Journal Description
Hematology Reports
Hematology Reports
- formerly Hematology Reviews - is an international, peer-reviewed, open access journal on all aspects of prevention, diagnosis and management of disorders of the blood, published quarterly online by MDPI (from Volume 14, Issue 1 - 2022). The Society of Hematologic Oncology Italy (SOHO Italy) is affiliated with Hematology Reports and their members receive discounts on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), PMC, PubMed, Embase, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 32.8 days after submission; acceptance to publication is undertaken in 5.5 days (median values for papers published in this journal in the first half of 2024).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
Impact Factor:
1.1 (2023);
5-Year Impact Factor:
1.1 (2023)
Latest Articles
Double Trouble: COVID-19 Infection Exacerbates Sickle Cell Crisis Outcomes in Hospitalized Patients—Insights from National Inpatient Sample 2020
Hematol. Rep. 2024, 16(3), 421-430; https://doi.org/10.3390/hematolrep16030041 (registering DOI) - 29 Jun 2024
Abstract
Background: This study investigated the impact of COVID-19 on patients with sickle cell crisis (SCC) using National Inpatient Sample (NIS) data for the year 2020. Methods: A retrospective cohort analysis was conducted utilizing International Classification of Diseases (ICD-10) codes to identify adults who
[...] Read more.
Background: This study investigated the impact of COVID-19 on patients with sickle cell crisis (SCC) using National Inpatient Sample (NIS) data for the year 2020. Methods: A retrospective cohort analysis was conducted utilizing International Classification of Diseases (ICD-10) codes to identify adults who were admitted with a principal diagnosis of sickle cell crisis. The primary outcomes examined were inpatient mortality, while the secondary outcomes assessed included morbidity, hospital length of stay, and resource utilization. Analyses were conducted with STATA. Multivariate logistic and linear regression analyses were used to adjust for confounding variables. Results: Of 66,415 adult patients with a primary SCC diagnosis, 875 were identified with a secondary diagnosis of COVID-19 infection. Unadjusted mortality rate was higher for SCC patients with COVID-19 (2.28%) compared to those without (0.33%), with an adjusted odds ratio (aOR) of 8.49 (p = 0.001). They also showed increased odds of develo** acute respiratory failure (aOR = 2.37, p = 0.003) and acute kidney injury requiring dialysis (aOR = 8.66, p = 0.034). Additionally, these patients had longer hospital stays by an adjusted mean of 3.30 days (p < 0.001) and incurred higher hospitalization charges by an adjusted mean of USD 35,578 (p = 0.005). Conclusions: The SCC patients with COVID-19 presented higher mortality rates, increased morbidity indicators, longer hospital stays, and substantial economic burdens.
Full article
Open AccessCase Report
A Rare Onset of T-Lymphoid Blast Crisis in Chronic Myeloid Leukemia with Two Distinct Blast Populations
by
Alessandra Mongia, Francesca Romano, Sara Ciullini Mannurita, Benedetta Peruzzi, Sara Bencini, Daniela Parrini, Laura Fasano and Alessandra Fanelli
Hematol. Rep. 2024, 16(3), 413-420; https://doi.org/10.3390/hematolrep16030040 - 27 Jun 2024
Abstract
Chronic myeloid leukemia (CML) is a myeloproliferative neoplasm characterized by bone marrow expansion and the proliferation of one or more myeloid cell lineages, predominantly driven by the expression of the constitutively active fusion product tyrosine kinase BCR:ABL1. Rarely, CML patients directly develop a
[...] Read more.
Chronic myeloid leukemia (CML) is a myeloproliferative neoplasm characterized by bone marrow expansion and the proliferation of one or more myeloid cell lineages, predominantly driven by the expression of the constitutively active fusion product tyrosine kinase BCR:ABL1. Rarely, CML patients directly develop a blast crisis (BC), mostly of myeloid origin. CML at blast crisis with a T-cell phenotype at diagnosis, without any prior history of CML, is extremely rare. Herein, we describe one rare CML case, in a young man showing an unusual and early T-lymphoid blastic crisis at diagnosis, as the first onset of a previously unknown CML. The multidisciplinary collaboration between laboratorians and clinicians for the diagnosis and management of this atypical case was crucial in outlining both a targeted pharmacological treatment and a successful hematopoietic stem cell transplantation.
Full article
Open AccessReview
Treatment of Immune Thrombocytopenia: Contextualization from a Historical Perspective
by
Daniel Martínez-Carballeira, Ángel Bernardo, Alberto Caro, Inmaculada Soto and Laura Gutiérrez
Hematol. Rep. 2024, 16(3), 390-412; https://doi.org/10.3390/hematolrep16030039 - 26 Jun 2024
Abstract
►▼
Show Figures
Immune thrombocytopenia (ITP) is an autoimmune disease characterized by an isolated decrease in platelet count and an increased risk of bleeding. The pathogenesis is complex, affecting multiple components of the immune system and causing both peripheral destruction of platelets and inadequate production in
[...] Read more.
Immune thrombocytopenia (ITP) is an autoimmune disease characterized by an isolated decrease in platelet count and an increased risk of bleeding. The pathogenesis is complex, affecting multiple components of the immune system and causing both peripheral destruction of platelets and inadequate production in the bone marrow. In this article, we review the treatment of ITP from a historical perspective, discussing first line and second line treatments, and management of refractory disease.
