectious diseases among the patients.
Full article

Figure 1
Open AccessReview
Targeting Neoantigens in Cancer: Possibilities and Opportunities in Breast Cancer
by
Zuhair Chaudhry, Anik Boyadzhyan, Kayvan Sasaninia and Vikrant Rai
Antibodies 2024, 13(2), 46; https://doi.org/10.3390/antib13020046 - 10 Jun 2024
Abstract
As one of the most prevalent forms of cancer worldwide, breast cancer has garnered significant attention within the clinical research setting. While traditional treatment employs a multidisciplinary approach including a variety of therapies such as chemotherapy, hormone therapy, and even surgery, researchers have
[...] Read more.
As one of the most prevalent forms of cancer worldwide, breast cancer has garnered significant attention within the clinical research setting. While traditional treatment employs a multidisciplinary approach including a variety of therapies such as chemotherapy, hormone therapy, and even surgery, researchers have since directed their attention to the budding role of neoantigens. Neoantigens are defined as tumor-specific antigens that result from a multitude of genetic alterations, the most prevalent of which is the single nucleotide variant. As a result of their foreign nature, neoantigens elicit immune responses upon presentation by Major Histocompatibility Complexes I and II followed by recognition by T cell receptors. Previously, researchers have been able to utilize these immunogenic properties and manufacture neoantigen-specific T-cells and neoantigen vaccines. Within the context of breast cancer, biomarkers such as tumor protein 53 (TP53), Survivin, Partner and Localizer of BRCA2 (PALB2), and protein tyrosine phosphatase receptor T (PTPRT) display exceeding potential to serve as neoantigens. However, despite their seemingly limitless potential, neoantigens must overcome various obstacles if they are to be fairly distributed to patients. For instance, a prolonged period between the identification of a neoantigen and the dispersal of treatment poses a serious risk within the context of breast cancer. Regardless of these current obstacles, it appears highly promising that future research into neoantigens will make an everlasting impact on the health outcomes within the realm of breast cancer. The purpose of this literature review is to comprehensively discuss the etiology of various forms of breast cancer and current treatment modalities followed by the significance of neoantigens in cancer therapeutics and their application to breast cancer. Further, we have discussed the limitations, future directions, and the role of transcriptomics in neoantigen identification and personalized medicine. The concepts discussed in the original and review articles were included in this review article.
Full article
(This article belongs to the Section Antibody-Based Therapeutics)
►▼
Show Figures

Figure 1
Open AccessArticle
Antibody Recognition of Human Epidermal Growth Factor Receptor-2 (HER2) Juxtamembrane Domain Enhances Anti-Tumor Response of Chimeric Antigen Receptor (CAR)-T Cells
by
Guangyu Zhou, Shengyu Fu, Yunsen Zhang, Shuang Li, Ziang Guo, Defang Ouyang, Tianlei Ying, Yinying Lu and Qi Zhao
Antibodies 2024, 13(2), 45; https://doi.org/10.3390/antib13020045 - 7 Jun 2024
Abstract
Chimeric antigen receptor (CAR) T cell therapy shows promise in treating malignant tumors. However, the use of human epidermal growth factor receptor-2 (HER2) CAR-T cells carries the risk of severe toxicity, including cytokine release syndrome, due to their “on-target off-tumor” recognition of HER2.
[...] Read more.
Chimeric antigen receptor (CAR) T cell therapy shows promise in treating malignant tumors. However, the use of human epidermal growth factor receptor-2 (HER2) CAR-T cells carries the risk of severe toxicity, including cytokine release syndrome, due to their “on-target off-tumor” recognition of HER2. Enhancing the quality and functionality of HER2 CARs could greatly improve the therapeutic potential of CAR-T cells. In this study, we developed a novel anti-HER2 monoclonal antibody, Ab8, which targets domain III of HER2, distinct from the domain IV recognition of trastuzumab. Although two anti-HER2 mAbs induced similar levels of antibody-dependent cellular cytotoxicity, trastuzumab-based CAR-T cells exhibited potent antitumor activity against HER2-positive cancer cells. In conclusion, our findings provide scientific evidence that antibody recognition of the membrane-proximal domain promotes the anti-tumor response of HER2-specific CAR-T cells.
Full article
(This article belongs to the Special Issue A Festschrift Celebrating Dr. Dimiter Stanchev Dimitrov: Antibodies, Innovation, and Impact on Infectious Disease and Cancer Research)
►▼
Show Figures