Full article

Figure 1
Open AccessReview
Neutropenia in Childhood—A Narrative Review and Practical Diagnostic Approach
by
Georgios Katsaras, Silouani Koutsi, Evdokia Psaroulaki, Dimitra Gouni and Pelagia Tsitsani
Hematol. Rep. 2024, 16(2), 375-389; https://doi.org/10.3390/hematolrep16020038 - 16 Jun 2024
Abstract
►▼
Show Figures
Neutropenia refers to a decrease in the absolute neutrophil count according to age and race norms and poses a common concern in pediatric practice. Neutrophils serve as host defenders and act crucially in acute inflammation procedures. In this narrative review, we systematically present
[...] Read more.
Neutropenia refers to a decrease in the absolute neutrophil count according to age and race norms and poses a common concern in pediatric practice. Neutrophils serve as host defenders and act crucially in acute inflammation procedures. In this narrative review, we systematically present causes of neutropenia in childhood, mainly adopting the pathophysiological classification of Frater, thereby studying (1) neutropenia with reduced bone marrow reserve, (2) secondary neutropenia with reduced bone marrow reserve, and (3) neutropenia with normal bone marrow reserve. Different conditions in each category are thoroughly discussed and practically approached from the clinician’s point of view. Secondary mild to moderate neutropenia is usually benign due to childhood viral infections and is expected to resolve in 2–4 weeks. Bacterial and fungal agents are also associated with transient neutropenia, although fever with severe neutropenia constitutes a medical emergency. Drug-induced and immune neutropenias should be suspected following a careful history and a detailed clinical examination. Cytotoxic chemotherapies treating malignancies are responsible for severe neutropenia and neutropenic shock. Rare genetic neutropenias usually manifest with major infections early in life. Our review of taxonomies clinical findings and associates them to specific neutropenia disorders. We consequently propose a practical diagnostic algorithm for managing neutropenic children.
Full article

Figure 1
Open AccessCase Report
Three-Way Translocation t(12;15;17) (p13;q24;q21) Found in Acute Promyelocytic Leukemia with Basophilic Differentiation
by
Sara Frazzetto, Lara Gullo, Gabriele Sapuppo, Manlio Fazio, Cristina Lo Faro, Giuliana Giunta, Ignazio Caravotta, Elisa Mauro, Marina Silvia Parisi, Anna Maria Triolo, Nunziatina Laura Parrinello, Maria Letizia Consoli, Loredana També, Daniela Cambria, Sara Marino, Grazia Scuderi and Francesco Di Raimondo
Hematol. Rep. 2024, 16(2), 367-374; https://doi.org/10.3390/hematolrep16020037 - 12 Jun 2024
Abstract
►▼
Show Figures
Acute promyelocytic leukemia is a rare form of acute myeloid leukemia in which immature promyelocytes abnormally proliferate in the bone marrow. In most cases, the disease is characterised by the translocation t(15;17) (q24;q21), which causes the formation of PML::RARA, an oncogenic fusion protein
[...] Read more.
Acute promyelocytic leukemia is a rare form of acute myeloid leukemia in which immature promyelocytes abnormally proliferate in the bone marrow. In most cases, the disease is characterised by the translocation t(15;17) (q24;q21), which causes the formation of PML::RARA, an oncogenic fusion protein responsible for blocking myeloid differentiation and survival advantage. Here, we present a case of acute promyelocytic leukemia with two unusual features: basophilic differentiation and a three-way translocation involving chromosomes 12, 15 and 17. In the few cases reported, basophilic differentiation was associated with a poor prognosis. In contrast, our patient responded promptly to the standard treatment with all-trans-retinoic acid (ATRA) and arsenic trioxide (ATO) and obtained complete remission. To our knowledge, this is the first report of basophilic acute promyelocytic leukemia with the three-way translocation t(12;17;15) (p13; q24;q21).
Full article

Figure 1
Open AccessReview
Skin Hypopigmentation in Hematology Disorders
by
Roberto Mazzetto, Paola Miceli, Alvise Sernicola, Jacopo Tartaglia and Mauro Alaibac
Hematol. Rep. 2024, 16(2), 354-366; https://doi.org/10.3390/hematolrep16020036 - 4 Jun 2024
Abstract
►▼
Show Figures
Hypopigmentation disorders pose significant diagnostic challenges in dermatology, sometimes reflecting underlying hematological conditions. This review explores the clinical presentations related to hypopigmentation in hematological disorders, focusing on vitiligo, morphea, and syndromic albinism. Vitiligo, an autoimmune disorder targeting melanocytes, involves interactions between genetic polymorphisms
[...] Read more.
Hypopigmentation disorders pose significant diagnostic challenges in dermatology, sometimes reflecting underlying hematological conditions. This review explores the clinical presentations related to hypopigmentation in hematological disorders, focusing on vitiligo, morphea, and syndromic albinism. Vitiligo, an autoimmune disorder targeting melanocytes, involves interactions between genetic polymorphisms and immune responses, particularly regarding CD8+ T cells and IFN-γ. Drug-induced vitiligo, notably by immune checkpoint inhibitors and small-molecule targeted anticancer therapies, underscores the importance of immune dysregulation. Morphea, an inflammatory skin disorder, may signal hematological involvement, as seen in deep morphea and post-radiotherapy lesions. Syndromic albinism, linked to various genetic mutations affecting melanin production, often presents with hematologic abnormalities. Treatment approaches focus on targeting the immune pathways specific to the condition, and when that is not possible, managing symptoms. Understanding these dermatological manifestations is crucial for the timely diagnosis and management of hematological disorders.