Figure 1
Open AccessArticle
Evaluating the Clinical Relevance of Antibodies against Non-Human Leukocyte Antigen in Kidney Transplantation
by
Shiv Bhutani, Shelley Harris, Michelle Carr, Marcus Russell-Lowe, Judith Worthington, Henry H. L. Wu, Rajkumar Chinnadurai and Kay Poulton
Antibodies 2024, 13(2), 44; https://doi.org/10.3390/antib13020044 - 6 Jun 2024
Abstract
►▼
Show Figures
Introduction: Kidney transplantation is the preferred modality of kidney replacement therapy for eligible patients with end-stage kidney disease (ESKD), given that it has been found to reduce mortality rates, improve quality of life, and is cost-effective compared to dialysis. Recent advancements in
[...] Read more.
Introduction: Kidney transplantation is the preferred modality of kidney replacement therapy for eligible patients with end-stage kidney disease (ESKD), given that it has been found to reduce mortality rates, improve quality of life, and is cost-effective compared to dialysis. Recent advancements in human leukocyte antigen (HLA) ty** and donor-specific antibody (DSA) detection have helped to reduce the risk of rejection, but antibody-mediated rejection (AMR) can still occur without DSA. Previous studies suggest that rejection can be attributed to antibodies against Non-Human Leucocyte Antigens (non-HLAs). We aimed to acquire further understanding of the prevalence and distribution of non-HLA antibodies in our local population and attempt to correlate these findings with graft outcomes, as well as assess whether non-HLA antibodies can be utilized to determine graft impairment and dysfunction. Methods: We conducted a retrospective study involving kidney transplant recipients between January 2010 and December 2020. All included individuals were aged over 18 and underwent kidney-alone transplants; were ABO- and HLA-compatible; and were matched at A, B, and DR loci (mismatch 0:0:0). HLA testing was negative at the time of transplantation. The samples from both cases of early graft rejection and the control group were tested for non-HLA antibodies using One Lambda LABScreenTM, Autoantibody kit groups 1, 2, and 3, as well as the Immucor LIFECODES non-HLA autoantibody assay. Results: A total of 850 kidney transplant recipients were included, in which 12 patients experienced early graft rejection within the first month post transplant and 18 patients who did not experience graft rejection were selected as study controls. Our study reported no correlation between the total burden of non-HLA antibodies and early rejection, most likely as the result of a small sample size. Nevertheless, a sub-analysis revealed that specific high-frequency pre-transplant non-HLA antibodies such as GSTT, CXCL11, CXCL10, and HNR, detected by LIFECODES, were associated with rejection (Fisher’s exact test with Bonferroni correction, p < 0.001). Most pre-transplant non-HLA antibody levels were reduced after transplantation, which was attributed to immunosuppression. Conclusion: The ‘high frequency’ non-HLA antibodies displayed an association with graft rejection, though the overall associations between the burden of non-HLA antibodies and rejection episodes remain inconclusive. Further work is needed to establish the rebound phenomenon of non-HLA antibodies, the development of de novo non-HLA antibodies in the long run, and their implications on graft survival.
Full article

Figure 1
Open AccessReview
Myelin Oligodendrocyte Glycoprotein-Antibody Associated Disease: An Updated Review of the Clinical Spectrum, Pathogenetic Mechanisms and Therapeutic Management
by
Panagiotis Gklinos and Ruth Dobson
Antibodies 2024, 13(2), 43; https://doi.org/10.3390/antib13020043 - 17 May 2024
Abstract
►▼
Show Figures
Clinical syndromes associated with antibodies against myelin oligodendrocyte glycoprotein (MOG) are now recognized as a distinct neurological disease entity, and are gaining increasing attention. The pathogenic mechanisms underlying MOG-antibody disease (MOGAD) remain incompletely understood. Case series, facilitated by registries, and observational studies over
[...] Read more.
Clinical syndromes associated with antibodies against myelin oligodendrocyte glycoprotein (MOG) are now recognized as a distinct neurological disease entity, and are gaining increasing attention. The pathogenic mechanisms underlying MOG-antibody disease (MOGAD) remain incompletely understood. Case series, facilitated by registries, and observational studies over the past few years have shed increasing light on the clinical aspects and therapeutic approaches of MOGAD. MOGAD may manifest with a variety of clinical syndromes, including acute disseminated encephalomyelitis (ADEM), autoimmune encephalitis, optic neuritis (ON) and transverse myelitis (TM). MOGAD can be either monophasic or relapsing. This review aims to provide a comprehensive updated description of the clinical spectrum, paraclinical features, and prognosis of MOG-antibody disease, as well as summarize its therapeutic considerations. Randomized clinical trials, standardized diagnostic criteria and treatment guidelines are the steps forward.
Full article

Figure 1
Open AccessArticle
Transcriptome Analysis Reveals the Induction of Apoptosis-Related Genes by a Monoclonal Antibody against a New Epitope of CD99 on T-Acute Lymphoblastic Leukemia
by
Nuchjira Takheaw, Kamonporn Kotemul, Ratthakorn Chaiwut, Supansa Pata, Witida Laopajon, Kuntalee Rangnoi, Montarop Yamabhai and Watchara Kasinrerk
Antibodies 2024, 13(2), 42; https://doi.org/10.3390/antib13020042 - 17 May 2024
Abstract
►▼
Show Figures
CD99 was demonstrated to be a potential target for antibody therapy on T-acute lymphoblastic leukemia (T-ALL). The ligation of CD99 by certain monoclonal antibodies (mAbs) induced T-ALL apoptosis. However, the molecular basis contributing to the apoptosis of T-ALL upon anti-CD99 mAb engagement remains
[...] Read more.
CD99 was demonstrated to be a potential target for antibody therapy on T-acute lymphoblastic leukemia (T-ALL). The ligation of CD99 by certain monoclonal antibodies (mAbs) induced T-ALL apoptosis. However, the molecular basis contributing to the apoptosis of T-ALL upon anti-CD99 mAb engagement remains elusive. In this study, using our generated anti-CD99 mAb clone MT99/3 (mAb MT99/3), mAb MT99/3 engagement strongly induced apoptosis of T-ALL cell lines, but not in non-malignant peripheral blood cells. By transcriptome analysis, upon mAb MT99/3 ligation, 13 apoptosis-related genes, including FOS, TNF, FASLG, BCL2A1, JUNB, SOCS1, IL27RA, PTPN6, PDGFA, NR4A1, SGK1, LPAR5 and LTB, were significantly upregulated. The epitope of CD99 recognized by mAb MT99/3 was then identified as the VDGENDDPRPP at residues 60–70 of CD99, which has never been reported. To the best of our knowledge, this is the first transcriptome data conducted in T-ALL with anti-CD99 mAb engagement. These findings provide new insights into CD99 implicated in the apoptosis of T-ALL. The identification of a new epitope and apoptosis-related genes that relate to the induction of apoptosis by mAb MT99/3 may serve as a new therapeutic target for T-ALL. The anti-CD99 mAb clone MT99/3 might be a candidate for further development of a therapeutic antibody for T-ALL therapy.
Full article

Figure 1
Open AccessArticle
Humoral Immunity across the SARS-CoV-2 Spike after Sputnik V (Gam-COVID-Vac) Vaccination
by
Alejandro Cornejo, Christopher Franco, Mariajose Rodriguez-Nuñez, Alexis García, Inirida Belisario, Soriuska Mayora, Domingo José Garzaro, José Luis Zambrano, Rossana Celeste Jaspe, Mariana Hidalgo, Nereida Parra-Giménez, Franklin Ennodio Claro, Ferdinando Liprandi, Jacobus Henri de Waard, Héctor Rafael Rangel and Flor Helene Pujol
Antibodies 2024, 13(2), 41; https://doi.org/10.3390/antib13020041 - 11 May 2024
Abstract
SARS-CoV-2 vaccines have contributed to attenuating the burden of the COVID-19 pandemic by promoting the development of effective immune responses, thus reducing the spread and severity of the pandemic. A clinical trial with the Sputnik-V vaccine was conducted in Venezuela from December 2020
[...] Read more.
SARS-CoV-2 vaccines have contributed to attenuating the burden of the COVID-19 pandemic by promoting the development of effective immune responses, thus reducing the spread and severity of the pandemic. A clinical trial with the Sputnik-V vaccine was conducted in Venezuela from December 2020 to July 2021. The aim of this study was to explore the antibody reactivity of vaccinated individuals towards different regions of the spike protein (S). Neutralizing antibody (NAb) activity was assessed using a commercial surrogate assay, detecting NAbs against the receptor-binding domain (RBD), and a plaque reduction neutralization test. NAb levels were correlated with the reactivity of the antibodies to the spike regions over time. The presence of Abs against nucleoprotein was also determined to rule out the effect of exposure to the virus during the clinical trial in the serological response. A high serological reactivity was observed to S and specifically to S1 and the RBD. S2, although recognized with lower intensity by vaccinated individuals, was the subunit exhibiting the highest cross-reactivity in prepandemic sera. This study is in agreement with the high efficacy reported for the Sputnik V vaccine and shows that this vaccine is able to induce an immunity lasting for at least 180 days. The dissection of the Ab reactivity to different regions of S allowed us to identify the relevance of epitopes outside the RBD that are able to induce NAbs. This research may contribute to the understanding of vaccine immunity against SARS-CoV-2, which could contribute to the design of future vaccine strategies.
Full article
(This article belongs to the Special Issue SARS-CoV-2: Immune Response Elicited by Infection or Vaccination)
►▼
Show Figures