Full article

Figure 1
Open AccessCase Report
Blinatumomab in Children with MRD-Positive B-Cell Precursor Acute Lymphoblastic Leukemia: A Report of 11 Cases
by
Yi-Lun Wang, Tsung-Yen Chang, Yu-Chuan Wen, Shu-Ho Yang, Yi-Wen Hsiao, Chia-Chi Chiu, Yu-Chieh Chen, Ruei-Shan Hu, Shih-Hsiang Chen, Tang-Her Jaing and Chih-Cheng Hsiao
Hematol. Rep. 2024, 16(2), 347-353; https://doi.org/10.3390/hematolrep16020035 - 3 Jun 2024
Abstract
Background/Objectives: Relapsed B-cell acute lymphoblastic leukemia (B-ALL) remains an unresolved matter of concern regarding adverse outcomes. This case study aimed to evaluate the effectiveness of blinatumomab, with or without door lymphocyte infusion (DLI), in treating measurable residual disease (MRD)-positive B-ALL. Methods: All patients
[...] Read more.
Background/Objectives: Relapsed B-cell acute lymphoblastic leukemia (B-ALL) remains an unresolved matter of concern regarding adverse outcomes. This case study aimed to evaluate the effectiveness of blinatumomab, with or without door lymphocyte infusion (DLI), in treating measurable residual disease (MRD)-positive B-ALL. Methods: All patients who received blinatumomab salvage therapy were included in this study. Eleven patients were included in the study. All patients were evaluated for MRD-negativity. Results: Before starting blinatumomab therapy, seven patients tested positive for MRD, three tested negative, and one had refractory disease. Hematopoietic cell transplantation (HCT) was reserved for five patients with persistent MRD. Six patients became MRD-negative and subsequent HCT was not performed. Only two patients relapsed; one patient died of relapse, and the other one received carfilzomib-based therapy and was MRD-negative thereafter. Nine patients were MRD-negative at a median follow-up of 28 months (15–52 months). Two of three MRD-positive post-transplant patients remained in complete molecular remission after preemptive DLI at the last follow-up date. In the first salvage, blinatumomab may achieve complete remission and bridging to HCT in pediatric patients with end-of-induction MRD-positive B-cell precursor ALL. Conclusions: The decision on how to treat post-transplant relapse continues to affect survival outcomes. Blinatumomab combined with DLI may extend the armamentarium of release options for high-risk pediatric patients. This approach is encouraging for high-risk ALL patients who are MRD-positive post-transplantation.
Full article
(This article belongs to the Topic Myeloma and Leukemia-Challenges and Current Treatment Options)
►▼
Show Figures

Figure 1
Open AccessArticle
Phase II Trial of Romidepsin as Consolidation Therapy after Gemcitabine, Dexamethasone, and Cisplatin in Elderly Transplant-Ineligible Patients with Relapsed/Refractory Peripheral T-Cell Lymphoma
by
Satoshi Yamasaki, Hiroatsu Iida, Akio Saito, Morio Matsumoto, Yoshiaki Kuroda, Tohru Izumi, Akiko M. Saito, Hiroaki Miyoshi, Koichi Ohshima, Hirokazu Nagai and Hiromi Iwasaki
Hematol. Rep. 2024, 16(2), 336-346; https://doi.org/10.3390/hematolrep16020034 - 28 May 2024
Abstract
Romidepsin is an important therapeutic option for patients with peripheral T-cell lymphoma (PTCL). However, the timing of romidepsin administration remains controversial. The objective of this study was to characterize the safety and efficacy of romidepsin as consolidation therapy after gemcitabine, dexamethasone, and cisplatin
[...] Read more.
Romidepsin is an important therapeutic option for patients with peripheral T-cell lymphoma (PTCL). However, the timing of romidepsin administration remains controversial. The objective of this study was to characterize the safety and efficacy of romidepsin as consolidation therapy after gemcitabine, dexamethasone, and cisplatin (GDP) therapy (GDPR). This study of patients treated between March 2019 and March 2021 was registered with the Japan Registry of Clinical Trials (registration number: jRCT0000000519). If complete response, partial response, or stable disease was confirmed after 2–4 GDP cycles, romidepsin was administered every 4 weeks for 1 year. Seven patients with relapsed/refractory (R/R) PTCL (T-follicular helper phenotype [n = 1] and angioimmunoblastic T-cell lymphoma [n = 6]) were included in this prospective study (PTCL-GDPR). After a median follow-up of 34 months of patients in PTCL-GDPR, the 2-year overall survival rate was 71%, and the overall response rate after treatment was 57%. Common adverse events in patients with PTCL-GDPR included hematological toxicities such as neutropenia, which improved with supportive treatment. There were no treatment-related mortalities. GDPR might be safe and effective in elderly transplant-ineligible patients with R/R PTCL; however, further investigation is required.
Full article
(This article belongs to the Special Issue Personalized Therapies and Clinical Outcomes for Older Patients with Lymphoma)
►▼
Show Figures

Figure 1
Open AccessCase Report
Morphological Clues of Acute Monocytic Leukemia in COVID-19-Induced Transient Leukoerythroblastic Reaction with Monocytosis
by
Ingrid S. Tam, Mohamed Elemary, John DeCoteau, Anna Porwit and Emina E. Torlakovic
Hematol. Rep. 2024, 16(2), 331-335; https://doi.org/10.3390/hematolrep16020033 - 28 May 2024
Abstract
►▼
Show Figures
Viral infections, including those caused by COVID-19, can produce striking morphologic changes in peripheral blood. Distinguishing between reactive changes and abnormal morphology of monocytes remains particularly difficult, with low consensus rates reported amongst hematopathologists. Here, we report a patient who developed transient monocytosis
[...] Read more.