Figure 1
Open AccessArticle
Human Antibodies against Herpes Simplex Virus 2 Glycoprotein G Do Not Neutralize but Mediate Antibody-Dependent Cellular Cytotoxicity
by
Jan-Åke Liljeqvist, Karin Önnheim, Petra Tunbäck, Kristina Eriksson, Staffan Görander, Malin Bäckström and Tomas Bergström
Antibodies 2024, 13(2), 40; https://doi.org/10.3390/antib13020040 - 11 May 2024
Abstract
Herpes simplex virus 2 (HSV-2) is a sexually transmitted infection affecting 491 million individuals globally. Consequently, there is a great need for both prophylactic and therapeutic vaccines. Unfortunately, several vaccine clinical trials, primarily employing the glycoprotein D of HSV-2 (gD-2), have failed. The
[...] Read more.
Herpes simplex virus 2 (HSV-2) is a sexually transmitted infection affecting 491 million individuals globally. Consequently, there is a great need for both prophylactic and therapeutic vaccines. Unfortunately, several vaccine clinical trials, primarily employing the glycoprotein D of HSV-2 (gD-2), have failed. The immune protection conferred by human anti-HSV-2 antibodies in genital infection and disease remains elusive. It is well-known that gD-2 elicits cross-reactive neutralizing antibodies, i.e., anti-gD-2 antibodies recognize gD in HSV-1 (gD-1). In contrast, anti-glycoprotein G in HSV-2 (mgG-2) antibodies are exclusively type-specific for HSV-2. In this study, truncated versions of gD-2 and mgG-2 were recombinantly produced in mammalian cells and used for the purification of anti-gD-2 and anti-mgG-2 antibodies from the serum of five HSV-2-infected subjects, creating a pool of purified antibodies. These antibody pools were utilized as standards together with purified mgG-2 and gD-2 antigens in ELISA to quantitatively estimate and compare the levels of cross-reactive anti-gD-1 and anti-gD-2 antibodies, as well as anti-mgG-2 antibodies in sera from HSV-1+2-, HSV-2-, and HSV-1-infected subjects. The median concentration of anti-mgG-2 antibodies was five times lower in HSV-1+2-infected subjects as compared with cross-reactive anti-gD-1 and anti-gD-2 antibodies, and three times lower in HSV-2 infected subjects as compared with anti-gD-2 antibodies. The pool of purified anti-gD-2 antibodies presented neutralization activity at low concentrations, while the pool of purified anti-mgG-2 antibodies did not. Instead, these anti-mgG-2 antibodies mediated antibody-dependent cellular cytotoxicity (ADCC) by human granulocytes, monocytes, and NK-cells, but displayed no complement-dependent cytotoxicity. These findings indicate that antibodies to mgG-2 in HSV-2-infected subjects are present at low concentrations but mediate the killing of infected cells via ADCC rather than by neutralizing free viral particles. We, and others, speculate that Fc-receptor mediated antibody functions such as ADCC following HSV-2 vaccination may serve as a better marker of protection correlate instead of neutralizing activity. In an mgG-2 therapeutic vaccine, our findings of low levels of anti-mgG-2 antibodies in HSV-2-infected subjects may suggest an opportunity to enhance the immune responses against mgG-2. In a prophylactic HSV-2 mgG-2 vaccine, a possible interference in cross-reactive immune responses in already infected HSV-1 subjects can be circumvented.
Full article
(This article belongs to the Section Humoral Immunity)
►▼
Show Figures

Figure 1
Open AccessArticle
Identification of a Fully Human Antibody VH Domain Targeting Anaplastic Lymphoma Kinase (ALK) with Applications in ALK-Positive Solid Tumor Immunotherapy
by
Chuan Chen, Zehua Sun, Zening Wang, Seungmin Shin, Abigail Berrios, John W. Mellors, Dimiter S. Dimitrov and Wei Li
Antibodies 2024, 13(2), 39; https://doi.org/10.3390/antib13020039 - 7 May 2024
Abstract
The anaplastic lymphoma kinase (ALK, CD247) is a potential target for antibody-based therapy. However, no antibody-based therapeutics targeting ALK have entered clinical trials, necessitating the development of novel antibodies with unique therapeutic merits. Single-domain antibodies (sdAb) bear therapeutic advantages compared to the full-length
[...] Read more.
The anaplastic lymphoma kinase (ALK, CD247) is a potential target for antibody-based therapy. However, no antibody-based therapeutics targeting ALK have entered clinical trials, necessitating the development of novel antibodies with unique therapeutic merits. Single-domain antibodies (sdAb) bear therapeutic advantages compared to the full-length antibody including deeper tumor penetration, cost-effective production and fast washout from normal tissues. In this study, we identified a human immunoglobulin heavy chain variable domain (VH domain) (VH20) from an in-house phage library. VH20 exhibits good developability and high specificity with no off-target binding to ~6000 human membrane proteins. VH20 efficiently bound to the glycine-rich region of ALK with an EC50 of 0.4 nM and a KD of 6.54 nM. Both VH20-based bispecific T cell engager (TCE) and chimeric antigen receptor T cells (CAR Ts) exhibited potent cytolytic activity to ALK-expressing tumor cells in an ALK-dependent manner. VH20 CAR Ts specifically secreted proinflammatory cytokines including IL-2, TNFα and IFNγ after incubation with ALK-positive cells. To our knowledge, this is the first reported human single-domain antibody against ALK. Our in vitro characterization data indicate that VH20 could be a promising ALK-targeting sdAb with potential applications in ALK-expressing tumors, including neuroblastoma (NBL) and non-small cell lung cancer.
Full article
(This article belongs to the Special Issue A Festschrift Celebrating Dr. Dimiter Stanchev Dimitrov: Antibodies, Innovation, and Impact on Infectious Disease and Cancer Research)
►▼
Show Figures

Figure 1
Open AccessArticle
Structural and Functional Characterization of Medicinal Plants as Selective Antibodies towards Therapy of COVID-19 Symptoms
by
Fatemeh Mollaamin
Antibodies 2024, 13(2), 38; https://doi.org/10.3390/antib13020038 - 7 May 2024
Abstract
Considering the COVID-19 pandemic, this research aims to investigate some herbs as probable therapies for this disease. Achillea millefolium (Yarrow), Alkanet, Rumex patientia (Patience dock), Dill, Tarragon, and sweet fennel, including some principal chemical compounds
[...] Read more.
Considering the COVID-19 pandemic, this research aims to investigate some herbs as probable therapies for this disease. Achillea millefolium (Yarrow), Alkanet, Rumex patientia (Patience dock), Dill, Tarragon, and sweet fennel, including some principal chemical compounds of achillin, alkannin, cuminaldehyde, dillapiole, estragole, and fenchone have been selected. The possible roles of these medicinal plants in COVID-19 treatment have been investigated through quantum sensing methods. The formation of hydrogen bonding between the principal substances selected in anti-COVID natural drugs and Tyr-Met-His (the database amino acids fragment), as the active area of the COVID protein, has been evaluated. The physical and chemical attributes of nuclear magnetic resonance, vibrational frequency, the highest occupied molecular orbital energy and the lowest unoccupied molecular orbital energy, partial charges, and spin density have been investigated using the DFT/TD-DFT method and 6-311+G (2d,p) basis set by the Gaussian 16 revision C.01 program toward the industry of drug design. This research has exhibited that there is relative agreement among the results that these medicinal plants could be efficient against COVID-19 symptoms.
Full article
(This article belongs to the Section Humoral Immunity)
►▼
Show Figures