Viral infections, including those caused by COVID-19, can produce striking morphologic changes in peripheral blood. Distinguishing between reactive changes and abnormal morphology of monocytes remains particularly difficult, with low consensus rates reported amongst hematopathologists. Here, we report a patient who developed transient monocytosis of 11.06 × 109/L with 32% promonocytes and 1% blasts during hospitalization that was secondary to severe COVID-19 infection. Three days later, the clinical status of the patient improved and the WBC had decreased to 8.47 × 109/L with 2.2 × 109/L monocytes. Flow cytometry studies did not reveal immunophenotypic findings specific for an overt malignant population. At no time during admission did the patient develop cytopenia(s), and she was discharged upon clinical improvement. However, the peripheral blood sample containing promonocytes was sent for molecular testing with an extended next-generation sequencing myeloid panel and was positive for pathogenic NPM1 Type A and DNMT3A R882H mutations. Subsequently, despite an essentially normal complete blood count, the patient underwent a bone marrow assessment that showed acute myeloid leukemia with 77% promonocytes. This case emphasizes the critical importance of a full work up to exclude acute leukemia when classical promonocyte morphology is encountered in the peripheral blood. Promonocytes are not a part of the reactive changes associated with COVID-19 and remain specific to myeloid neoplasia.
Full article

Figure 1
Open AccessReview
Appropriate Treatment Intensity for Diffuse Large B-Cell Lymphoma in the Older Population: A Review of the Literature
by
Satoshi Yamasaki
Hematol. Rep. 2024, 16(2), 317-330; https://doi.org/10.3390/hematolrep16020032 - 24 May 2024
Abstract
Most patients with diffuse large B-cell lymphoma (DLBCL) are >65 years of age, with the number of patients expected to increase in the coming years. A comprehensive geriatric assessment that carefully evaluates fitness status and comorbidities is essential for selecting the appropriate treatment
[...] Read more.
Most patients with diffuse large B-cell lymphoma (DLBCL) are >65 years of age, with the number of patients expected to increase in the coming years. A comprehensive geriatric assessment that carefully evaluates fitness status and comorbidities is essential for selecting the appropriate treatment intensity. Although generally healthy patients or those <80 years of age may benefit from standard immunochemotherapy, unfit/frail patients or patients >80 years old may require reduced-intensity chemotherapy or less-toxic drugs. Some new drugs are currently being tested as single or combined agents for first-line treatment, aiming to improve the outcomes of conventional chemotherapy. This review systematically collates and discusses the outcomes associated with the use of immunochemotherapy in older patients with DLBCL, as well as considering the impact of full-dose immunochemotherapy on quality of life in older and frail patients, summarizing the rationale for reduced dosing in the older population, and presenting recommendations for selecting patients likely to benefit from reduced dosing. If preliminary efficacy and safety data are confirmed in future clinical trials, non-chemotherapy-based immunotherapy approaches could become an alternative potentially curative option in frail patients and those >80 years of age with DLBCL.
Full article
(This article belongs to the Special Issue Personalized Therapies and Clinical Outcomes for Older Patients with Lymphoma)
►▼
Show Figures

Figure 1
Open AccessCase Report
EBV-Positive Nodal T- and NK-Cell Lymphoma Mimicking Anaplastic Large Cell Lymphoma: A Case Report
by
Brooj Abro, Pamela Allen, Saja Asakrah, Kyle Bradley and Linsheng Zhang
Hematol. Rep. 2024, 16(2), 308-316; https://doi.org/10.3390/hematolrep16020031 - 23 May 2024
Abstract
►▼
Show Figures
EBV-positive nodal T- and NK-cell lymphoma (EBV+ NT/NKCL) is a recently recognized entity in the 5th edition of the WHO Classification of Tumors of Hematopoietic and Lymphoid Tissues. Notably, CD30 positivity is frequently observed in (EBV+ NT/NKCL), creating diagnostic challenges to distinguish it
[...] Read more.
EBV-positive nodal T- and NK-cell lymphoma (EBV+ NT/NKCL) is a recently recognized entity in the 5th edition of the WHO Classification of Tumors of Hematopoietic and Lymphoid Tissues. Notably, CD30 positivity is frequently observed in (EBV+ NT/NKCL), creating diagnostic challenges to distinguish it from ALK-negative anaplastic large cell lymphoma (ALCL). Furthermore, cases of EBV+ ALCL have been documented in the literature, predating the inclusion of EBV+ nodal cytotoxic T-cell lymphoma as a variant of peripheral T-cell lymphoma. We present a case of a 47-year-old male presenting with multiple lymphadenopathies. The histomorphologic and immunophenotypic features of the lymph node closely resemble ALK-negative ALCL, characterized by uniform CD30 expression and a subcapsular distribution of lymphoma cells. However, the lymphoma cells exhibit diffuse positivity for EBV, consistent with EBV+ NT/NKCL. A case of ALK-negative ALCL with an immunophenotype identical to the EBV-positive case is included for comparison. Given that EBV+ NT/NKCL represents an aggressive neoplasm requiring unique clinical management compared to ALK-negative ALCL, it is critical to accurately differentiate EBV+ NT/NKCL from ALK-negative ALCL with a cytotoxic T-cell immunophenotype.
Full article

Figure 1
Open AccessCase Report
Complications of Brentuximab Therapy in Patients with Hodgkin’s Lymphoma and Concurrent Autoimmune Pathology—A Case Series
by
Oana Diana Preda, Sorina Bădeliță, Iulia Ursuleac, Ruxandra Maria Irimia, Sonia Balanica, Monica Cojocaru, Cristina Cotruta, Camelia Dobrea and Daniel Coriu
Hematol. Rep. 2024, 16(2), 299-307; https://doi.org/10.3390/hematolrep16020030 - 20 May 2024
Abstract
►▼
Show Figures
Background: Brentuximab Vedotin (BV) has revolutionized the treatment landscape for Hodgkin’s lymphoma, yet its effects on pre-existing autoimmune disorders remain elusive. Methods: Here, we present four cases of patients with concurrent autoimmune conditions—Crohn’s disease, vitiligo, type I diabetes, and minimal change
[...] Read more.
Background: Brentuximab Vedotin (BV) has revolutionized the treatment landscape for Hodgkin’s lymphoma, yet its effects on pre-existing autoimmune disorders remain elusive. Methods: Here, we present four cases of patients with concurrent autoimmune conditions—Crohn’s disease, vitiligo, type I diabetes, and minimal change disease—undergoing BV therapy for Hodgkin’s lymphoma. The patients were treated with A-AVD instead of ABVD due to advanced-stage disease with high IPI scores. Results: Our findings reveal the surprising and complex interplay between BV exposure and autoimmune manifestations, highlighting the need for multidisciplinary collaboration in patient management. Notably, the exacerbation of autoimmune symptoms was observed in the first three cases where T-cell-mediated autoimmunity predominated. Additionally, BV exposure precipitated autoimmune thrombocytopenia in the vitiligo patient, underscoring the profound disruptions in immune regulation. Conversely, in the minimal change disease case, a disease characterized by a blend of B- and T-cell-mediated immunity, the outcome was favorable. Conclusions: This paper underscores the critical importance of vigilance toward autoimmune flare-ups induced by BV in patients with concurrent autoimmune conditions, offering insights for tailored patient care.
Full article

Figure 1
Open AccessCase Report
Anti-PF4 ELISA-Negative, SRA-Positive Heparin-Induced Thrombocytopenia
by
Abraham Attah, Chelsea Peterson, Max Jacobs, Rama Bhagavatula, Deep Shah, Robert Kaplan and Yazan Samhouri
Hematol. Rep. 2024, 16(2), 295-298; https://doi.org/10.3390/hematolrep16020029 - 9 May 2024
Abstract
►▼
Show Figures
Heparin products are frequently used in the inpatient setting to prevent and treat venous thromboembolism, but they simultaneously put patients at risk of develo** heparin-induced thrombocytopenia (HIT). The 4Ts score determines the pretest probability of HIT. Diagnosis is made with a screening antiplatelet
[...] Read more.
Heparin products are frequently used in the inpatient setting to prevent and treat venous thromboembolism, but they simultaneously put patients at risk of develo** heparin-induced thrombocytopenia (HIT). The 4Ts score determines the pretest probability of HIT. Diagnosis is made with a screening antiplatelet factor (PF4) immunoassay and the serotonin-release assay (SRA) as a confirmatory test. Anti-PF4 assays have high sensitivity (98%) but lower specificity (50%) and result in frequent false-positive tests. We present a rare case from our institution of a patient with anti-PF4–Polyanion ELISA-negative, SRA-positive HIT and describe the challenges in making a timely diagnosis in this case.
Full article
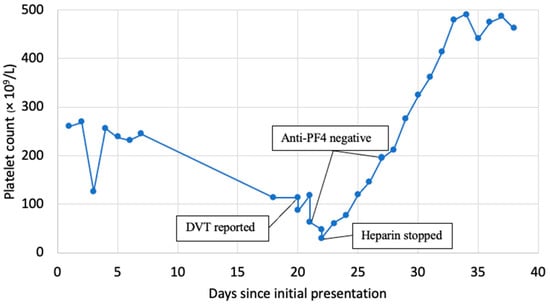
Figure 1
Open AccessArticle
Treatment Patterns and Clinical Outcomes of Patients with Moderate to Severe Acute Graft-Versus-Host Disease: A Multicenter Chart Review Study
by
David Michonneau, Raynier Devillier, Mikko Keränen, Marie Thérèse Rubio, Malin Nicklasson, Hélène Labussière-Wallet, Martin Carre, Anne Huynh, Elisabet Viayna, Montserrat Roset, Jonathan Finzi, Minja Pfeiffer, Daniel Thunström, Núria Lara, Lorenzo Sabatelli, Patrice Chevallier and Maija Itälä-Remes
Hematol. Rep. 2024, 16(2), 283-294; https://doi.org/10.3390/hematolrep16020028 - 6 May 2024
Abstract
►▼
Show Figures
Acute graft-versus-host disease (aGVHD) remains a barrier to successful allogeneic hematopoietic stem cell transplantation (HSCT) outcomes. Contemporary comprehensive analyses of real-world clinical outcomes among patients who develop aGVHD post-HSCT are needed to better understand the unmet needs of this patient population. This multicenter,
[...] Read more.
Acute graft-versus-host disease (aGVHD) remains a barrier to successful allogeneic hematopoietic stem cell transplantation (HSCT) outcomes. Contemporary comprehensive analyses of real-world clinical outcomes among patients who develop aGVHD post-HSCT are needed to better understand the unmet needs of this patient population. This multicenter, retrospective chart review describes treatment patterns and clinical outcomes among patients (≥18 years old) from Finland, Sweden, and France who developed grades II–IV aGVHD after their first HSCT (January 2016–June 2017). From 13 participating centers, 151 patients were included. The median (Q1, Q3) age at HSCT was 56 (45, 62) years old. One line of aGVHD treatment was received by 47.7%, and the most common first-line treatment was methylprednisolone (alone or in a combination regimen, 74.2%; monotherapy, 25.8%). Among patients treated with methylprednisolone, 79.5% achieved a complete or partial response. The median (Q1, Q3) number of treatment lines was 2.0 (1.0, 3.0). The median (Q1, Q3) time to obtain an aGVHD diagnosis from transplant was 29.5 (21.0, 44.0) days, and 14.5 (7.0, 34.0) days to achieve the best response for 110 evaluable patients. At 6 and 12 months, 53.6% and 49.0%, respectively, achieved a complete response. Chronic GVHD occurred in 37.7% of patients, and aGVHD reoccurred in 26.5%. Following aGVHD diagnosis, mortality rates were 30.0% at 6 months and 37.3% at 12 months. Findings from this study demonstrate a continuing unmet need for new therapies that control aGVHD and improve mortality.