Figure 1
Open AccessEditor’s ChoiceArticle
A Conditionally Activated Cytosol-Penetrating Antibody for TME-Dependent Intracellular Cargo Delivery
by
Carolin Sophie Dombrowsky, Dominic Happel, Jan Habermann, Sarah Hofmann, Sasi Otmi, Benny Cohen and Harald Kolmar
Antibodies 2024, 13(2), 37; https://doi.org/10.3390/antib13020037 - 2 May 2024
Abstract
►▼
Show Figures
Currently, therapeutic and diagnostic applications of antibodies are primarily limited to cell surface-exposed and extracellular proteins. However, research has been conducted on cell-penetrating peptides (CPP), as well as cytosol-penetrating antibodies, to overcome these limitations. In this context, a heparin sulfate proteoglycan (HSPG)-binding antibody
[...] Read more.
Currently, therapeutic and diagnostic applications of antibodies are primarily limited to cell surface-exposed and extracellular proteins. However, research has been conducted on cell-penetrating peptides (CPP), as well as cytosol-penetrating antibodies, to overcome these limitations. In this context, a heparin sulfate proteoglycan (HSPG)-binding antibody was serendipitously discovered, which eventually localizes to the cytosol of target cells. Functional characterization revealed that the tested antibody has beneficial cytosol-penetrating capabilities and can deliver cargo proteins (up to 70 kDa) to the cytosol. To achieve tumor-specific cell targeting and cargo delivery through conditional activation of the cell-penetrating antibody in the tumor microenvironment, a single-chain Fc fragment (scFv) and a VL domain were isolated as masking units. Several in vitro assays demonstrated that fusing the masking protein with a cleavable linker to the cell penetration antibody results in the inactivation of antibody cell binding and internalization. Removal of the mask via MMP-9 protease cleavage, a protease that is frequently overexpressed in the tumor microenvironment (TME), led to complete regeneration of binding and cytosol-penetrating capabilities. Masked and conditionally activated cytosol-penetrating antibodies have the potential to serve as a modular platform for delivering protein cargoes addressing intracellular targets in tumor cells.
Full article

Figure 1
Open AccessEditor’s ChoiceArticle
Balancing the Affinity and Tumor Cell Binding of a Two-in-One Antibody Simultaneously Targeting EGFR and PD-L1
by
Julia Harwardt, Felix Klaus Geyer, Katrin Schoenfeld, David Baumstark, Vera Molkenthin and Harald Kolmar
Antibodies 2024, 13(2), 36; https://doi.org/10.3390/antib13020036 - 2 May 2024
Abstract
The optimization of the affinity of monoclonal antibodies is crucial for the development of drug candidates, as it can impact the efficacy of the drug and, thus, the dose and dosing regimen, limit adverse effects, and reduce therapy costs. Here, we present the
[...] Read more.
The optimization of the affinity of monoclonal antibodies is crucial for the development of drug candidates, as it can impact the efficacy of the drug and, thus, the dose and dosing regimen, limit adverse effects, and reduce therapy costs. Here, we present the affinity maturation of an EGFR×PD-L1 Two-in-One antibody for EGFR binding utilizing site-directed mutagenesis and yeast surface display. The isolated antibody variants target EGFR with a 60-fold-improved affinity due to the replacement of a single amino acid in the CDR3 region of the light chain. The binding properties of the Two-in-One variants were confirmed using various methods, including BLI measurements, real-time antigen binding measurements on surfaces with a mixture of both recombinant proteins and cellular binding experiments using flow cytometry as well as real-time interaction cytometry. An AlphaFold-based model predicted that the amino acid exchange of tyrosine to glutamic acid enables the formation of a salt bridge to an arginine at EGFR position 165. This easily adaptable approach provides a strategy for the affinity maturation of bispecific antibodies with respect to the binding of one of the two antigens.
Full article
(This article belongs to the Section Antibody Discovery and Engineering)
►▼
Show Figures