Full article

Figure 1
Open AccessReview
Current Therapeutic Sequencing in Chronic Lymphocytic Leukemia
by
Samir Mouhssine, Nawar Maher, Sreekar Kogila, Claudio Cerchione, Giovanni Martinelli and Gianluca Gaidano
Hematol. Rep. 2024, 16(2), 270-282; https://doi.org/10.3390/hematolrep16020027 - 30 Apr 2024
Abstract
►▼
Show Figures
The treatment landscape of chronic lymphocytic leukemia (CLL), the most frequent leukemia in adults, is constantly changing. CLL patients can be divided into three risk categories, based on their IGHV mutational status and the occurrence of TP53 disruption and/or complex karyotype. For the
[...] Read more.
The treatment landscape of chronic lymphocytic leukemia (CLL), the most frequent leukemia in adults, is constantly changing. CLL patients can be divided into three risk categories, based on their IGHV mutational status and the occurrence of TP53 disruption and/or complex karyotype. For the first-line treatment of low- and intermediate-risk CLL, both the BCL2 inhibitor venetoclax plus obinutuzumab and the second generation BTK inhibitors (BTKi), namely acalabrutinib and zanubrutinib, are valuable and effective options. Conversely, venetoclax-based fixed duration therapies have not shown remarkable results in high-risk CLL patients, while continuous treatment with acalabrutinib and zanubrutinib displayed favorable outcomes, similar to those obtained in TP53 wild-type patients. The development of acquired resistance to pathway inhibitors is still a clinical challenge, and the optimal treatment sequencing of relapsed/refractory CLL is not completely established. Covalent BTKi-refractory patients should be treated with venetoclax plus rituximab, whereas venetoclax-refractory CLL may be treated with second generation BTKi in the case of early relapse, while venetoclax plus rituximab might be used if late relapse has occurred. On these grounds, here we provide an overview of the current state-of-the-art therapeutic algorithms for treatment-naïve patients, as well as for relapsed/refractory disease.
Full article
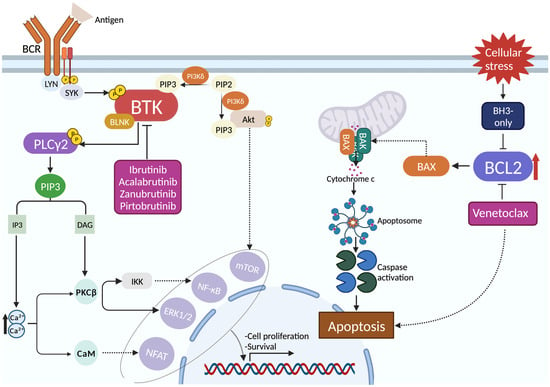
Figure 1
Open AccessCase Report
Is There an Association between a Tonsillar Diffuse Large B-Cell Lymphoma Arising after a Neck Squamous Cell Carcinoma of Occult Primary? A Case Report and Extensive Literature Review
by
Dimitris Tatsis, Athena Niakou, Konstantinos Paraskevopoulos, Stavroula Papadopoulou and Konstantinos Vahtsevanos
Hematol. Rep. 2024, 16(2), 260-269; https://doi.org/10.3390/hematolrep16020026 - 29 Apr 2024
Abstract
►▼
Show Figures
Objectives: The aim of this review is to focus on the possibility of patients with squamous cell carcinoma to develop a second primary disease such as DLBCL, perhaps because of the irradiation of the head and neck area. Materials and methods: A case
[...] Read more.
Objectives: The aim of this review is to focus on the possibility of patients with squamous cell carcinoma to develop a second primary disease such as DLBCL, perhaps because of the irradiation of the head and neck area. Materials and methods: A case of an 89-year-old man is reported, who initially underwent surgical and complementary treatment for neck squamous cell carcinoma of occult primary and later for tonsillar diffuse large B-cell non-Hodgkin lymphoma. Results: The second primary was considered a recurrence in the neck of the original cancer of unknown primary, so a new surgical management was decided. The final pathology report described a diffuse large B-cell non-Hodgkin lymphoma. Conclusions: The importance of maintaining follow-ups for patients with occult primary cancers who are at an elevated risk of develo** a metastasis or a second primary carcinoma outbreak is highlighted.
Full article

Figure 1
Open AccessCase Report
Eculizumab Treatment of Massive Hemolysis Occurring in a Rare Co-Existence of Paroxysmal Nocturnal Hemoglobinuria and Myasthenia Gravis
by
Ráhel Réka Bicskó, Árpád Illés, Zsuzsanna Hevessy, Gergely Ivády, György Kerekes, Gábor Méhes, Tünde Csépány and Lajos Gergely
Hematol. Rep. 2024, 16(2), 255-259; https://doi.org/10.3390/hematolrep16020025 - 19 Apr 2024
Abstract
►▼
Show Figures
The co-occurrence of myasthenia gravis (MG) and paroxysmal nocturnal hemoglobinuria (PNH) is rare; only one case has been published so far. We report a 63-year-old Caucasian female patient who was diagnosed with MG at the age of 43. Thymoma was also detected, and
[...] Read more.
The co-occurrence of myasthenia gravis (MG) and paroxysmal nocturnal hemoglobinuria (PNH) is rare; only one case has been published so far. We report a 63-year-old Caucasian female patient who was diagnosed with MG at the age of 43. Thymoma was also detected, and so it was surgically resected, which resulted in reasonable disease control for nearly 20 years. Slight hemolysis began to emerge, and then myasthenia symptoms progressed, so immunosuppressive therapy was started. Due to progressive disease and respiratory failure, the patient underwent plasmapheresis, and ventilatory support was stopped. Marked hemolysis was present, and diagnostic tests confirmed PNH with type III PNH cells. Her myasthenia symptoms aggravated, mechanical ventilation had to be started again, and due to the respiratory acidosis, massive hemolysis occurred. After two plasmapheresis sessions, the patient received eculizumab at 600 mg, resulting in prompt hemolysis control. After the second dose of the treatment, the patient was extubated. Still, due to their inability to cough, she developed another respiratory failure and pneumonia–sepsis, resulting in the patient’s death. This case highlights the rare association between these two serious diseases and similar immune-mediated pathophysiology mechanisms involving the complement system.
Full article
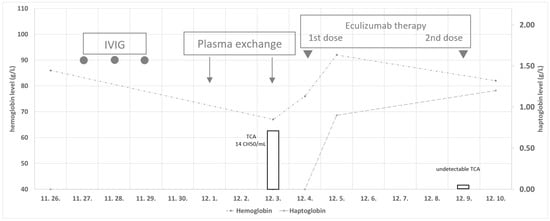
Figure 1
Open AccessReview
A 2024 Update on Menin Inhibitors. A New Class of Target Agents against KMT2A-Rearranged and NPM1-Mutated Acute Myeloid Leukemia
by
Anna Candoni and Gabriele Coppola
Hematol. Rep. 2024, 16(2), 244-254; https://doi.org/10.3390/hematolrep16020024 - 18 Apr 2024
Abstract
►▼
Show Figures
Menin inhibitors are new and promising agents currently in clinical development that target the HOX/MEIS1 transcriptional program which is critical for leukemogenesis in histone-lysine N-methyltransferase 2A-rearranged (KMT2Ar) and in NPM1-mutated (NPM1mut) acute leukemias. The mechanism of action of this new class of agents
[...] Read more.
Menin inhibitors are new and promising agents currently in clinical development that target the HOX/MEIS1 transcriptional program which is critical for leukemogenesis in histone-lysine N-methyltransferase 2A-rearranged (KMT2Ar) and in NPM1-mutated (NPM1mut) acute leukemias. The mechanism of action of this new class of agents is based on the disruption of the menin–KMT2A complex (consisting of chromatin remodeling proteins), leading to the differentiation and apoptosis of AML cells expressing KMT2A or with mutated NPM1. To date, this new class of drugs has been tested in phase I and II clinical trials, both alone and in combination with synergistic drugs showing promising results in terms of response rates and safety in heavily pre-treated acute leukemia patients. In this brief review, we summarize the key findings on menin inhibitors, focusing on the mechanism of action and preliminary clinical data on the treatment of acute myeloid leukemia with this promising new class of agents, particularly revumenib and ziftomenib.
Full article
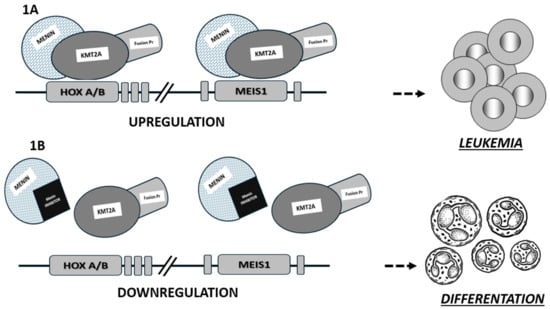
Figure 1
Open AccessCommunication
Post-Transplant Cyclophosphamide versus Anti-Thymocyte Globulin in Patients with Hematological Malignancies Treated with Allogeneic Hematopoietic Stem Cell Transplantation from Haploidentical and Matched Unrelated Donors: A Real-Life Experience
by
Bianca Serio, Gabriella Storti, Matteo D’Addona, Lidia Santoro, Camilla Frieri, Danilo De Novellis, Luana Marano, Giovanna De Santis, Roberto Guariglia, Ilenia Manfra, Eleonora Urciuoli, Serena Luponio, Serena Marotta, Denise Morini, Michela Rizzo, Fausto Palmieri, Nicola Cantore, Valentina Giudice, Antonio Maria Risitano and Carmine Selleri
Hematol. Rep. 2024, 16(2), 234-243; https://doi.org/10.3390/hematolrep16020023 - 17 Apr 2024
Abstract
►▼
Show Figures
Background: Post-transplant cyclophosphamide (PTCY) is widely used as graft versus host disease (GvHD) prophylaxis in allogeneic hematopoietic stem cell transplant (HSCT) recipients, with reported clinical benefits in patients who underwent transplant from a matched unrelated donor (MUD). However, real-life data on clinical efficacy
[...] Read more.
Background: Post-transplant cyclophosphamide (PTCY) is widely used as graft versus host disease (GvHD) prophylaxis in allogeneic hematopoietic stem cell transplant (HSCT) recipients, with reported clinical benefits in patients who underwent transplant from a matched unrelated donor (MUD). However, real-life data on clinical efficacy and safety of PTCY in haploidentical and MUD transplantations are still poor. Methods: In our real-life retrospective observational study, we included a total of 40 consecutive adult patients who underwent haploidentical or MUD HSCT for various hematological malignancies and who received PTCY (n = 24) or ATG (n = 16) as GvHD prophylaxis at Hematology Units from hospitals of Salerno and Avellino, Italy, and clinical outcomes were compared. Results: We showed protective effects of PTCY against disease relapse with the relapse rate after transplantation of 16% versus 50% in the ATG arm (p = 0.02). All-cause mortality was lower (36% vs. 75%; p = 0.02) and the 2-year overall survival was slightly superior in patients administered PTCY (61% vs. 42%; p = 0.26). Conclusions: We support the use of PTCY, even in a real-life setting; however, the optimization of this protocol should be further investigated to better balance relapse prevention and GvHD prophylaxis.