Figure 1
Open AccessCommunication
Antibodies against Platelet Glycoproteins in Clinically Suspected VITT Patients
by
Romy T. Meier, Leendert Porcelijn, Suzanne Hofstede-van Egmond, Camila Caram-Deelder, Jonathan M. Coutinho, Yvonne M. C. Henskens, Marieke J. H. A. Kruip, An K. Stroobants, Jaap J. Zwaginga, C. Ellen van der Schoot, Masja de Haas and Rick Kapur
Antibodies 2024, 13(2), 35; https://doi.org/10.3390/antib13020035 - 1 May 2024
Abstract
►▼
Show Figures
Vaccine-induced thrombotic thrombocytopenia (VITT) is a rare but severe complication following COVID-19 vaccination, marked by thrombocytopenia and thrombosis. Analogous to heparin-induced thrombocytopenia (HIT), VITT shares similarities in anti-platelet factor 4 (PF4) IgG-mediated platelet activation via the FcγRIIa. To investigate the involvement of platelet-antibodies
[...] Read more.
Vaccine-induced thrombotic thrombocytopenia (VITT) is a rare but severe complication following COVID-19 vaccination, marked by thrombocytopenia and thrombosis. Analogous to heparin-induced thrombocytopenia (HIT), VITT shares similarities in anti-platelet factor 4 (PF4) IgG-mediated platelet activation via the FcγRIIa. To investigate the involvement of platelet-antibodies in VITT, we analyzed the presence of platelet-antibodies directed against glycoproteins (GP)IIb/IIIa, GPV and GPIb/IX in the serum of 232 clinically suspected VITT patients determined based on (suspicion of) occurrence of thrombocytopenia and/or thrombosis in relation to COVID-19 vaccination. We found that 19% of clinically suspected VITT patients tested positive for anti-platelet GPs: 39%, 32% and 86% patients tested positive for GPIIb/IIIa, GPV and GPIb/IX, respectively. No HIT-like VITT patients (with thrombocytopenia and thrombosis) tested positive for platelet-antibodies. Therefore, it seems unlikely that platelet-antibodies play a role in HIT-like anti-PF4-mediated VITT. Platelet-antibodies were predominantly associated with the occurrence of thrombocytopenia. We found no association between the type of vaccination (adenoviral vector vaccine versus mRNA vaccine) or different vaccines (ChAdOx1 nCoV-19, Ad26.COV2.S, mRNA-1273, BTN162b2) and the development of platelet-antibodies. It is essential to conduct more research on the pathophysiology of VITT, to improve diagnostic approaches and identify preventive and therapeutic strategies.
Full article
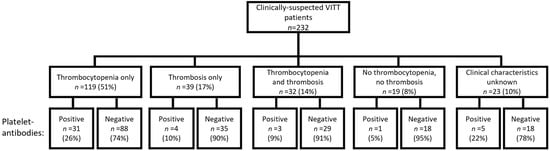
Figure 1
Open AccessBrief Report
Characterization of a Trispecific PD-L1 Blocking Antibody That Exhibits EGFR-Conditional 4-1BB Agonist Activity
by
Laura Rubio-Pérez, Susana Frago, Marta Compte, Rocío Navarro, Seandean L. Harwood, Rodrigo Lázaro-Gorines, Marina Gómez-Rosel, Oana Hangiu, Noelia Silva-Pilipich, Lucía Vanrell, Cristian Smerdou and Luis Álvarez-Vallina
Antibodies 2024, 13(2), 34; https://doi.org/10.3390/antib13020034 - 24 Apr 2024
Abstract
Immune checkpoint blockade has changed the treatment paradigm for advanced solid tumors, but the overall response rates are still limited. The combination of checkpoint blockade with anti-4-1BB antibodies to stimulate tumor-infiltrating T cells has shown anti-tumor activity in human trials. However, the further
[...] Read more.
Immune checkpoint blockade has changed the treatment paradigm for advanced solid tumors, but the overall response rates are still limited. The combination of checkpoint blockade with anti-4-1BB antibodies to stimulate tumor-infiltrating T cells has shown anti-tumor activity in human trials. However, the further clinical development of these antibodies has been hampered by significant off-tumor toxicities. Here, we generated an anti-4-1BB/EGFR/PD-L1 trispecific antibody consisting of a triple-targeting tandem trimerbody (TT) fused to an engineered silent Fc region. This antibody (IgTT-4E1-S) was designed to combine the blockade of the PD-L1/PD-1 axis with conditional 4-1BB costimulation specifically confined to the tumor microenvironment (TME). The antibody demonstrated simultaneous binding to purified EGFR, PD-L1, and 4-1BB in solution, effective blockade of the PD-L1/PD1 interaction, and potent 4-1BB-mediated costimulation, but only in the presence of EGFR-expressing cells. These results demonstrate the feasibility of IgTT-4E1-S specifically blocking the PD-L1/PD-1 axis and inducing EGFR-conditional 4-1BB agonist activity.
Full article
(This article belongs to the Special Issue Emerging Antibody Engineering Strategies and Applications for Immunotherapy of Cancer)
►▼
Show Figures
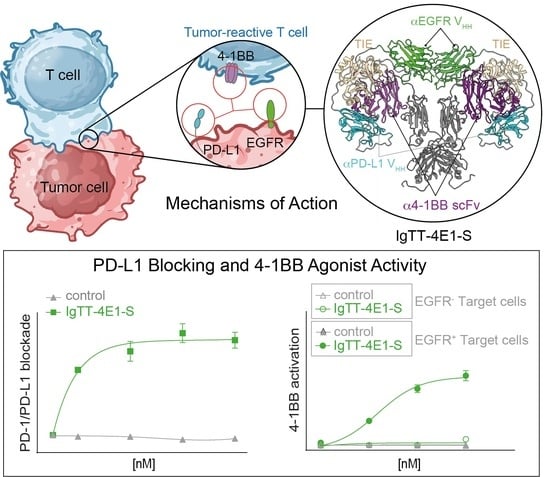
Graphical abstract
Open AccessFeature PaperEditor’s ChoiceArticle
Cross-Reactivity of N6AMT1 Antibodies with Aurora Kinase A: An Example of Antibody-Specific Non-Specificity
by
Baiba Brūmele, Evgeniia Serova, Aleksandra Lupp, Mihkel Suija, Margit Mutso and Reet Kurg
Antibodies 2024, 13(2), 33; https://doi.org/10.3390/antib13020033 - 22 Apr 2024
Abstract
►▼
Show Figures
Primary antibodies are one of the main tools used in molecular biology research. However, the often-occurring cross-reactivity of primary antibodies complicates accurate data analysis. Our results show that three commercial polyclonal antibodies raised against N-6 adenine-specific DNA methyltransferase 1 (N6AMT1) strongly cross-react with
[...] Read more.
Primary antibodies are one of the main tools used in molecular biology research. However, the often-occurring cross-reactivity of primary antibodies complicates accurate data analysis. Our results show that three commercial polyclonal antibodies raised against N-6 adenine-specific DNA methyltransferase 1 (N6AMT1) strongly cross-react with endogenous and recombinant mitosis-associated protein Aurora kinase A (AURKA). The cross-reactivity was verified through immunofluorescence, immunoblot, and immunoprecipitation assays combined with mass spectrometry. N6AMT1 and AURKA are evolutionarily conserved proteins that are vital for cellular processes. Both proteins share the motif ENNPEE, which is unique to only these two proteins. We suggest that N6AMT1 antibodies recognise this motif in N6AMT1 and AURKA proteins and exhibit an example of “specific” non-specificity. This serves as an example of the importance of controls and critical data interpretation in molecular biology research.
Full article

Figure 1

Journal Menu
► ▼ Journal Menu-
- Antibodies Home
- Aims & Scope
- Editorial Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Antibodies, Cancers, Immuno, IJMS, Vaccines
Anti-Tumor Immune Responses 2.0
Topic Editors: Massimo Zollo, Renata GrifantiniDeadline: 31 August 2024

Conferences
Special Issues
Special Issue in
Antibodies
Review Collection on Humoral Immunity
Guest Editors: Jagadeesh Bayry, Linqi ZhangDeadline: 31 July 2024
Special Issue in
Antibodies
Design, Production and Characterization of Peptide Antibodies—Volume II
Guest Editors: Nicole H. Trier, Gunnar HouenDeadline: 31 August 2024
Special Issue in
Antibodies
Antibodies in Laboratory Diagnostic Techniques
Guest Editor: Kinga LisDeadline: 25 September 2024
Special Issue in
Antibodies
Unravelling Effector Functions of B cells in Infectious Diseases and Cancer
Guest Editor: Farhat AfrinDeadline: 20 November 2024
Topical Collections
Topical Collection in
Antibodies
Computational Antibody and Antigen Design
Collection Editor: Buyong Ma