Full article
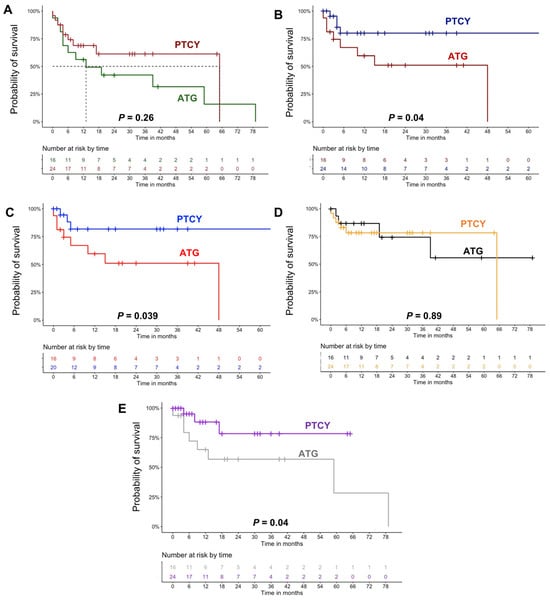
Figure 1
Open AccessArticle
Monitoring Humoral Response Following BNT162b2 mRNA Vaccination against SARS-CoV-2 in Hematopoietic Stem-Cell Transplantation Patients: A Single-Center Prospective Study along with a Brief Review of Current Literature
by
John V. Asimakopoulos, Eleni Lalou, George Seferlis, Maria Malliarou, Eliana Konstantinou, Ioannis Drandakis, Ioannis Vasilopoulos, Angeliki N. Georgopoulou, Anastasia Kopsaftopoulou, Alexandros Machairas, Alexia Piperidou, Anestis Karapaschalidis, Maria-Ekaterini Lefaki, Dimitrios Galopoulos, Maria-Panagiota Arapaki, Panagiota Petsa, Ekaterini Benekou, Marina P. Siakantaris, Athanasios G. Papavassiliou, Panagiotis Tsaftaridis, Panayiotis Panayiotidis, Theodoros P. Vassilakopoulos, Angeliki Papapanagiotou and Maria K. Angelopoulouadd
Show full author list
remove
Hide full author list
Hematol. Rep. 2024, 16(2), 220-233; https://doi.org/10.3390/hematolrep16020022 - 16 Apr 2024
Abstract
►▼
Show Figures
Data on antibody response (AR) after vaccination against SARS-CoV2 in hematopoietic stem-cell transplantation setting (HSCT) were initially scarce, mainly due to the exclusion of such patients from approval studies. Shortly after the worldwide application of vaccination against SARS-CoV-2 in vulnerable populations such as
[...] Read more.
Data on antibody response (AR) after vaccination against SARS-CoV2 in hematopoietic stem-cell transplantation setting (HSCT) were initially scarce, mainly due to the exclusion of such patients from approval studies. Shortly after the worldwide application of vaccination against SARS-CoV-2 in vulnerable populations such as patients with hematologic malignancies, limited single-center trials, including HSCT patients, were published. However, there was a great heterogeneity between them regarding the type of underlying malignancy, co-current treatment, type of vaccine, method of AR measurement, and time point of AR measurement. Herein, we present the results of a prospective study on AR after vaccination for SARS-CoV-2 using the BNT162b2 vaccine in a cohort of 54 HSCT recipients—mostly autologous from a single Unit—along with a broad review of the current literature. In our cohort, the AR positivity rate at 1 month was 80.8% and remained positive in 85.7% of patients at 3 months after vaccination. There were only nine non-responders, who were more heavily pretreated and more frequently hypogammaglobulinemic compared to responders. High antibody titers (AT), [AT ≥ 1000 U/mL], were detected in 38.5% and 30.6% of the patients at m1 and m3, respectively. A significant decline in AT between m1 and m3 was demonstrated—p < 0.0001; median AT1 and AT3 were 480.5 and 293 U/mL, respectively. A novel finding of our study was the negative impact of IgA hypogammaglobulinemia on response to vaccination. Other negative significant factors were treatment with anti-CD20 antibody at vaccination and vaccination within 18 months from HSCT. Our data indicate that HSCT recipients elicit a positive response to the BNT162b2 vaccine against SARS-CoV-2 when vaccinated at 6 months post-transplant, and vaccination should be offered to this patient population even within the post-pandemic COVID-19 era.
Full article

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics

Conferences
Special Issues
Special Issue in
Hematology Reports
State of the Art Papers from the XXXI IACRLD Symposium
Guest Editors: Ali Bazarbachi, Claudio Cerchione, Giovanni Martinelli, Giuseppe SaglioDeadline: 1 December 2024
Special Issue in
Hematology Reports
Personalized Therapies and Clinical Outcomes for Older Patients with Lymphoma
Guest Editor: Yamasaki SatoshiDeadline: 31 December 2024
Special Issue in
Hematology Reports
Modern Radiotherapy for the Treatment of Lymphoma and Myeloma: Integration with Systemic Treatment and Advanced Imaging Techniques
Guest Editors: Andrea Guerini, Michela Buglione Di Monale E BastiaDeadline: 20 January 2025